Case Report
The Beneficial Effect of Functional Electrical Stimulation of Miller Fisher syndrome (MFS) with Special Context to Guillain-Barré Syndrome Rehabilitation of a 25-year-Old Patient –A Systematic Review and Meta-Analysis
Borah AK1*, Goswami P2and Saikia H2
1Department of Neuro-Physiotherapy, Mahatma Gandhi University, District Ri Bhoi, Meghalaya, India
2Department of Physiotherapy and Orthopedic Rehabilitation, Sanjevani Hospital, Maligaon, Guwahati, Assam, India
2Department of Physiotherapy and Orthopedic Rehabilitation, Sanjevani Hospital, Maligaon, Guwahati, Assam, India
*Corresponding author:Arup Kumar Borah, Department of Neuro-Physiotherapy, Mahatma Gandhi University, District Ri Bhoi, Meghalaya, India. E-mail Id: barup614@gmail.com
Article Information:Submission: 14/04/2026; Accepted: 05/05/2026; Published: 07/05/2026
Copyright: © 2026 Borah AK, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Miller Fisher syndrome is a rare and atypical variation of Guillain-Barré syndrome, which includes the clinical trial of are flexia, ataxia, and ophthalmoplegia. Miller Fisher syndrome is commonly associated with the involvement of the lower cranial and facial nerves. Miller Fisher syndrome is one of the types of Guillain-Barré syndrome. Guillain-Barré syndrome has been defined to be the foremost incapacitating form of neurological disease following the disease polio. Guillain-Barré syndrome is a broad category that encompasses several types of acute immune-mediated polyneuropathies, the most common of which is acute inflammatory demyelinating polyradiculoneuropathy. It is considered part of the continuum of immune-mediated acute polyneuropathies, alongside GBS and Bickerstaff brainstem encephalitic (BBE). Charles Miller Fisher (1913-2012) first formally described this syndrome in 1956, and it has been recognized as a distinct clinical entity with in the GBS spectrum. We apply Functional Electrical Stimulation method of externally controlling muscles when signals from the brain can no longer control movement. This can happen after a spinal cord injury, stroke or neurological disorder such a multiple sclerosis. FES improves the quality of life (QoL) for patients with neurological disabilities across the globe. We also describe the patient’s clinical course,
diagnostic method, and therapy. The study demonstrates the value of early detection, quick action in treating Miller Fisher syndrome, and the possibility of full recovery with adequate therapy. Techniques utilized in physical therapy emphasize performing everyday tasks along with strengthening muscles. Miller fisher syndrome has a generally good prognosis, with most patients achieving full or near-full recovery within 8 to 12 weeks, and usually within 6 months.
Recovery involves the gradual reversal of ataxia (loss of balance), opthalmoplegia (eye movement issues), and arereflexia (lost reflexes). Early treatment with IVIg or plasmapheresis is key. In this paper we study a case report of a 25-year-old male patient who displayed the characteristic symptoms of Mill Fisher Syndrome. We also
investigate the patient’s clinical course, diagnostic method, and therapy. Hence, this case demonstrates the value of early detection, quick action in treating Miller Fisher syndrome, and the possibility of full recovery with adequate physiotherapy treatment. Techniques applied in physical therapy emphasize performing everyday tasks along with strengthening muscles.
Categories: Neurology; Pain Management; Physical Medicine and Rehabilitation.
Categories: Neurology; Pain Management; Physical Medicine and Rehabilitation.
Keywords:Miller fisher syndrome; Guillain-Barré syndrome; Physical recovery; Neurological recovery; Pain control
Abbreviations
MFS − Miller Fisher Syndrome; GBS − Guillain - Barré Syndrome;
AIDP − Acute Inflammatory Demyelinating Polyradiculopathy; SCAT
3/5 − Sport Concussion Assessment Tools – 5 steps Neurological
Screen; EMG − Electromyography; EEG − Electroencephalography;
DR − Direct Response (facial nerve, motor); NC − Nerve Conduction.
