Case Report
Multiple System Atrophy Type C and Its Neuropsychiatric Manifestations: Challenges in Diagnosis and Management-A Case Report
Kothekar M*, Parkar S, Ghodke J, Desai S, Dhar A and Purohit S
Department of Psychiatry, Vedantaa Institute of Medical Sciences, Dahanu, Palghar
*Corresponding author:Dr. Mansi Kothekar, Vedantaa Institute of Medical Sciences, Dahanu, Palghar. E-mail Id: dr.mansikothekar16@gmail.com
Article Information:Submission: 02/04/2026; Accepted: 16/04/2026; Published: 20/04/2026
Copyright: © 2026 Kothekar M, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: Psychotic symptoms in Multiple System Atrophy (MSA) are uncommon, affecting approximately 5–10% of patients, and are frequently attributed to dopaminergic therapy or advanced disease. Early-onset psychosis intrinsic to MSA–C remains underrecognized and may result in misdiagnosis as a primary psychiatric disorder, delaying appropriate management.
Case Summary: A 54-year-old man with no prior psychiatric history presented with a one-week history of persecutory delusions and multimodal hallucinations. The psychosis was accompanied by executive dysfunction (MMSE 23/30; impaired Trail Making Test) and had emerged in the setting of progressive cerebellar ataxia, parkinsonism, and autonomic dysfunction (constipation, bladder dysfunction) — all antedating dopaminergic exposure. MRI demonstrated periventricular T2/FLAIR hyperintensities without the pathognomonic ‘hot cross bun’ sign. Based on consensus diagnostic criteria, a diagnosis of probable MSA–C was established. Olanzapine 5 mg nightly led to gradual resolution of psychosis without extrapyramidal worsening over two weeks.
Conclusion: This case illustrates that psychosis may be an early, intrinsic manifestation of MSA–C, preceding dopaminergic therapy and motor symptom dominance. Late-onset psychosis co-occurring with executive dysfunction and autonomic features should prompt systematic evaluation for neurodegenerative synucleinopathies. Recognition of underlying MSA has critical implications for antipsychotic selection and prognostication.
Case Summary: A 54-year-old man with no prior psychiatric history presented with a one-week history of persecutory delusions and multimodal hallucinations. The psychosis was accompanied by executive dysfunction (MMSE 23/30; impaired Trail Making Test) and had emerged in the setting of progressive cerebellar ataxia, parkinsonism, and autonomic dysfunction (constipation, bladder dysfunction) — all antedating dopaminergic exposure. MRI demonstrated periventricular T2/FLAIR hyperintensities without the pathognomonic ‘hot cross bun’ sign. Based on consensus diagnostic criteria, a diagnosis of probable MSA–C was established. Olanzapine 5 mg nightly led to gradual resolution of psychosis without extrapyramidal worsening over two weeks.
Conclusion: This case illustrates that psychosis may be an early, intrinsic manifestation of MSA–C, preceding dopaminergic therapy and motor symptom dominance. Late-onset psychosis co-occurring with executive dysfunction and autonomic features should prompt systematic evaluation for neurodegenerative synucleinopathies. Recognition of underlying MSA has critical implications for antipsychotic selection and prognostication.
Keywords:Multiple System Atrophy-C; Parkinsonism; Psychosis; Neurodegeneration; Late-onset psychosis; Synucleinopathy; Antipsychotic selection
Introduction
Late-onset psychosis presents a significant diagnostic challenge
in neuropsychiatric practice. While primary psychotic disorders can
emerge in midlife, secondary causes – particularly neurodegenerative
disorders - must be systematically excluded before a primary
psychiatric diagnosis is assigned.
Among synucleinopathies, psychosis is well characterised in Parkinson’s disease (PD) and dementia with Lewy bodies (DLB). In contrast, psychotic manifestations in Multiple System Atrophy (MSA) are less frequently emphasised in the literature and are often attributed to dopaminergic therapy or advanced disease stages. [9,10]
Among synucleinopathies, psychosis is well characterised in Parkinson’s disease (PD) and dementia with Lewy bodies (DLB). In contrast, psychotic manifestations in Multiple System Atrophy (MSA) are less frequently emphasised in the literature and are often attributed to dopaminergic therapy or advanced disease stages. [9,10]
MSA is a progressive α-synucleinopathy characterised by
variable combinations of autonomic failure, parkinsonism, and
cerebellar ataxia.[1,9] It arises from oligodendroglial cytoplasmic
inclusions containing misfolded α-synuclein, leading to multisystem
neurodegeneration.[1] Consensus criteria classify MSA into MSA-P
(parkinsonian predominant) and MSA-C (cerebellar predominant)
subtypes.[11] The prevalence is estimated at 3-5 per 100,000
individuals, with onset typically in the fifth to seventh decades of life.
