Research Article
In vitro Regeneration of Dendrobium terminale, a CITES-Listed Orchid for Sustainable Conservation
Dutta S and Anjalika Roy*
Department of Botany (DST-FIST and UGC-DRS SAP-II) Visva-Bharati, Santiniketan, India
*Corresponding author:Anjalika Roy, Department of Botany (DST-FIST and UGC-DRS SAP-II) Visva-Bharati, Santiniketan, India. E-mail Id: anjaika.roy@visva-bharati.ac.in
Copyright: © Dutta S, et al. 2025. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information: Submission: 02/06/2025; Accepted: 23/07/2025; Published: 26/07/2025
Abstract
The present study develops an efficient and reproducible protocol for the rapid in vitro mass propagation of the critically endangered epiphytic orchid Dendrobium terminale Par. & Rchb. Juvenile seedlings were produced from seed suspension culture. Dendrobium terminale is included in Appendix II of
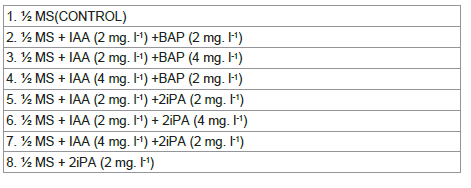
CITES (The Convention on International Trade in Endangered Species of Wild Fauna and Flora), showing its alarming position in natural habitat, ultimately emphasizing the need for its multiplication and conservation. Half MS media alone or supplemented with a combination of various concentrations of growth
regulators: 6-Benzyl-aminopurine, BAP (2 and 4mg/L), 2iPA and Indole acetic acid, IAA (2mg/L and 4mg/L), were used, which produced callus, PLB, multiple shoots and leaves. The concentrations and combinations of auxin and cytokinin have a significant impact on plant growth, with certain combinations resulting
in synergistic effects. It has been observed that BAP (4mg /L) and IAA (2 mg/L) were found to be most effective in promoting plant growth among the different combinations and concentrations of BAP, 2iPA, with IAA. The BAP (4 mg l-1) and IAA (2 mg l-1) recorded the highest number of PLB formation and significant
leaf growth. The combination of IAA (2 mg l-1) and 2iPA (2 mg l-1) also promotes a higher percentage of PLB formation (75%) without necrosis. Whereas higher concentrations of cytokinin (e.g., 2iPA (4 mg l-1) tend to favour more callus formation.
Keywords:In vitro; Dendrobium terminale; Callus; PLB; IAA (indole acetic acid); BAP (benzyl aminopurine); 2iPA
Introduction
Orchid plants, which are members of the Orchidaceae family, are
renowned for their exquisite flowers and distinctive morphological
features. The orchidaceae family is quite large, with 27,000 accepted
species and more than 31,000–35,000 species are estimated to exist
[1]. Orchids reign throughout the world, in both high alpine and
tropical regions. They can be classified as terrestrial, epiphytic, or
saprophytic depending on the kind of habitat they grow in, except
aquatic systems. Many diverse civilizations and tribes use orchids for
their decorative purposes, and others use them as food and as herbal
remedies [2].
Among flowering plants, orchids are the most vulnerable species
worldwide. Orchid species are severely threatened due to a variety
of factors, including over exploitation, illegal trading, land invasion,
and climate change [3]. They receive a variety of pollinators, like as
insects, tiny birds, and even bats, in addition to more light and air.
Over the millennia, this special relationship between orchids and their
pollinator has changed, giving the floral parts incredible proportions
and shapes[4].e germination rate of orchid seeds in nature is
extremely low due to their small size, non-endospermic origin, and
reduced embryo encased in a somewhat translucent covering [4].
In addition to their challenging germination process, orchids have
an extremely lengthy life cycle when left in their natural habitat. These
plants need five to ten years to bloom and set fruitful seeds. Orchid
vegetative propagation is likewise a very slow process, so for large-
scale production can be done by in vitro techniques. Nonetheless, a
lot of horticultural plants are originally cultivated from tissue culture
(rather than seeds) and maintained in highly enriched media, which
eliminates the requirement for a symbiotic fungus [5]. It is now
acknowledged that the plant tissue culture approach offers a viable
substitute for large-scale orchid propagation and conservation
of vulnerable, endangered, and threatened species. With these
techniques, a large number of identical clones can be grown from a
single protocorm or shoot tip explant [6].
Dendrobium Sw. is one of the three largest genera in the family
Orchidaceae, with about 800-1500 species worldwide. The majority
of Dendrobium species are terrestrial, very few are epiphytes in
primary forests, and are less frequent lithophytes with worldwide
distribution, except the coldest and driest areas of the world [7].