Purpose:
To describe a diagnostic approach and rehabilitation, the
management a rare case of Miller Fisher Syndrome.Objectives:
Miller Fisher syndrome objectives focus on early recognition of
the clinical triad – ataxia, areflexia and opthalmoplegia and prompt
management to improve patient outcomes. Key goals include
distinguishing MFS from other Guillain-Barré syndrome variants,
confirming diagnosis with anti-GQ1b antibodies, initiating immunetargeted
therapy, and preventing respiratory complications. Identify
treatment considerations including FES for patients with Miller Fisher
syndrome. Review the abnormal findings found upon evaluation of
Miller Fisher syndrome compared to those found in other forms of
Guillain-Barré syndrome.Case presentation
Patient information:
A 25-year-old young boy came to Sajeevani Hospital, Guwahati,
Assam India with a chief complaint of bilateral symmetrical weakness
in both upper and lower extremities over the preceding 45 days, with
more of an impact on his legs than his arms. The patient also had
history of chronic cough for 2 weeks before the onset. Patient noticed
imbalance and tendency to fall on either side during walking. The
patient had no relevant medical history of tuberculosis, diabetes or
hypertension. Neurological examinations finding showed ataxia gait
when the patient performed tandem gait test, concussion protocols
(SCAT3/5). The assessment and inquiry confirmed that the patient
was diagnosed as Miller Fisher syndrome (MFS), a variant of Guillain-
Barré syndrome. But, the patient was admitted and to shift for
specialized care in the neuro-intensive care unit (NICU) before being
moved to the general ward. Furthermore, the patient was referred to
neurophysiotherapy for further improvement in quality of life (QoL).Clinical Findings:
As MRI findings and shows enhancement of the cauda equina
nerve roots, consistent with the Landry-Guillain-Barre syndrome
spectrum. The examination process started only after obtaining parolcontract.
The patient’s head end is raised to 30 degrees in a supine
lying position. The patient was conscious and followed command
properly. His body type was ectomorph. The patient relied on his
auxiliary muscles for respiration since he had trouble breathing. Both
eyes showed signs of ptosis. Upon examination, opthalmoplegia was
determined by the paralysis of the ocular muscles. Both deep and
superficial reflexes remain unaltered. In the lower limb, superficial
reflexes were reduced while deep reflexes were not elicited. Thus,
