Review Article
Morphological and Phytopharmacological Investigations in Jasminum Species – A Review
Soorya SS1 and Sumitha VR2
1Department of Botany, Mahatma Gandhi College, Thiruvananthapuram Kerala, India.
2Department of Botany, NSS College, Pandalam, Kerala, India.
2Department of Botany, NSS College, Pandalam, Kerala, India.
*Corresponding author:Soorya SS, Research Scholar, Department of Botany, Mahatma Gandhi College, Thiruvananthapuram Kerala, India Email Id: sooryasindhu97@gmail.com
Copyright: © Soorya SS, et al. 2025. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information:Submission: 20/07/2025; Accepted: 11/08/2025; Published: 13/08/2025
Abstract
The genus Jasminum comprises deciduous shrubs and climbers belonging to the family Oleaceae and includes more than 200 species that are both cultivated and found growing wild worldwide. These species are valued for their traditional applications in medicine, perfumery, and cultural practices. Jasminum species are rich in diverse phytochemicals, including alkaloids, flavonoids, saponins, phenols, tannins, and glycosides. Bioactive constituents isolated from these species have been reported to exhibit therapeutic potential against cancer, metabolic disorders, infectious diseases, and inflammatory conditions. Pharmacological studies have demonstrated that Jasminum extracts possess antimicrobial, anti-inflammatory, anticancer, antioxidant, antiviral, antidiabetic, and anthelmintic activities. The essential oils of Jasminum are extensively used in perfumery and aromatherapy, while the flowers hold ceremonial and cultural significance in several countries. This review provides a comprehensive summary of current knowledge on the morphological, phytochemical, and pharmacological attributes of Jasminum species. It also identifies key bioactive compounds and phytopharmacological activities, emphasizing their
potential as valuable resources for drug discovery and therapeutic development.
Keywords:Jasminum species; Morphology; Phytochemicals; Pharmacological effects
Introduction
Herbal medicines and plant-based chemicals are widely used from
ancient period throughout the world and the traditional knowledge of
plants has become an important tool in the invention of new drugs and
nutraceuticals [1] The medicinal value of plants possess important
dimensions in the past few decades for the discovery diverse array of
secondary metabolites from plant extracts rather than minerals and
primary metabolites [2]. More than 80% individuals from developed
countries use traditional medicines and its compounds derived from
medicinal plants. Medicinal plants serve as one of the most valuable
sources for deriving herbal medicines [3]. Jasminum species are widely
associated with aromatic, pharmaceuticals, phytochemicals and
cosmetic industries [4]. Jasminum species are commonly cultivated
across various regions of India. The genus Jasminum, comprising
both shrubs and climbers, is taxonomically classified under the family
Oleaceae commonly known as the olive family and encompasses over
200 species globally. Of these, nearly 40 species have been
documented as naturally occurring or cultivated within the Indian
subcontinent. Jasmine is traditionally recognized as a culturally
significant floricultural crop in India. The major jasmine species
grown in the state are Kakada (Jasminum multiflorum Burm. F.), Sooji
Mallige (Jasminum auriculatum Vanl.) Gundu mallige (Jasminum
sambac Ait) and Jaji Mallige (Jasminum grandiflorum Linn). Jasmine
is grown for its highly scented flowers. The roots, leaves and flowers of
Jasminum plants are used for the treatment of diarrhoea, abdominal
pain, fever, conjunctivitis and dermatitis [5]. The extensive traditional
use and commercial importance of Jasminum species, comprehensive
analysis summarizing their phytochemical diversity, pharmacological
properties, and therapeutic applications remain limited. Hence, this
systematic review that consolidates existing knowledge, elucidates the
phytopharmacological activities of the genus, and identifies future
directions for their exploration in drug discovery and nutraceutical
development.
Plant Description:
Jasminum is the largest genus of the olive family Oleaceae with
∼200 species, which includes flowering shrubs and vines. Jasminum
species are widely considered as medicinal plant and has been used
since ancient times [6]. In Southern and Eastern parts of India
Jasmines are considered as the most important ornamental flowering
plant and widely cultivated for their appealing appearance and
pleasant fragrance. Jasminum species are cultivated for ornamental
gardening, landscape enhancement, the preparation of garlands, hair
adornment, and the extraction of essential oils. The basic chromosome
number of Oleaceae family is x=13 [7].Distribution:
The Jasminum genus is considered native to the warm climates of
tropical and temperate areas, with its distribution extending from
Portugal and The Canary Islands across Southern Europe to Taiwan,
Tahiti, and Australia. Jasmine is particularly valued for its fragrant
flowers and the extraction of essential oil. The Indo-Malayan region
is considered the centre of origin of Jasminum species and India
exhibiting significant genetic diversity within the genus. Although the
distribution of the Jasminum genus is globally tropical, a significant
concentration of species is found in India, China, and the Malayan
region. In India, the major jasmine-producing states are Tamil Nadu
and Karnataka [8].Morphological Characteristics:
Most Jasminum species possess climbing branches without
tendrils, although some species exhibit a bushy or creeping growth
habit. The leaves, which may be simple or compound, exhibit a variety
of shapes including acuminate, lanceolate, cordate, pointed, and oval.