About 30 species have shown common distributional patterns and are
present in almost all the states of North-East India [8]. Among them,
Dendrobium terminale are is included in Appendix II of CITES (The
Convention on International Trade in Endangered Species of Wild
Fauna and Flora), where its status is now threatened with extinction,
which means strict regulation in trading is required for their survival
[9]. The attractive flowers are 10-12 mm tall, white or pink with many
longitudinal pink stripes on the lip, and are borne singly or in pairs
close to the apex of the stem [10]. For many reasons, including their
slow development, low seed germination rate, and low regeneration
rate, orchid conservation is an international concern. The main
factors affecting their natural population decline include habitat
damage, overharvesting and illegal trafficking, and pressure from
human population growth [11]. The most economically successful
tissue culture technique is the alternative means of plant vegetative
propagation known as micropropagation. The in vitro approach
through the application of plant tissue culture technology provides an
excellent opportunity for effective conservation by mass propagating
orchids in a short period [12]. The significant advantage offered by
micropropagation over conventional methods is that a large number
of plants can be produced from a single individual, independent
of the seasons [13]. Several valuable species of Dendrobium have
been reported to be propagated through asymbiotic germination of
immature seeds or direct shoot regeneration of PLBs from different
explants. [14]. The objective of the current study is to establish of
suitable protocol for successful in vitro culture of Dendrobium
terminale for PLB and callus formation.
Materials and Methods
Two-month-old juvenile seedlings of Dendrobium terminale Par.
& Rchb has been used as plant material, which has been developed
from seed suspension culture. The work was carried out in the
Cytogenetics and Plant Tissue Culture Laboratory, Department of
Botany, Siksha Bhavana, Visva Bharati, Santiniketan, during 2023
and 2024. Murashige and Skoog [15] (1962), a medium in half
concentration were used for culturing the planting materials. The MS
medium was supplemented with different plant growth regulators
(BAP, 2iPA, and IAA) in different concentrations. The culture is
maintained under 16±5°C temperature and 8 hours of photoperiod
in four replications for 60 days.
The 1 L stalk solution was prepared by adding 2.3 g of half MS
media in 200ml of distilled water, then 30 g of sucrose was added. The
solution was supplemented with various concentrations of the growth
regulators listed in [Table 1] and properly mixed. Then, by adding
distilled water, the mixture volume is made up to 1 L. By adding
either 0.1 N NaOH or 0.1 M HCl, the medium’s pH was adjusted to
5.5. After that, 8gms of Agar powder was added to the mixture and
dissolved by heating. The prepared media were poured into different
culture tubes, then covered with a cotton plug and autoclaved at
121°C for 20 minutes under 15 psi pressure. After cooling at room
temperature, the medium was used for inoculation and incubation. A
plant growth regulator-free medium was used as a control.
Results and Discussion
The studies recorded that seed suspension growth of Dendrobium
terminale is successful in ½ MS medium. Thus, ½ MS medium
supplemented with combination of IAA with BAP, 2iPA and 2iPA
alone was inoculated with a single seedling. After 60 days of growth,
every change in growth was carefully observed and recorded (Figure
1). In control, very little growth has been observed. The combination
of IAA (2 mg l-1) + BAP (mg l-1) showed relatively high PLB formation
and leaf growth. Whereas IAA (2 mg l-1) + BAP (4 mg l-1) has
resulted in significantly increased PLB formation, leaf growth, and
Figure 1: Multiple shoots and root formation in vitro culture of Dendrobium
terminale (A) Microscopic seeds of D. terminale obtain from manual
pollination (10X compound microscope), (B) Developing globular PLB, (C)
mature PLB and Callus, (D) Mature PLB with leaf primordia, (E) Multiple
shoots, (F) Well rooted multiple shoots on ½ MS medium supplemented with
IAA (2 mg l-1) and BAP (1 mg l-1)
root development compared to the control and other combinations.
However, IAA (4 mg l-1) + BAP (2 mg l-1) leads to a decrease in PLB
formation (25%) and an increase in callus (50%), without necrosis.
The combination of IAA with 2iPA responded differently in different
combinations. The IAA (2 mg l-1) + 2iPA (2 mg l-1) showed moderate
PLB formation and leaf growth than IAA(2mg/L) + 2iPA (4 mg l-1)
and control. The combination of IAA (2 mg l-1) + 2iPA (4 mg l-1)
induced higher PLB and callus formation. The combination of IAA
(4 mg l-1) + 2iPA (2 mg l-1) equals the proportion of PLB and callus
formation. The addition of 2iPA (2 mg l-1) alone in culture medium
lowers the PLB but induces callus formation.
Certain researchers have reported the healthy growth of orchid
protocorms in media having a balanced supply of organic and
inorganic nutrients. According to research on D. aphyllum tissue
culture on MS media, the seeds of the plant responded progressively
to varying kinetin concentrations [16]. The combination of IAA (2
mg l-1) and BAP (4 mg l-1) seems to be the most effective in promoting
plant growth, as it leads to the highest number of PLBs, significant
leaf growth. A simple and efficient growth protocol was developed
for Dendrobium lowii, an endangered and Borneo’s endemic epiphyte
orchid, using four-month-old protocorms as explant sources produced
by asymbiotic seeds germination [17]. Dendrobium lowii protocorms
were cultivated on Knudson C (KC) medium supplemented with
plant growth regulators (NAA, Zeatin, and BAP) at varying doses.