clinical outcomes showed that both lower limbs had a loss of muscle
strength. Compared to the bilateral upper limb weakness, there was
a greater lower-limb weakness. The patient was unable to do basic
everyday tasks including sitting, standing and walking due to severe
exhaustion as well as physical pain. In the upper limb, superficial
sensations of touch, pain, and warmth were determined to remain
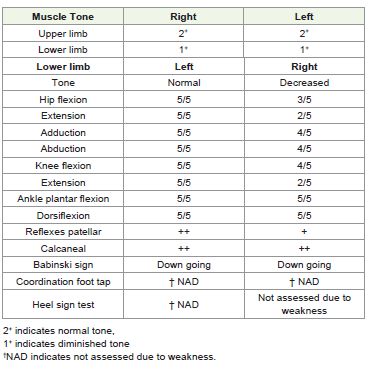
intact, but they were decreased in the lower limb. Now, the tone
grading scale indicating that the upper limb tone was normal, and on
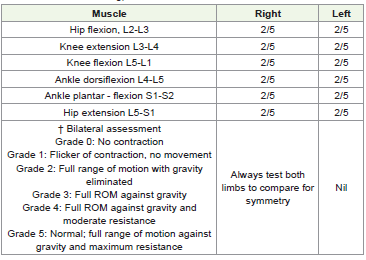
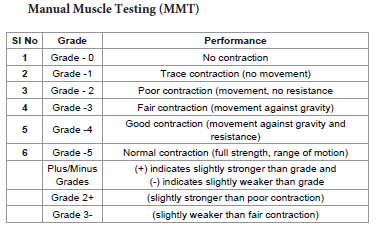
the lower limb, the tone was decreased [Table 1]. Grade-2 power was
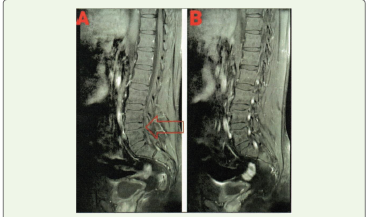
found in both lower The MRI report is shown in [Figure 1]. [Table 4]
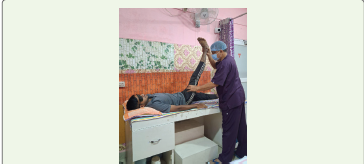
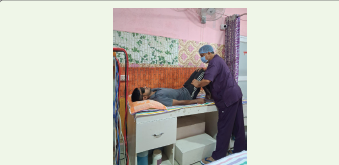
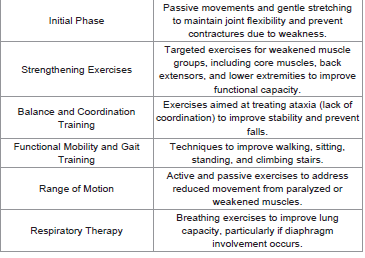
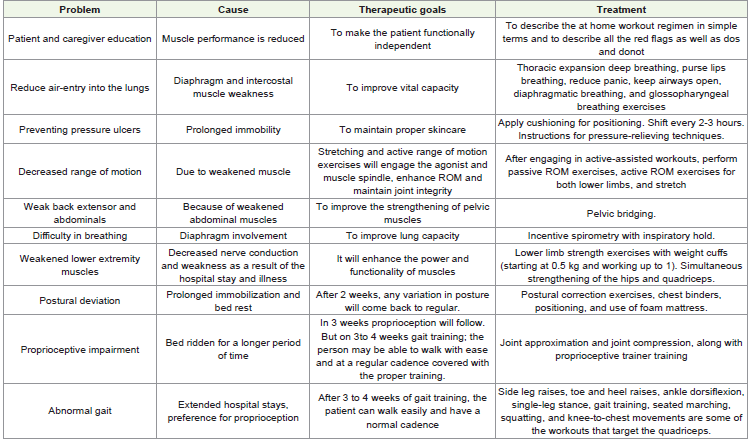
shows physiotherapy intervention. [Figure 2], [Figure 3] ,[Figure 4], and [Figure 5] show the exercises performed by the patient. [Figure 7] and outcome measures. limbs and during manual muscle testing
[Table 2] . (MMT). The supinator jerk, triceps, and biceps There was
no response on the knee and ankle [Table 3], all responded normally
to reflex tests. The bilateral lower limb’s hamstring and tibialis anterior muscles were found to be tight.
Figure 1:MRI scan showing (A) disc desiccation at the L4-L5 disc level, and
(B) conus medularis and roots of the cauda equina depicted enhancement
on contrast, raising the possibility of Guillain-Barré syndrome
Introduction
Functional Electrical stimulation (FES) systems can radically
change people’s lives. They increase a person’s functional ability,
thereby improving quality of life. The patient’s new found ability
to participate more actively in society reduces their isolation and
dependence on others. Miller Fisher syndrome is a rare disorder that
causes your immune system to attack nerves, usually starting in your
face. It can affect the nerves that control your eye muscles, so you may
have trouble moving your eyes. But, MFS also can cause problems
with your coordination and reflexes. Symptoms usually follow a
viral or bacterial infection. It is more common in Asian descent.
MFS affects males twice as often as females. Miller Fisher syndrome
is not contagious, so it doesn’t spread from person to person. This
syndrome is very rare. It affects about 1 to 2 people per 1,000,000
people worldwide.