Leaf coloration ranges from dark green to light green. The leaf texture
varies and can be rough, smooth, pubescent, glabrous, leathery,
or velvety, with either an even or uneven leaf base. The flowers of
Jasminum species are white, yellow, or very pale pink in colour, and
are strongly fragrant. They usually possess five petals, although the
number can range from four to nine. Flowers are usually produced in
cymose clusters comprising at least three blossoms, although single
flowers may occasionally appear at the ends of branchlets. Each flower
generally contains two locules and one to four ovules. The flower buds
exhibit various shapes, including conical-pointed, conical-rounded,
and ovate-rounded forms. The fruit of Jasminum is a two-lobed black
berry [9].Scientific Classification:
Kingdom: Plantae
Division: Magnoliophyta
Class: Magnoliopsida
Order: Lamiales
Family: Oleaceae
Genus: Jasminum [10]
Kingdom: Plantae
Division: Magnoliophyta
Class: Magnoliopsida
Order: Lamiales
Family: Oleaceae
Genus: Jasminum [10]
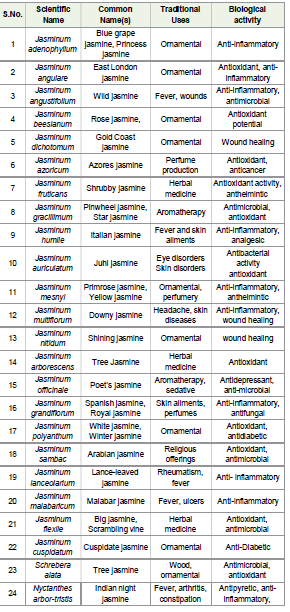
Table 1:Traditionally and Therapeutically Valuable Jasminum
Species [11]
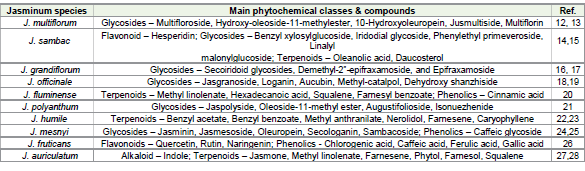
Phytochemical Activities:
Phytochemical analysis of leaf and flower extracts of Jasminum
multiflorum examined the presence of various bioactive compounds,
including tannins, sterols, cardiac glycosides, flavonoids, alkaloids,
and terpenoids [12]. The important phytochemical constituents
identified from this species are secoiridoids, which are derived from
iridoid-based bicyclic ring structures known as iridanes. The aqueous
extract of Jasminum multiflorum contains secoiridoid glycosides,
including multifloroside, hydroxyoleoside-11-methylester, 10-
hydroxyoleuropein, 10-hydroxyligustroside, jusmultiside, and
multiflorin [13]. Phytochemical estimation of Jasminum sambac
showed the presence of alkaloids phenols flavonoids, tannins,
saponin, and phytosterols [14]. Jasminum sambac was characterized
with a wide range of phytochemicals. Its major phytoconstituents
were iridoidal glycoside, benzyl xylosylglucoside, phenylethyl
primeveroside, linalyl malonylglucoside, dotriacontanoic acid,
hesperidin, oleanolic acid, daucosterol, and volatile compounds [15].
Chemical constituents isolated from various parts of Jasminum
grandiflorum include secoiridoid glycosides such as demethyl-2″-
epifraxamoside, 2″-epifraxamoside, and jasminanhydride. [16].
Phytochemical estimation of Jasminum grandiflorum revealed the
presence of secoiridoids, terpenoids, flavonoids, and tannins [17].
