Review Article
Origin, Geographical Distribution, And Genetic Diversity of Crop Brassica Species: A Review
Ganesh G*
Department of Botany, M.A.L.D. Govt. Degree College Gadwal,Jogulamba Gadwal District, Telangana, India
*Corresponding author:Dr. G.Ganesh, Department of Botany, M.A.L.D. Govt. Degree College Gadwal, Jogulamba Gadwal District, Telangana, India. E-mail Id: ganijaann55@gmail.com
Copyright: © Ganesh G. 2026. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information:Submission: 03/01/2026; Accepted: 22/01/2026; Published: 24/01/2026
Abstract
The genus Brassica comprises some of the most economically important oilseed and vegetable crops cultivated worldwide. These crops contribute significantly to global food security, nutrition, and agricultural sustainability. The present paper provides a comprehensive synthesis of the origin, geographical
distribution, and genetic diversity of major crop Brassica species. Archaeobotanical and molecular evidence suggests that primary domestication of diploid Brassica species (B. rapa, B. nigra, and B. oleracea) occurred independently in the Mediterranean and Central Asian regions between 3000–5000 years BP.
These diploid species subsequently gave rise to amphidiploid species (B. juncea, B. napus, and B. carinata) through natural hybridization and polyploidization, as described by U’s Triangle. Genetic diversity studies using molecular markers (SSR, SNP, AFLP) reveal high intra-specific variation, particularly in B. rapa
(Nei’s gene diversity, H = 0.42–0.65; derived from genome-wide SNP analyses by Bird et al., 2020 and Park et al., 2023) [1] and B. juncea (H = 0.38–0.60; reported using SNP and GBS approaches by Hasan et al., 2023 and Gupta et al., 2024), reflecting wide ecological adaptation. Understanding the origin, distribution, and diversity of crop Brassicas is essential for crop improvement, conservation of genetic resources, and climate-resilient breeding programs.
Introduction
The genus Brassica (family Brassicaceae) includes a wide range of
cultivated crops used as vegetables, oilseeds, condiments, and fodder.
Major Brassica crops such as mustard, rapeseed, cabbage, cauliflower,
broccoli, and turnip are integral components of agricultural systems
across temperate and subtropical regions. India and China together
contribute more than 35% of the global production of rapeseed–
mustard, while Europe dominates vegetable Brassica cultivation.
(FAO,2022; FAOSTAT).
The remarkable morphological variation observed in Brassica crops, particularly within B. oleracea (cabbage, cauliflower, broccoli, kale), has made the genus a classical model for studying domestication, polyploidy, and crop evolution. Advances in cytogenetics and molecular biology have further clarified the evolutionary relationships among cultivated species, especially through the concept of U’s Triangle.
The remarkable morphological variation observed in Brassica crops, particularly within B. oleracea (cabbage, cauliflower, broccoli, kale), has made the genus a classical model for studying domestication, polyploidy, and crop evolution. Advances in cytogenetics and molecular biology have further clarified the evolutionary relationships among cultivated species, especially through the concept of U’s Triangle.
Objectives
The present study aims to:
1. Review the origin and domestication history of the major crop Brassica species.
2. Examine the global geographical distribution of cultivated Brassica species in relation to their centers of diversity and patterns of genetic variation.
3. Synthesize cytogenetic and molecular (genomic and SNP-based) evidence on genetic diversity and discuss its implications for crop improvement and conservation.
1. Review the origin and domestication history of the major crop Brassica species.
2. Examine the global geographical distribution of cultivated Brassica species in relation to their centers of diversity and patterns of genetic variation.
3. Synthesize cytogenetic and molecular (genomic and SNP-based) evidence on genetic diversity and discuss its implications for crop improvement and conservation.
Materials and Methods
This review is based on a comprehensive survey of the published
literature from 2010 to 2025, with a particular emphasis on recent
studies published between 2019 and 2025, to capture advances in
genomics and high-throughput marker technologies. Peer-reviewed
research articles, review papers, book chapters, and authoritative
databases were consulted using major scientific platforms, including
Scopus, Web of Science, PubMed, and Google Scholar.
Studies were selected based on the following criteria:
(i) Clear taxonomic identification of cultivated Brassica species
or their wild relatives;
(ii) Use of well-established molecular markers, including SSRs, SNP arrays, genotyping-by-sequencing (GBS), or wholegenome resequencing;
(iii) Adequate sample size and genome-wide coverage to reliably estimate genetic diversity parameters (e.g., H, Ho, π); and
(iv) Relevance to questions of origin, geographical distribution, domestication, or genetic diversity. Emphasis was placed on SNP-based and genomic studies due to their higher resolution and reproducibility.
Data from selected studies were synthesized qualitatively and quantitatively to compare diversity patterns across species and regions. FAOSTAT reports were additionally consulted to compile information on global production and distribution trends.