MFS usually follows an infectious trigger, most commonly Cmapylobacter jejuni, Haemophilus influenza, or viral agents. Molecular mimicry leads to autoimmune attack on gangliosides, particularly GQ1b, explaining the cranial and sensory involvement. MFS predominantly affects the cranial nerves III, IV and VI, as well as proprioceptive pathways. Although, initially alarming, MFS has a favourable prognosis, with most patients recovering within 8-12 weeks. But, severe forms may involve limb weakness or respiratory compromise, blurring distinctions with GBS variants.
Neurological symptoms in Miller Fisher Syndrome have been associated with anti-GQ1b IgG antibodies. Generally, it takes 10-15 weeks for signs and symptoms to improve completely, and almost all patients have a good prognosis. Oculomotor, dorsal ganglion neurons, and muscle spindles all express the GQ1b ganglioside [1]. Ataxia, opthalmoplegia, paradoxical hyperreflexia and alternate consciousness are all symptoms of Bickerstaff brainstem encephalitis. Fisher’s syndrome has been associated with antibodies to ganglioside Q1b [2]. An upper respiratory or digestive tract disorder frequently comes before this severe polyneuropathy. Thus, 85-90% of all Miller Fisher syndrome patients, there is an antiganglioside antibody known as anti –GQ1b, which is self-reactive to the GQ1b ganglioside component of the nerve [3]. The most prevalent triggering infection is caused by Campylobacter jejuni, this associated illness with MFS, Bickerstaff brainstem encephalitis is characterized by a triad of acute bilateral opthalmoplegia, ataxia and encephalitis, with overlap with some forms of Guillain−Barré syndrome [4].
MFS usually follows an infectious trigger, most commonly Cmapylobacter jejuni, Haemophilus influenza, or viral agents. Molecular mimicry leads to autoimmune attack on gangliosides, particularly GQ1b, explaining the cranial and sensory involvement. MFS predominantly affects the cranial nerves III, IV and VI, as well as proprioceptive pathways. Although, initially alarming, MFS has a favourable prognosis, with most patients recovering within 8-12 weeks. But, severe forms may involve limb weakness or respiratory compromise, blurring distinctions with GBS variants.
Neurological symptoms in Miller Fisher Syndrome have been associated with anti-GQ1b IgG antibodies. Generally, it takes 10-15 weeks for signs and symptoms to improve completely, and almost all patients have a good prognosis. Oculomotor, dorsal ganglion neurons, and muscle spindles all express the GQ1b ganglioside [1]. Ataxia, opthalmoplegia, paradoxical hyperreflexia and alternate consciousness are all symptoms of Bickerstaff brainstem encephalitis. Fisher’s syndrome has been associated with antibodies to ganglioside Q1b [2]. An upper respiratory or digestive tract disorder frequently comes before this severe polyneuropathy. Thus, 85-90% of all Miller Fisher syndrome patients, there is an antiganglioside antibody known as anti –GQ1b, which is self-reactive to the GQ1b ganglioside component of the nerve [3]. The most prevalent triggering infection is caused by Campylobacter jejuni, this associated illness with MFS, Bickerstaff brainstem encephalitis is characterized by a triad of acute bilateral opthalmoplegia, ataxia and encephalitis, with overlap with some forms of Guillain−Barré syndrome [4].
Acute inflammatory immune mediated polyradiculopathy,
diminished or absent myotatic reflexes, distal areflexia and proximal
hyporeflexia, and albumin cytologic dissociation are diagnostic
criteria for Guillain-Barré syndrome. However, symptoms typically
include tingling, diminished strength of the muscle cell, as well as
discomfort [5]. Often the earliest noticeable signs of the Guillain-
Barré syndrome are symmetrical paresthesia that grows distally.
The four main subtypes of this condition are acute motor axonal
neuropathy, acute motor and sensory axonal neuropathy, and acute
inflammatory demyelinating polyradiculoneoropathy. Four studies
conducted in Western nations indicated in winter high, whereas
research from Northern China, India, Bangladesh and Latin America
indicated a summer peak [6].