The primary phytochemical investigation of Jasminum officinale
leaves confirmed the occurrence of various bioactive compounds
including, flavonoids, alkaloids, glycosides, terpenoids, coumarins,
steroids, saponins, and tannins [18]. Flower buds of Jasminum
officinale were identified with six iridoid glycosides as aucubin,
loganin, jasgranoside B, deacetyl asperulosidic acid, methylcatalpol,
and dehydroxyshanzhiside [19]. Phytochemical examination of the
chloroform and methanolic extracts of Jasminum fluminense revealed
the presence of alkaloids, flavonoids, tannins, and glycosides. Analysis
of the chloroform and methanolic leaf extracts indicated the presence
of terpenoids, phenolic compounds, methyl linolenate, hexadecanoic
acid, linolenic acid, squalene, and D-α-tocopherol. Similarly,
phytochemical screening of the chloroform and methanolic flower
extracts showed the presence of lipids, terpenoids, and alkaloids,
including hexadecanoic acid, eicosanoic acid, linolenic acid, linoleoyl
chloride, (2E,6E)-farnesylbenzoate, hexatriacontane, glucose
benzyloxime pentaacetate, and phenylethanolamine.Phytochemical assessment of leaves and flowers of Jasminum
polyanthum revealed the presence of alkaloids, phenols, quinones,
saponins, and terpenoids. The important phytochemical constituents
are from secoiridoid class of compounds - jaspolyside, oleoside-11-
methyl ester, 7,11-oleoside dimethyl ester, methyl glucooleoside,
augustifolioside, oleuropein, and isonuezhenide [21]. Preliminary
phytochemical screening of the extract of Jasminum humile confirmed
the presence of several bioactive compounds, including alkaloids,
flavonoids, glycosides, tannins, saponins, phenolic compounds,
coumarins, steroids, anthraquinones, betacyanins, carbohydrates,
fixed oils, and resins [22]. Gas chromatography–mass spectrometry
analysis of essential oils extracted from freshly collected flowers of
Jasminum humile revealed the presence of seventeen distinct chemical
constituents, predominantly belonging to the terpenoid and phenolic
classes. Esters and sesquiterpenes were the main constituents. Esters
formed the major class of compounds, and included benzyl acetate,
benzyl benzoate, benzyl salicylate, methyl anthranilate, methyl
jasmonate, and methyl epijasmonate. The sesquiterpene fraction was
primarily composed of nerolidol, farnesene, and caryophyllene [23].
Caffeic glycoside, secoiridoid glucoside, and flavonoids were the
major phytochemicals isolated from the leaves of Jasminum mesnyi.
Several glucosides have been isolated from the methanolic leaf extract
including hydroxyljasmesosidic acid, jasminin, jasmoside,
jasmesoside, oleuropein, secologanin, sambacoside, and jasminin.
[24]. The leaves of Jasminum mesnyi contain essential oil, with
coumarin identified as the main component. The oil also contains high
amounts of monoterpenols such as linalool, terpineol, and geraniol
[25]. Phytochemical and biological studies were conducted in the
flowering branches of Jasminum fruticans shown the presence of
phytochemicals such as phenols, flavonoids and phenolic acid.
Phenolic profiling of J. fruticans extracts by LC/ESI-MS/MS indicated
the presence of p-coumaric acid, ferulic acid, hyperoside, rutin,
quercetin, naringenin, chlorogenic acid, caffeic acid, and gallic acid
[26]. Alkaloids, glycosides, terpenoids, flavonoids, steroids, tannins,
and phenolic compounds were identified in the phytochemical
analysis of Jasminum auriculatum [27]. GC-MS analysis of flower
absolutes from Jasminum auriculatum revealed the presence of
several lipid derivatives, terpenoids, and alkaloids including methyl
hexadecanoate, (Z)-jasmone, methyl linolenate, indole, (2E,6E) α-
farnesene, (E)-phytol, (2E,6E)-farnesol, 2,3-epoxy squalene,
squalene, δ-jasmine lactone, and nerolidol [28].