[9]
Although motor and autonomic symptoms predominate,
neuropsychiatric manifestations including depression, cognitive
impairment, sleep disturbances, and - less commonly - psychosis
are increasingly recognised.[6,12] Neuropathological studies have
demonstrated that α-synuclein pathology in MSA may extend beyond
classical motor circuits to involve limbic and associative cortical
regions, [2,3] providing a plausible substrate for early psychiatric
manifestations.
We report a case in which psychosis emerged as an early and
clinically dominant feature of probable MSA-C, preceding both
dopaminergic therapy and full motor syndrome characterisation.
We discuss the neurobiological mechanisms, differential diagnostic
considerations, and pharmacological implications of this presentation.
Case Presentation
A 54-year-old man with no prior psychiatric history presented
with a one-week history of persecutory delusions and multimodal
hallucinations. He expressed a fixed belief that unknown persons
intended to cause him harm, was observed muttering to himself,
and was noted to gesture toward unseen stimuli. Sleep was markedly
reduced and appetite had declined.
Over the preceding year, he had developed progressive imbalance, frequent falls, and difficulty with postural stability. Two weeks prior to psychiatric presentation, he experienced constipation, abdominal distension, and incomplete bladder emptying – autonomic disturbances consistent with MSA. [6,9] There was no history of substance use, head trauma, seizures, or family history of psychiatric illness. No fluctuating sensorium was recorded on any assessment.
Over the preceding year, he had developed progressive imbalance, frequent falls, and difficulty with postural stability. Two weeks prior to psychiatric presentation, he experienced constipation, abdominal distension, and incomplete bladder emptying – autonomic disturbances consistent with MSA. [6,9] There was no history of substance use, head trauma, seizures, or family history of psychiatric illness. No fluctuating sensorium was recorded on any assessment.
Mental Status Examination:
On assessment, the patient was dishevelled with a stooped
posture. Psychomotor slowing and limited eye contact were noted.
Spontaneous speech was decreased with increased latency. Mood was
anxious (“I feel unsafe”); affect was restricted and congruent. Thought
process was goal-directed but slowed. Thought content revealed
systematised persecutory delusions. Perceptual abnormalities
included second- and third-person auditory hallucinations and
intermittent visual hallucinations.Cognitive assessment demonstrated:
Orientation to time, place, and person intact; impaired recent
memory; MMSE score of 23/30; and impaired Trail Making Test
performance indicative of executive dysfunction. Insight was absent
and judgment impaired. Executive dysfunction of this nature is
consistent with prior neuropsychological studies demonstrating
frontal-subcortical deficits in MSA, reflecting disruption of
dorsolateral prefrontal–striatal circuitry. [4,5,12]Neurological Examination and Neuroimaging:
Neurological examination revealed coarse tremors, reduced arm
swing, short-stepped gait, cerebellar incoordination, and impaired
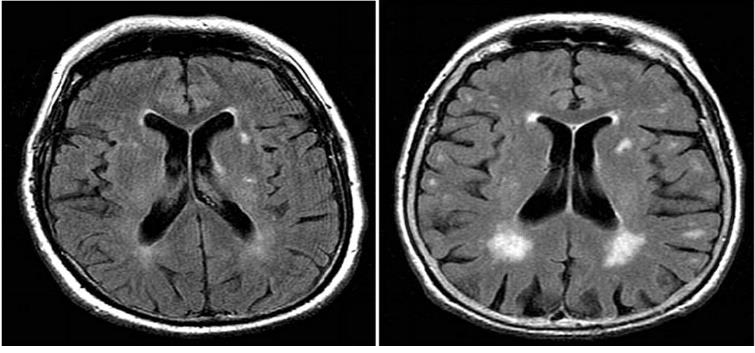
Figure 1:MRI Findings T2/ Flair-periventricular hyperintensities
unremarkable for cerebello-pontine T2 hyperintensities. (likely due to the
disease’s rapid progression)
proprioception — a mixed cerebellar and parkinsonian phenotype
consistent with MSA-C. [1,11]
Brain MRI demonstrated periventricular T2/FLAIR hyperintensities. The pathognomonic ‘hot cross bun’ sign of pontine degeneration was absent; however, absence of this finding does not exclude MSA, particularly in early stages. [1,9] Blood investigations including complete blood count, metabolic panel, thyroid function, serum B12, VDRL, and HIV serology were within normal limits, excluding common metabolic and infective causes of psychosis. An electroencephalogram showed no epileptiform activity.
Based on the Second Consensus Statement on the Diagnosis of Multiple System Atrophy,[11] the combination of cerebellar dysfunction, parkinsonism, and autonomic failure supported a diagnosis of probable MSA-C.