(ii) Use of well-established molecular markers, including SSRs, SNP arrays, genotyping-by-sequencing (GBS), or wholegenome resequencing;
(iii) Adequate sample size and genome-wide coverage to reliably estimate genetic diversity parameters (e.g., H, Ho, π); and
(iv) Relevance to questions of origin, geographical distribution, domestication, or genetic diversity. Emphasis was placed on SNP-based and genomic studies due to their higher resolution and reproducibility.
Data from selected studies were synthesized qualitatively and quantitatively to compare diversity patterns across species and regions. FAOSTAT reports were additionally consulted to compile information on global production and distribution trends.
Results and Discussion
Origin and Domestication:
The origin of cultivated Brassica species is closely linked to the
Mediterranean and Central Asian regions. The three primary diploid
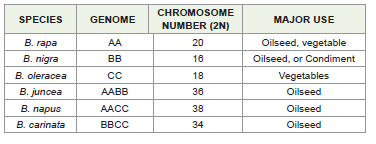
species—B.rapa (AA, 2n=20), B. nigra (BB, 2n=16), and B.oleracea
(CC, 2n=18)—represent the ancestral gene pools. Archaeological
evidence suggests that B.oleracea was domesticated along the
Mediterranean coast, while B.rapa originated in Central Asia and
later diversified in East Asia.The amphidiploid species arose through natural hybridization:
B. juncea (AABB, 2n=36) from B. rapa × B. nigra
B. napus (AACC, 2n=38) from B. rapa × B. oleracea
B. carinata (BBCC, 2n=34) from B. nigra × B. oleracea
B. carinata (BBCC, 2n=34) from B. nigra × B. oleracea
Geographical Distribution:
Crop Brassicas are widely distributed across Asia, Europe, North
America, and Australia. B. juncea dominates the Indian subcontinent,
accounting for nearly 80% of mustard cultivation in India. B. napus
is extensively grown in Europe, Canada, and China as a major oilseed
crop. Vegetable forms of B.oleracea are cultivated globally under
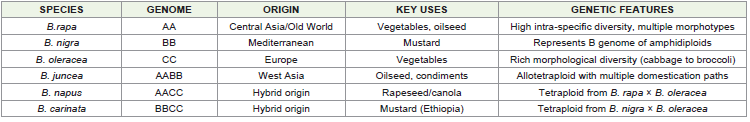
temperate climatic conditions.Among six cultivated brassicas, B.oleracea L. is a vegetable brassica, and the remaining five species are oilseed brassicas grouped into rapeseed and mustard. The origin of the cultivated Brassica species is as follows:
Brassica rapa (B.campestris L.): has a primary centre of diversity in the Indian gene centre, namely in the Himalayan
region (Hedge,1976) [2]. Russian workers, particularly Sinskaia
and Vavilov. Regarded Central Asia, Afganistan and the adjoining
northwest India as one of the independent centres of the origin of
oleiferous B. rapa. It is widely believed that brown sarson is the oldest
type (Singh 1958) [3] from which toria developed, probably in the
foothills of the Himalayas, as an early maturing type in response to
particular ecological requirements, and that yellow sarson probably
originated in the Indo-Gangetic Plains as a spontaneous mutant of
toria for superior quality. Yellow sarson was mentioned in Indian
Sanskrit documents from 1500 BC. The species B.rapa L. (2n = 2x =
20, genome AA) is one progenitor of the both amphidiploid B.juncea
(L.) Czern. (2n = 4x = 36; genome AA BB) and B.napus L. (2n = 4x =
38; genome AA CC).
Brassica nigra (L.): Koch was cultivated as a spice as early as 3000
BC and it has been probably evolved as a second crop plant from
weedy types. The origin may have taken place in the Mediterranean
area or Asia; it is now widespread throughout Europe, Africa, Asia,
India is the Far East (Hemingway, 1976) [4]. It has now been largely
replaced by B. juncea due to the latter better adapted to modern agrotechnology.
In India, it occupies only a limited area in Karnataka,
Tamil Nadu and the northern states. It is used as a condiment and
for pickle making. The species B.nigra L. Koch. (2n = 2x = 16; genome
BB) is one progenitor of both amphidiploids, B.carinata A. Braun.
(2n =4x =34; genome BB CC) and B.juncea L. (2n = 4x =36; genome
AA BB).
Brassica oleracea L.: is originated in the Mediterranean area and Western Europe. The hybridization of W-European forms with
forms from the E-Mediterranean area has resulted in a wide range
of variation. The species B.oleracea L. displays an interesting genetic
diversity, represented by 14 cultivated types (Dias 1995) [5] and 10
wild taxa in the B.oleracea cytodeme (Harberd 1972; Snogerup et al.