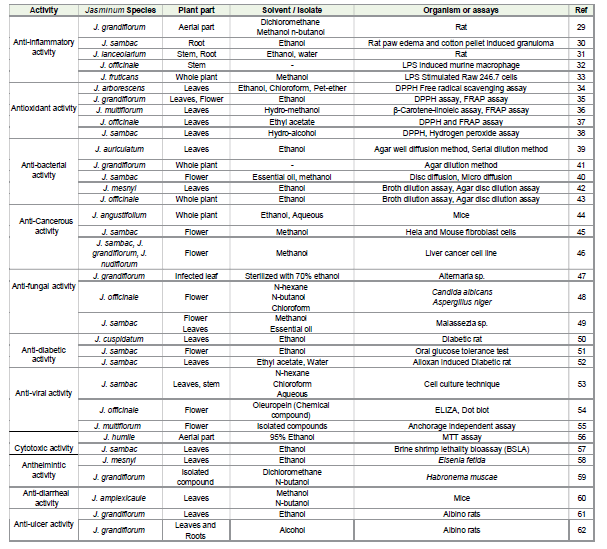
Pharmacological Properties:
Anti-inflammatory activityThe anti-inflammatory potential of the total methanolic extract from the aerial parts of Jasminum grandiflorum L. (JTME) was investigated using two experimental rat models-acetic acid-induced ulcerative colitis and adjuvant-induced arthritis. In vitro antiinflammatory activity of JTME methanolic fractions was tested calorimetrically and described the inhibitory COX and LOX activities were determined by using colorimetric inhibitor screening assay kits. The anti-inflammatory activity of JTME was shown in a dose-dependent manner at 400 mg/kg. It lowered the levels of proinflammatory cytokines such as IFN-γ, TNF-α, IL-1, IL-6 and MPO in the intestines and also protected the tight junctions of intestinal cells by maintaining the levels of claudin and occludin [29]. The antiinflammatory activity of Jasminum sambac was analysed using carrageenan-induced rat paw edema and cotton pellet-induced granuloma. The ethanolic root extract of J. sambac demonstrated a significant anti-inflammatory effect against carrageenan-induced inflammation at all dose levels, with the highest effect observed at 400 mg/kg. A significant reduction in granuloma formation was observed at 200 and 400 mg/kg in the cotton pellet-induced inflammation model [30].
Evidence indicates that 70% ethanolic and aqueous extracts of
Jasminum lanceolarium (EJL) exhibit significant anti-inflammatory
activity, as demonstrated by the suppression of COX-2 and 5-LOX
expression in carrageenan-induced inflamed rats. The extracts of J.
lanceolarium showed strong anti-inflammatory effect at 400 mg/kg.
Eleven compounds were isolated from the active extracts, including
six lignanoids and five iridoids, which exhibited significant antiinflammatory
activity with IC₅₀ values ranging from 1.76 to 5.22 mg/
mL when tested for their ability to inhibit phospholipase A₂. The antiinflammatory
effect was attributed to their ability to inhibit the
breakdown of membrane phospholipids and reduce the production of
both COX-2 and 5-LOX, thereby lowering prostaglandin release [31].
Compounds isolated from Jasminum officinale, including the
identified sesquiterpenoids Jasminol A, G, and H (nor-cinalbicanetype)
and Jasminol B (eremophilene-type), were analyzed to evaluate
the anti-inflammatory activity of the stem extract. The evaluation
performed using lipopolysaccharide (LPS)-induced murine
macrophage RAW264.7 cells. All four compounds showed moderate
inhibition of LPS-induced nitric oxide (NO) production and exhibited
low cytotoxicity, with CC₅₀ values greater than 200 μm [32]. The antiinflammatory
effects of the extract of Jasminum fruticans were
investigated along with two other species, Centaurea pichleri subsp.
pichleri and Conyza canadensis, by measuring the levels of tumour
necrosis factor-α (TNF-α), interferon-γ (IFN-γ), nitric oxide (NO),
and prostaglandin E₂ (PGE₂) in RAW 264.7 macrophage cells. The
methanolic extract of J. fruticans was found to be less active in antiinflammatory
action compared with the other two plant extracts. The
activity of J. fruticans in the control group was recorded as 11 ± 9 μM,
while in the LPS-treated group it was 4.28 ± 2.41 μM. The effects
of the J. fruticans extract on TNF-α, IFN-γ, PGE₂, and NO levels were
estimated as ± 2109.82, ± 115.63, ± 2121.62, and ± 55.72 μM,
respectively [33].
Antioxidant activity:
The antioxidant activity of Jasminum arborescens leaf extracts
prepared in petroleum ether, chloroform, and ethanol has been
evaluated in vitro. The antioxidant potential of the solvent extracts at
varying concentrations was evaluated using Ferric Reducing
Antioxidant Power (FRAP) and DPPH free radical scavenging assays.
The ethanolic extract showed higher antioxidant activity compared
with the chloroform and petroleum ether extracts [34]. The ethanolic
leaf and dried flower extracts of Jasminum grandiflorum showed
potent DPPH radical scavenging activity with an IC₅₀ value of 15 μg/
mL, comparable to that of the standard ascorbic acid (IC₅₀ = 12 μg/
mL). The extracts also showed nitric oxide radical scavenging activity
with an IC₅₀ of 98 μg/mL, comparable to that of curcumin (IC₅₀ = 92
μg/mL). In addition, the extracts also exhibited strong reducing
power, with an IC₅₀ of 19.5 μg/mL, comparable to quercetin (IC₅₀ =
15.5 μg/mL) [35].The hydromethanolic leaf extract of Jasminum multiflorum
(Burm. f.) Andrews was analyzed for its antioxidant potential.