On the other hand, the prevalence of Guillain-Barré syndrome
varies from 0.6 to 2.66 per 100,000 annually; according to a Metaanalysis
and men appear more likely than women to get Guillain-
Barré syndrome [7]. But, the autoimmune illness of Guillain-Barré
syndrome is diverse. It affects 0.4 to 1.7 cases per million people
annually [8]. Plasma exchange (PE) is considered as the best option and
first-line therapy method for gastrointestinal (GI) bleeding syndrome.
Individuals have experienced shorter times on ventilator assistance,
quicker healing and earlier mobility due to the benefits of plasma
exchange. The term multidisciplinary-care adhering to Guillain-
Barré syndrome implies healthcare administration involving more
than two specialists such as healthcare, physiotherapy, occupational
therapy, nutritional counseling and additional healthcare providers
[9].
Even though physicians might not regularly encounter individuals
with Guillain-Barré syndrome on daily basis, such individuals and the
family demands seen substantial [10].
Treatment protocols:
Initiate immunotherapy promptly, usually with intravenous
immunoglobulin (IVIg) or plasmapheresis as these are the standard
of care.Recovery – Most patients’ recovery fully within 8-12 months. Effects on Functional Electrical stimulation (FES)-Functional electrical stimulation has demonstrated potential to improve eye movement disorders (opthalmoplegia) faster than conventional, nonstimulated therapy.
Case Evidence – A 25 years old male patient with severe MFS showed improved ocular motility within 10 days TENS treatment, compared to an usual 5 months recovery.
Mechanism – we apply Electrical stimulation as well as functional electrical stimulation (FES) may help reestablish nerve-muscle connections (neuro-modulation) and prevent atrophy.
Method – management:
Miller Fisher syndrome is mainly treated with adequate
supportive care, pain control, respiratory support as needed and
immunotherapy. Although used in the past, oral or intravenous (IV)
steroids are no longer recommended in the treatment of GBS or MFS
because they are ineffective. Corticosteroids may slow recovery from
GBS and they are recommended only in the setting of neuropathic or
radicular pain. IV immunoglobulin (IVIg) and plasma exchange (PE)
are effective treatments for Guillain Barré syndrome and severe cases
of Miller Fisher syndrome.Post-Operative and Rehabilitation Role – Physical Therapy
treatment is essential for managing ataxia and muscle weakness.
A patient discharged with GBS or MFS may need a lengthy and
intense program of physiotherapy to recover function. Complete
recovery is dependent on many factors, including the severity of
neurological deficits at onset, the age of the patient, complications,
motivation, and goals of the patient, among others.
A thorough physical and occupational therapy assessment in the hospital is essential to identify the patient’s needs and goals of therapy. Patients with GBS and MFS frequently begin acute care and Physiotherapy treatment in the intensive care unit, then progress to a sub-acute setting in a rehabilitation department or outside nursing/ rehab facility and eventually translate to home-based or outpatient therapy. The assessment includes a patient/caregiver interview, sensory function, skin inspection, testing joint range motion, manual muscle testing, functional testing (e.g., ADL/IADL pre and postillness), mobility, respiration (e.g., vital capacity and inspiratory force), autonomic dysfunction, and endurance.
A thorough physical and occupational therapy assessment in the hospital is essential to identify the patient’s needs and goals of therapy. Patients with GBS and MFS frequently begin acute care and Physiotherapy treatment in the intensive care unit, then progress to a sub-acute setting in a rehabilitation department or outside nursing/ rehab facility and eventually translate to home-based or outpatient therapy. The assessment includes a patient/caregiver interview, sensory function, skin inspection, testing joint range motion, manual muscle testing, functional testing (e.g., ADL/IADL pre and postillness), mobility, respiration (e.g., vital capacity and inspiratory force), autonomic dysfunction, and endurance.