1990) [6,7]. The species B.olerucea L. (2n=2x=18, genome CC) is one
progenitor of the both amphidiploids B.carinata A. Braun. (2n =4x=
34; genome BB CC) and B.napus L. (2n=4x=30; genome AA CC).
Brassica juncea (L.): Czern. Is an amphidiploid species with the A genome of B.rapa L. and the B genome of B. nigra (L.) Koch.
Perhaps it is the oldest of the cultivated amphidiploids. Central
Asia-Himalayas is a primary centre of diversity for this species with
migration to China, India and the Caucasus (Hemingway 1976) [4].
The species has probably evolved in the Middle East, when its putative
diploid progenitors are sympatric (Prakash and Hinata 1980) [8-16]. It is highly polymorphic and includes both leafy and oleiferous
variants. B. juncea may have arisen several times, as secondary centres
of diversity exist in China, East India, and the Caucasus (Hemingway
1976) [4]. In India, B. juncea (L.) Czern is the predominant species
that accounts for nearly 80 per cent hectarage of the oilseed brassicas.
Brassica napus L.: is an amphidiploid species with the A genome of B.rapa L. and the C genome of B. oleracea. B. napus L. seems to be of relatively recent origin in the South West and Mediterranean
regions. The species evolved in South Europe and is divided into two
Nopobrassica (L.) Hanelt originated following hybridisation between
turnip and kale. Rape (B. napus L. ssp. napus), a valuable oil crop,
perhaps has a separate origin in the western Mediterranean areas.
B.napus L. lacks a true petiole, as does B. rapa L., but only partial
clasping of the stem occurs. The crop B. napus L.is mostly grown in
Canada, Europe, Japan and Chile as an oilseed and fodder crop. On
the Indian sub-continent, B. rapa L. and B. juncea (L.) Czern. still
predominate compared to the introduced B. napus L. material.
Brassica carinata: A. Braun is an amphidiploid with the B genome of B. nigra (L.) Koch and the C genome of B. oleracea L. have several characteristics that make it a suitable candidate for a winter crop in
Mediterranean countries. It is found and cultivated in North Eastern
Africa and Ethiopia in particular, and is believed to have originated
in the Ethiopian highlands (Vaughan 1956). Leaves are attached to
the stem with a true petiole. It is an important oil crop as well as a leaf
vegetable and shows promise agronomically in many other parts of
the world. Compared to other cultivated Brassica species, the range of
variation of B. carinata is not very high.
Genetic Diversity: Genetic diversity studies indicate substantial
variability within and among Brassica species. Molecular marker
analyses reveal that B.rapa exhibits the highest diversity due to its
wide ecological amplitude and multiple domestication events. In
contrast, B.napus exhibits relatively narrow genetic diversity (H
= 0.20–0.35), which is attributed to its recent origin and intensive
breeding. Conservation of wild relatives and landraces is therefore
critical for broadening the genetic base of cultivated Brassicas.
Importantly, these diversity patterns have direct implications for applied breeding and climate resilience. Species and populations exhibiting higher heterozygosity and nucleotide diversity, such as B. rapa and B. juncea, offer broader allelic pools for the selection of traits related to drought tolerance, heat stress, salinity resistance, and disease resistance. Conversely, the relatively narrow genetic base of B.napus underscores the need for targeted introgression from diverse germplasm, including landraces and wild relatives, to enhance adaptive capacity under changing climatic conditions. Genomic diversity information can thus guide parent selection, heterosis breeding, and pre-breeding programs aimed at developing resilient Brassica cultivars suited to future agro-climatic challenges.
Importantly, these diversity patterns have direct implications for applied breeding and climate resilience. Species and populations exhibiting higher heterozygosity and nucleotide diversity, such as B. rapa and B. juncea, offer broader allelic pools for the selection of traits related to drought tolerance, heat stress, salinity resistance, and disease resistance. Conversely, the relatively narrow genetic base of B.napus underscores the need for targeted introgression from diverse germplasm, including landraces and wild relatives, to enhance adaptive capacity under changing climatic conditions. Genomic diversity information can thus guide parent selection, heterosis breeding, and pre-breeding programs aimed at developing resilient Brassica cultivars suited to future agro-climatic challenges.
Conclusion
The genus Brassica represents an excellent example of crop
evolution through domestication, hybridization, and polyploidy.
The wide geographical distribution and rich genetic diversity of crop
Brassicas have enabled their adaptation to diverse agro-climatic
conditions. However, genetic erosion due to modern breeding
necessitates urgent conservation of traditional varieties and wild
relatives. A thorough understanding of origin, distribution, and
genetic diversity will support future breeding strategies aimed at
improving yield, quality, and stress tolerance in Brassica crops.
References
Citation
Ganesh G. Origin, Geographical Distribution, And Genetic Diversity of Crop Brassica Species: A Review. J Plant Sci Res. 2026;13(1): 292.