The antioxidant activity was evaluated using the Ferric Reducing
Antioxidant Power (FRAP) assay and the β-carotene–linoleic acid
assay. At a concentration of 75 μg/mL, J. multiflorum extract showed
strong antioxidant activity with 68.23 ± 0.35% inhibition in the β-
carotene-linoleic acid assay and a TEAC value of 60.30 ± 0.60 μmol
Trolox g⁻¹ in the FRAP assay [36]. The antioxidant properties of
the ethyl acetate extract of Jasminum officinale L. leaves have been
investigated. Antioxidant activity was evaluated using the DPPH
radical scavenging assay and FRAP assay. The ethyl acetate fraction
(EAF) of J. officinale exhibited strong DPPH scavenging activity, with
an IC₅₀ value of 33.85 ± 1.09 μg/mL. The FRAP assay revealed an IC₅₀
value of 15.14 ± 0.25 μM (expressed as Fe2+ equivalents). In addition,
the EAF showed significant α-amylase inhibitory activity, ranging
from 13.25% to 74.51%, with an IC₅₀ value of 47.40 ± 0.29 μg/mL
[37]. The free radical scavenging activity of the 70% hydroalcoholic
leaf extract of Jasminum sambac was evaluated using DPPH, nitric
oxide, and hydrogen peroxide radicals. The inhibitory concentration
(IC₅₀) in the DPPH assay was 122 μg/mL for the extract. The total
antioxidant capacity and reducing power were 155.40 μg/mL and
44.28 μg/mL, respectively. The minimum inhibitory concentrations
for nitric oxide and hydrogen peroxide were 173.94 μg/mL and
125 μg/mL, respectively. The extract showed higher free radical
scavenging activity against hydrogen peroxide compared with nitric
oxide and DPPH, and exhibited moderate reducing power and total
antioxidant capacity [38].
Antibacterial activity:
Antimicrobial activity of Jasminum auriculatum leaves was
evaluated using the agar well diffusion method, and MIC values were
determined by the serial dilution method. The ethanolic extract of J.
auriculatum exhibited inhibitory effects against all tested
microorganisms. The extent of inhibition varied among the
organisms, with zones of inhibition ranging from 11 to 16 mm.
Ciprofloxacin and fluconazole (100 μg/mL) showed inhibition zone
diameters ranging from 15–24 mm and 16–17 mm, respectively.
The extract demonstrated strong antibacterial activity against
Pseudomonas aeruginosa, with a MIC of 0.78 mg/mL and a zone of
inhibition of 16.65 ± 0.6 mm [39].The antibacterial activity of the essential oil and methanolic
extract of Jasminum sambac flowers was evaluated against E. faecalis,
S. enterica, B. cereus, E. coli, and S. pyogenes, using microdilution and
disc diffusion techniques. Both extracts exhibited inhibitory effects
against Gram-positive and Gram-negative bacteria. The essential oil
showed a broader inhibition zone (8–41 mm) than the methanolic
extract (7–17 mm). In particular, the essential oil demonstrated strong
bactericidal activity against E. faecalis, with a minimum inhibitory
concentration (MIC) of 4 μg/mL [40]. The antimicrobial efficacy of
Jasminum grandiflorum absolute was assessed against selected
Gram-positive and Gram-negative bacterial strains, as well as the
fungal pathogen, Candida albicans, using agar diffusion and agar
dilution assays. Jasmine absolute showed moderate to strong
antimicrobial activity against Gram-negative bacteria such as P.
aeruginosa, E. coli, Salmonella spp., and Klebsiella spp., as well as the
Gram-positive bacterium E. faecalis [41]. The ethanolic leaf extract
of Jasminum mesnyi was studied for its antibacterial activity using
broth dilution and agar disc diffusion assays. Significant growth
inhibition of Vibrio parahaemolyticus and Aeromonas hydrophila
was observed with the diethyl ether fraction, producing inhibition
zones of 19 mm and 17 mm, respectively, at a concentration of
250 μg/mL. The disc diffusion assay demonstrated growth inhibition
against Gram-negative bacteria (A. hydrophila, V. parahaemolyticus,
Escherichia coli) and Gram-positive bacteria (Bacillus anthracis,
Bacillus subtilis, Staphylococcus aureus) on agar plates. The hexane
fraction (HF) also showed inhibitory effects on the growth of B.
anthracis, B. subtilis, and S. aureus. [42].