Task Oriented Training – Functional Electrical stimulation
combined with task-oriented training is highly effective for regaining
functional movement, similar to its use in other polyneuropathies.
Safety – Nerve stimulation and functional electrical stimulation
are generally considered a safe, adjunct therapy to standard care.
Symptoms of Miller Fisher Syndrome
There’s no way to prevent Miller Fisher syndrome. But you can
take steps to reduce your risk of getting a viral or bacterial infection.
Be sure to:
Outlook for someone with Miller Fisher syndrome: The outlook for people who receive treatment for Miller Fisher syndrome is good. It’s rarely fatal and usually doesn’t cause any serious or long-term health complications. Most people recover within two to six months.
Outlook for someone with Miller Fisher syndrome: The outlook for people who receive treatment for Miller Fisher syndrome is good. It’s rarely fatal and usually doesn’t cause any serious or long-term health complications. Most people recover within two to six months.
Clinical aspects of Miller Fisher syndrome (MFS):
A lumber puncture was performed to examine the cerebrospinal
fluid (CSF). The ultimate diagnosis of Miller Fisher syndrome, a
variant of Guillain-Barré syndrome, was made possible by the lumbar
puncture (lower back, usually L3/L4 below the spinal cord itself)
and clinical findings. The complete blood count was examined. MRI
reports show in [Figure 1].ata availability statement:
The original contributions presented in the study are included in
the article / supplementary material; further inquiries can be directed
to the corresponding authors.Ethics statement:
Written informed consent was obtained from the individual(s)
for the publication of any potentially identifiable images or data
included in this article.Conflict of interest:
The authors declare that the research was conducted in the
absence of any commercial or financial relationships that could be
constructed as a potential conflict of interest.All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in the article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Discussion
Diagnosis of Miller Fisher syndrome in this patient is supported
by the clinical findings of acute onset ophthalmoplegia, ataxia and
areflexia following an episode of upper respiratory tractus infection.
Although analysis CSF and serological testing anti GQ1B antibodies
was not performed, but electromyography finding of this patient
supported our clinical diagnosis. Miller Fisher syndrome has a
positive prognosis, and the decision to initiate immunomodulatory
therapy should be guided by the patient’s overall condition.
The patient had shown gradual improvement in symptoms
including power, ataxia, ophthalmoplegia at fourth and eight week
follow up.
As a result of its unique manifestation and uncommon evaluation, the above neurological case presents significant challenges. The surveillance of complications and crucial medical care among those diagnosed with Guillain-Barré syndrome has given way to a newly developed intensive approach (plasma exchange and intravenous immunoglobulin infusion), which reduces this condition’s course and improves outcomes, particularly among individuals with severe illness. This approach for handling followed earlier research that suggested treating the root cause of the disease, treating acute neuropathy along with its consequences, and promoting the individual’s sustained recovery which constitute the most important aspects of managing Guillain-Barré syndrome. Research has demonstrated to benefit critically ill patients in intensive care units, and speedy rehabilitation promotes a rapid cure. Vital signs showed extraordinary improvement, primarily attributable to the team and intensive care unit scheduled interventions [11]. Atypical Guillain-Barré syndrome is a rare version of ataxia, ophthalmoplegia, and areflexia that complies with the medical range established by Cullier. GQ1b antibodies found in the fibers of oculomotor nerves, neurons of the posterior spinal root ganglia, and neuromuscular spindles elicit an across-reaction that results in Miller Fisher syndrome following an episode of digestive problems as well as pulmonary infection or immunization [12].