But our study showed that ½ MS supplemented with IAA, BAP, and
2iPA has a significant impact on plant growth with synergistic effects.
The plant growth regulators BAP, 2iPA and IAA are very suitable
for quick micropropagation because they have significant role in cell
division and root-shoot formation, leading correct direction of plant
developmental pathway under in vitro conditions mimicking natural
growth hormones.The combination of IAA (2 mg l-1) and 2iPA (2 mg
l-1) promotes the highest percentage of direct somatic embryogenesis
(75%) without inducing necrosis. Higher concentrations of cytokinin
2iPA (4 mg l-1) tend to favour the formation of callus over PLB.
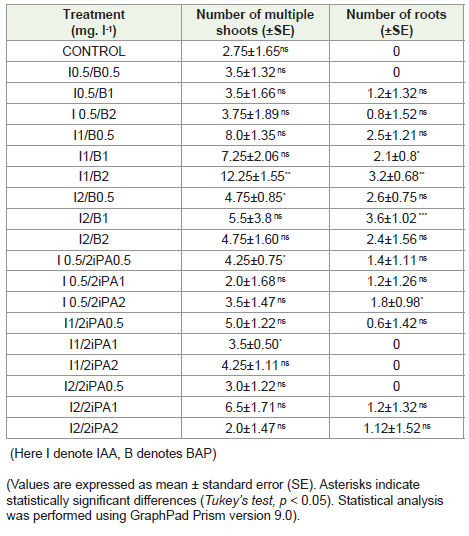
The results indicate that the combination of IAA (1 mg/L) with
BAP (2 mg/L) (I1/B2) was the most effective treatment, significantly
enhancing both multiple shoot formation (12.25 ± 1.55) and root
induction (3.2 ± 0.68), marked by a high level of statistical significance
(p < 0.01). This suggests a strong synergistic effect between these
concentrations of auxin and cytokinin. Another effective combination
was I2/B1, which yielded the highest number of roots (3.6 ± 1.02)
with high significance (p < 0.001), although its shoot number was
comparatively moderate (5.5 ± 3.8) and not statistically significant.
Other combinations, such as I1/B0.5 and I1/B1, also showed improved
shoot (8.0 ± 1.35 and 7.25 ± 2.06, respectively) and root numbers,
although without consistent significance. In contrast, treatments
involving 2iP generally resulted in lower shoot and root counts, with
no combination showing comparable performance to the BAP + IAA
treatments. The control and low concentrations of IAA and cytokinins
(e.g., I0.5/B0.5 or I0.5/B1) resulted in poor shoot proliferation and
little to no rooting. Overall, the combination of IAA 1 mg/L and BAP
2 mg/L emerged as the optimal condition for simultaneous shoot and
root development in Dendrobium terminale, underlining the greater
efficacy of BAP over 2iP in this orchid species. The superior response
observed with the combination of BAP (6-benzylaminopurine) and
IAA (indole-3-acetic acid) in the micropropagation of Dendrobium
terminale can be attributed to their synergistic roles in promoting
protocorm-like body (PLB) formation and shoot induction. BAP, a
potent cytokinin, effectively stimulates cell division and shoot bud
initiation, while IAA, a natural auxin, supports cell elongation and
root meristem development. When applied together in an optimal
ratio (BAP 2 mg. l-1+ IAA 1 mg. l-1), they create a hormonal balance
that favours organized tissue development over callusing, enhancing
both shoot proliferation and early root induction. Compared to other
plant growth regulator combinations
Conclusion
In conclusion, this study successfully established an in vitro
culture protocol for Dendrobium terminale, a threatened and
conservation-priority orchid species. After 60 days of culture, the
maximum protocorm-like body (PLB) formation occurred on MS
medium supplemented with BAP (4 mg l⁻¹) and IAA (2 mg l⁻¹). This
was followed by a combination of 2iPA (4 mg l⁻¹) and IAA (2 mg l⁻¹),
while the lowest PLB induction was observed with IAA (2 mg l⁻¹) and
2iPA (2 mg l⁻¹). The medium containing 2iPA (2 mg l⁻¹) was found
to promote greater leaf development. For root induction, the most
effective response was obtained from MS medium fortified with IAA
(2 mg l⁻¹) and BAP (1 mg l⁻¹).
References
Citation
Dutta S, Roy A. In vitro Regeneration of Dendrobium terminale, a CITES-Listed Orchid for Sustainable Conservation. J Plant Sci Res. 2025;12(2): 285