In vitro antibacterial activity of ethanolic extracts from the
flowers, stems, leaves, and roots of Jasminum officinale was assessed
against Enterococcus faecalis, Staphylococcus aureus, Escherichia
coli, and Pseudomonas aeruginosa using broth dilution and agar
diffusion methods. The flowers, stems, and leaves showed a minimum
inhibitory concentration (MIC) of 2 mg/mL against all bacteria. The
root extract had an MIC of 2 mg/mL against P. aeruginosa and 4 mg/
mL against E. faecalis, S. aureus, and E. coli. All extracts showed
significant antibacterial activity in the agar diffusion assay [43].
Anti-cancerous activity:
The whole plant of Jasminum angustifolium was studied for its
anticancer effect. Male Swiss albino mice were used to evaluate the
anticancer efficacy of ethanolic (EEJA) and aqueous (AEJA) extracts
of J. angustifolium against Ehrlich ascites carcinoma (EAC)-induced
tumors. The average life span of the EAC tumor control group was
18.2 ± 1.3 days. In contrast, oral administration of EEJA and AEJA at
a dose of 500 mg/kg increased the average life span to 39.4 ± 0.5 days
and 36.7 ± 0.7 days, respectively. Treatment with 5-fluorouracil (5-
FU) resulted in an average life span of 37.5 ± 1 day, confirming its
potent antitumor effect. A considerable reduction in body-weight gain
in animals treated with EEJA and AEJA further supported their
antitumor potential [44]. The anticancer activity of Jasminum sambac
has been evaluated in Swiss albino mice induced with Dalton’s ascites
lymphoma. The methanolic flower extract of J. sambac exhibited
dose-dependent inhibition of cancer cell proliferation in both HeLa
and mouse fibroblast cell lines. The IC₅₀ values for cancer and normal
cells were determined to be 93.8 μg/mL and 123.3 μg/mL respectively,
at concentrations ranging from 25 to 400 μg/mL. significant
improvements in blood profiles and reductions in AST, ALT, ACP,
ALP, and LDH levels were observed following oral administration of
the methanolic extract at a dose of 100 mg/kg body weight, supporting
the anticancer potential of Jasminum sambac [45].Methanolic extracts from five different Jasminum species-
Jasminum grandiflorum, Jasminum azoricum, Jasminum sambac
(single-flower), Jasminum sambac (double-flower), and Jasminum
nudiflorum were investigated for their potential anticancer properties
against a liver cancer cell line (HepG2). The percentage inhibition and
concentration-dependent response were evaluated for J. azoricum
treated HepG2 cells. At a concentration of 10 μg/mL, cell viability was
41.85 ± 0.89%, while at 100 μg/mL, it was reduced to 2.25 ± 0.10%.
These findings indicate that J. azoricum exhibits potent anticancer
activity and effectively inhibiting HepG2 cell proliferation at both
concentrations [46].
Antifungal activity:
Plant extracts of Jasminum grandiflorum were screened for
antifungal activity under in vitro conditions. Healthy leaf tissues,
along with adjacent infected tissues, were surface-sterilized using
ethanol and rinsed with sterile distilled water. The disinfected
leaf segments were inoculated onto potato dextrose agar (PDA)
medium. Leaves of J. grandiflorum showed dark-coloured lesions
with concentric rings. The isolated pathogen produced brownish
mycelium and conidia arranged in chains, with both vertical and
horizontal septations. Morphological analysis identified the pathogen
as belonging to the genus Alternaria, and molecular characterization
using primers AaF and AaR confirmed the species-level identity as
Alternaria alternata [47]. The antifungal effects of flower extracts
from Jasminum officinale was evaluated against Candida albicans and
Aspergillus niger. The n-hexane fraction exhibited minimal antifungal
activity, with zones of inhibition measuring 2.1 ± 1.3 mm and 3.2
± 1.8 mm, respectively. The n-butanol fraction demonstrated greater
antifungal activity than the standard drug against C. albicans, with a
zone of inhibition of 20.9 ± 1.2 mm. Moderate antifungal efficacy was
observed in the chloroform fraction, with inhibition zones measuring
13.1 ± 1.3 mm against C. albicans and 12.3 ± 0.6 mm against A. niger
[48].The antifungal activity of Jasminum sambac against Malassezia
species was evaluated using broth microdilution and disc diffusion
methods. Essential oils and methanolic extracts of the flowers and
leaves were prepared using solvent extraction and hydrodistillation
techniques. Sabouraud dextrose agar was used to culture skin samples
collected from individuals with dandruff, and fungal growth was
confirmed microscopically using the Tween assimilation test. Two
Malassezia species were isolated and identified based on their
morphology and ability to assimilate Tween. The leaf and flower
extracts of J. sambac showed antifungal activity, with inhibition
zones of 11.10 ± 1.92 mm, 12.90 ± 1.68 mm, and 13.06 ± 0.26 mm.