As a result of its unique manifestation and uncommon evaluation, the above neurological case presents significant challenges. The surveillance of complications and crucial medical care among those diagnosed with Guillain-Barré syndrome has given way to a newly developed intensive approach (plasma exchange and intravenous immunoglobulin infusion), which reduces this condition’s course and improves outcomes, particularly among individuals with severe illness. This approach for handling followed earlier research that suggested treating the root cause of the disease, treating acute neuropathy along with its consequences, and promoting the individual’s sustained recovery which constitute the most important aspects of managing Guillain-Barré syndrome. Research has demonstrated to benefit critically ill patients in intensive care units, and speedy rehabilitation promotes a rapid cure. Vital signs showed extraordinary improvement, primarily attributable to the team and intensive care unit scheduled interventions [11]. Atypical Guillain-Barré syndrome is a rare version of ataxia, ophthalmoplegia, and areflexia that complies with the medical range established by Cullier. GQ1b antibodies found in the fibers of oculomotor nerves, neurons of the posterior spinal root ganglia, and neuromuscular spindles elicit an across-reaction that results in Miller Fisher syndrome following an episode of digestive problems as well as pulmonary infection or immunization [12].
Etiology of this case is likely viral (both influenza-like and
gastrointestinal), which might cause a reaction of autoimmune origin
that could, in a few instances, result in Guillain-Barré syndrome
along with the uncommon Miller Fisher syndrome [13].
Specific subgroups of Guillain-Barré syndrome can express
distinct antiganglioside indicators. The characteristic Miller Fisher
syndrome outcomes for our patient’s situation had been bilateral
ophthalmoplegia, hyporeflexia, and antiganglioside positive along
with elevated cerebrospinal fluid. Depending on when the lumbar
puncture is done, albumin-cytological separation is the usual
cerebrospinal fluid finding for Guillain-Barré syndrome [14].
Furthermore, common prior occurrences among patients
incorporate illnesses, especially gastrointestinal and infections of
the airways nearly (83%). Thus, this clinical case study provides an
example of how physiotherapy can help individuals with Miller Fisher
syndrome Guillain-Barré syndrome (GBS) achieve an independent
return. The severity and duration of symptoms are brought into
focus, making a conventional treatment strategy more challenging to
follow, and calls for additional study into how innovative therapies
affect healing [15].
The functional state has been evaluated applying the Hughes disability scale, whereas the muscle power was evaluated using the Medical Research Council (MRC) grades [16]. Presently, there hasn’t been any epidemiological research that explicitly evaluates the occurrence and rate of Miller Fisher syndrome [17]. Bersch et al. studied that based on task-focused functional training and a 16 week intensive functional electrical stimulation; the outcomes show that a patient with chronic Guillain-Barré syndrome with hyperreflexia can enhance their fine motor abilities [18]. It is possible to reach early independent functioning with the right medical care and physiotherapy treatment [19,20].
The functional state has been evaluated applying the Hughes disability scale, whereas the muscle power was evaluated using the Medical Research Council (MRC) grades [16]. Presently, there hasn’t been any epidemiological research that explicitly evaluates the occurrence and rate of Miller Fisher syndrome [17]. Bersch et al. studied that based on task-focused functional training and a 16 week intensive functional electrical stimulation; the outcomes show that a patient with chronic Guillain-Barré syndrome with hyperreflexia can enhance their fine motor abilities [18]. It is possible to reach early independent functioning with the right medical care and physiotherapy treatment [19,20].
Conclusion and direction to future research
Mildly affected Guillain-Barré syndrome patients may still have
considerable problems in mobility at three and six months after
onset of the disease. The positive effect of PE (plasma exchange aka,
plasmaparesis). It is a primary first-line immunotherapy treatment
used to treat this rare neurological disorder by filtering harmful
autoantibodies out of the blood [21].
References
10. Khan F (2004) Rehabilitation in Guillian Barre syndrome. Aust Fam Physician. 2004, 33:1013-1017.
Citation
Borah AK, Goswami P, Saikia H. The Beneficial Effect of Functional Electrical Stimulation of Miller Fisher syndrome (MFS) with Special Context to Guillain-Barré Syndrome Rehabilitation of a 25-year-Old Patient –A Systematic Review and Meta-Analysis. Indian J Neurol. 2026;7(1): 167.