The minimum inhibitory concentrations (MICs) ranged from 80 to
160 mg/mL. The study concluded that Malassezia-associated skin
infections can be effectively treated with J. sambac extracts [49].
Antidiabetic activity:
Streptozotocin-induced diabetic rats were treated with the
ethanolic extract of Jasminum cuspidatum leaves. A significant
reduction in blood glucose levels was observed with the extract
at doses of 200 mg/kg and 400 mg/kg compared with the diabetic
control group. Treatment with glibenclamide (4 mg/kg) also produced
a significant decrease in blood glucose levels. The extract at 200 mg/kg
showed less glucose-lowering activity compared with the 400 mg/kg
dose and glibenclamide [50]. The antidiabetic potential of the
ethanolic flower extract of Jasminum sambac has been evaluated
through glucose tolerance studies. The extract was tested using oral
glucose tolerance tests as well as alloxan and streptozotocin-induced
diabetes models. In all models, animals treated with the extract
showed significantly lower blood glucose levels compared with the
diabetic control group. However, the antidiabetic activity of the J.
sambac extract was found to be less effective than the standard drug
[51].Ethyl acetate and aqueous extracts of Jasminum sambac leaves
were tested in alloxan-induced diabetic rats. The aqueous extract (300
mg/kg) produced a significant reduction in plasma glucose levels. The
ethyl acetate extract (EAE) was less effective compared with the aqueous
extract, whereas glibenclamide (10 mg/kg) significantly reduced
blood glucose levels compared with the diabetic control group [52].
Antiviral activity:
The n-hexane, chloroform, and aqueous leaf and stem extracts of
Jasminum sambac were analysed for their viricidal activity against
the foot-and-mouth disease virus using cell culture techniques.
The n-hexane extract showed no viricidal activity, whereas the
alcoholic and aqueous extracts exhibited potential antiviral activity at
concentrations of 1000–2000 μg/mL. The chloroform extract was
cytotoxic at 1000 μg/mL and 2000 μg/mL in BHK-21 cells, but
antiviral activity was observed at lower concentrations of 125 μg/mL
and 250 μg/mL. The antiviral potential of J. sambac was attributed to
oleuropein, a chemical constituent in the flowers known to inhibit the
secretion of hepatitis B surface antigen (HBsAg) [53].Oleuropein, a phytochemical extracted from the flowers of Jasminum officinale, was investigated for its antiviral effects against the HepG2 2.2.15 cell line (hepatitis B virus) and for its impact on duck hepatitis B virus (DHBV) replication in ducklings. ELISA was used to measure the concentrations of hepatitis B e antigen (HBeAg) and hepatitis B surface antigen (HBsAg) in the cell culture medium, while DHBV levels in duck serum were analyzed using a dot blot assay. Oleuropein inhibited the secretion of HBsAg in HepG2 2.2.15 cells in a dose-dependent manner, with an IC₅₀ value of 23.2 μg/mL. It also reduced viremia in DHBV-infected ducks [54]. The antiviral activity of compounds isolated from Jasminum multiflorum was assessed against hepatocellular carcinoma cells infected with hepatitis C virus (HCV). The antiviral efficacy was validated using anchorage independent assays, transwell migration assays, and cell-spreading assays. The flower extract of J. multiflorum demonstrated a selective antiviral effect on HepG2 and Huh-7 cell lines. Treatment with the leaf and flower extracts of J. multiflorum resulted in significant reductions in viral load, with decreases of 80.6 ± 2.1% and 91 ± 0.8%, respectively [55].
Cytotoxic activity:
The Cytotoxic activity of a 95% ethanolic extract from Jasminum
humile aerial parts was assessed against MCF-7, HepG-2 and THP-
1 cell lines using the MTT assay, with doxorubicin employed as
the positive control. The extract exhibited cytotoxic effects in a
concentration-dependent manner. High cytotoxic potential was
observed in the ethanolic extract of Jasminum humile, as evidenced
by its lowest IC₅₀ values recorded against THP-1 (46.63 μg/mL) and
HepG-2 (59.47 μg/mL) cell lines. These results suggest that aerial
parts of Jasminum humile possess strong cytotoxic potential [56].The dried leaves of Jasminum sambac (L.) were evaluated for
cytotoxicity using a brine shrimp lethality bioassay. The crude
ethanolic extract showed strong activity against Artemia salina, with
LC₅₀ and LC₉₀ values of 50 μg/mL and 100 μg/mL, respectively. The
extract caused concentration-dependent mortality in brine shrimp
nauplii, indicating potent cytotoxic properties [57].
Anthelmintic activity:
The anthelmintic activity of the ethanolic leaf extract of Jasminum
mesnyi was evaluated using adult Indian earthworms (Eisenia fetida).
The study demonstrated that the plant extract at concentrations of 20
mg/mL and 40 mg/mL induced paralysis and death in the worms.
Distilled water was used as the control, while Albendazole (10 mg/
mL) served as the reference drug. The results demonstrated that the
extract exhibited significant anthelmintic activity [58].The dichloromethane (DCM-F) and n-butanol (BuOH-F) fractions of Jasminum grandiflorum were separated and evaluated for their anthelmintic activity. The isolated compounds were tested against two groups of helminths—cestodes and arthropods. Four main compounds were found in the most active BuOH-F fraction: two flavonoids (kaempferol-3-O-neohesperoside and rutin) and two secoiridoid glycosides (oleuropein and ligstroside). In the BuOH-F fraction, rutin exhibited the strongest anthelmintic activity at 41.04 μg/mL against H. muscae adult worms. These findings confirmed the anthelmintic potential of Jasminum grandiflorum L. [59].
Antidiarrheal activity:
The methanolic leaf extract and its fractions of Jasminum
amplexicaule were evaluated for antidiarrheal activity using
various experimental models in mice, including castor oilinduced,
magnesium sulphate-induced, anti-enteropooling, and
gastrointestinal motility assays. The methanolic extract (ME)
demonstrated significant antidiarrheal activity at doses of 100, 200,
and 400 mg/kg. The n-butanol fraction (BUF) exhibited lower activity
than the methanolic extract [60].Anti-ulcer activity:
The hydroalcoholic extract of Jasminum grandiflorum L.
leaves was evaluated for anti-ulcer activity using an aspirin and
pylorus ligation-induced ulcer model in albino rats. The extract
was administered orally at doses of 100 and 200 mg/kg. Treatment
with the extract resulted in a significant reduction in the volume of
gastric juice, total acidity, free acidity, and ulcer index, along with an
increase in gastric pH. A dose-dependent and statistically significant
(P < 0.01) decrease in the ulcerative lesion index was observed when
compared to the standard drug, omeprazole (30 mg/kg, orally). These
findings indicated the gastroprotective potential of J. grandiflorum L.
in experimentally induced gastric ulcers [61].The anti-ulcer activity of alcoholic extracts from the leaves and roots of Jasminum grandiflorum was evaluated in albino rats using aspirin- and pylorus ligation-induced gastric ulcer models. Both leaf and root extracts exhibited significant anti-ulcer effects, as evidenced by increased gastric pH and reduced gastric juice volume, total acidity, and free acidity [62].
Conclusion
This review examines the morphological and phytopharmacological aspects of Jasminum species, highlighting their importance as both ornamental and medicinal plants for human
use. Jasminum species are distributed pantropically, with the majority
concentrated in India, China, and Malaya. Native to tropical
and subtropical regions, jasmines are commercially cultivated in
countries such as India, Thailand, China, and the Philippines for their
fresh flowers. These species are rich in a wide array of phytochemicals,
including alkaloids, tannins, sterols, terpenoids, flavonoids, cardiac
glycosides, saponins, phytosterols, and various bioactive compounds
such as iridoids, secoiridoids, lactones, and secoiridoid glycosides—
namely oleuropein, secologanin, multifloroside, oleopolyanthoside,
hydroxyoleoside 11-methyl ester, augustifolioside, multiflorin,
sambacosides, jaspolyside, jaspofoliamoside, jaspolinaloside,
jusmultiside, jasmoside, and jasminin. Pharmacological studies on
Jasminum species have revealed their diverse biological activities,
including anti-inflammatory, antioxidant, antimicrobial, antidiabetic,
anti-aging, antiviral, antidiarrheal, anticancer, and cytotoxic
properties. These bioactive constituents possess promising potential
for the development of standardized formulations such as
nutraceuticals, phytopharmaceuticals, and herbal therapeutics. In
addition to their medicinal value, Jasminum essential oils are widely
used in aromatherapy, perfumery, and the cosmetic industry due to
their fragrance and therapeutic benefits. Exploration of novel delivery
systems and commercialization strategies could not only enhance the
clinical applicability of Jasminum phytoconstituents but also expand
their potential in global healthcare and economic sectors.
Acknowledgement
I am grateful to my mentor and colleagues for their constructive
suggestions and valuable insights during the preparation of this
review paper. I also extend my sincere thanks to the library and online
databases of MG College, Thiruvananthapuram, Kerala, for providing
access to relevant scientific literature.
References
Citation
Soorya SS, Sumitha VR. Morphological and Phytopharmacological Investigations in Jasminum Species – A Review. J Plant Sci Res. 2025;12(2): 286