Brain MRI demonstrated periventricular T2/FLAIR hyperintensities. The pathognomonic ‘hot cross bun’ sign of pontine degeneration was absent; however, absence of this finding does not exclude MSA, particularly in early stages. [1,9] Blood investigations including complete blood count, metabolic panel, thyroid function, serum B12, VDRL, and HIV serology were within normal limits, excluding common metabolic and infective causes of psychosis. An electroencephalogram showed no epileptiform activity.
Based on the Second Consensus Statement on the Diagnosis of Multiple System Atrophy,[11] the combination of cerebellar dysfunction, parkinsonism, and autonomic failure supported a diagnosis of probable MSA-C.
Differential Diagnosis:
The differential diagnosis considered included:• Late-onset schizophrenia: No prior psychiatric history; motor and autonomic features atypical for primary psychosis.
• Psychotic disorder due to neurocognitive disorder: Cognitive impairment was mild and executive-predominant without frank dementia at presentation.
• Dementia with Lewy bodies (DLB): Absence of characteristic cognitive fluctuations and predominant cerebellar features argued against this diagnosis.
• Parkinson’s disease psychosis: Cerebellar-predominant phenotype and absence of prior dopaminergic therapy argued against a PD-related aetiology.
• Delirium: No fluctuating consciousness or identifiable acute precipitant; investigations excluded metabolic and infective causes.
Critically, psychosis antedated dopaminergic therapy in this patient, implicating intrinsic neuropathological mechanisms rather than medication-induced phenomena. [10,12]
Management and Outcome:
Olanzapine 5 mg nightly was initiated following a risk-benefit
discussion. Gradual resolution of psychotic symptoms was observed
over two weeks, with no worsening of extrapyramidal or cerebellar
features on serial examination.Typical (first-generation) antipsychotics were avoided given the
risk of exacerbating parkinsonian features in the context of nigrostriatal
degeneration.[8] In parkinsonian disorders, antipsychotic selection
requires careful consideration of D2-receptor blockade and associated
motor vulnerability. Although clozapine and pimavanserin have the
strongest evidence base in PD-related psychosis, cautious low-dose
use of selected second-generation antipsychotics may be considered
when closely monitored - particularly when clozapine prescribing
infrastructure is unavailable.[8]
The patient was counselled regarding the progressive nature of MSA and referred for multidisciplinary input including neurology, physiotherapy, and speech therapy. Family members received caregiver education.
The patient was counselled regarding the progressive nature of MSA and referred for multidisciplinary input including neurology, physiotherapy, and speech therapy. Family members received caregiver education.
Discussion
Neurobiological Basis of Psychosis In MSA–C MSA is
characterised by oligodendroglial α-synuclein inclusions producing
degeneration of the striatonigral and olivopontocerebellar pathways.
[1,9] Neuropathological studies have demonstrated convergence of
α-synuclein pathology into limbic structures, including the amygdala
and anterior cingulate cortex, [2,3] regions critically implicated
in emotional salience, threat appraisal, and the pathogenesis of
delusions.
Executive dysfunction in this patient implicates additional disruption of dorsolateral prefrontal–striatal circuitry. [4,5] Impaired top-down regulatory control over limbic salience networks may predispose to delusional ideation and aberrant threat perception. Furthermore, cerebellar degeneration in MSA–C disrupts cerebellothalamo- cortical circuits, and the cerebellum has been increasingly implicated in psychosis through mechanisms of cognitive dysmetria and predictive processing failure.[7] The convergent degeneration of frontostriatal and cerebellar networks in MSA–C may thus synergistically increase vulnerability to psychotic experiences. Psychiatric manifestations in MSA — including hallucinations and delusions — have been documented in both clinical and clinicopathological studies, [12,13] though they remain less common than affective or sleep-related symptoms and are under emphasised in clinical guidance.
Executive dysfunction in this patient implicates additional disruption of dorsolateral prefrontal–striatal circuitry. [4,5] Impaired top-down regulatory control over limbic salience networks may predispose to delusional ideation and aberrant threat perception. Furthermore, cerebellar degeneration in MSA–C disrupts cerebellothalamo- cortical circuits, and the cerebellum has been increasingly implicated in psychosis through mechanisms of cognitive dysmetria and predictive processing failure.[7] The convergent degeneration of frontostriatal and cerebellar networks in MSA–C may thus synergistically increase vulnerability to psychotic experiences. Psychiatric manifestations in MSA — including hallucinations and delusions — have been documented in both clinical and clinicopathological studies, [12,13] though they remain less common than affective or sleep-related symptoms and are under emphasised in clinical guidance.
Clinical Implications:
This case highlights several clinically important points:• Late-onset psychosis warrants systematic neurological evaluation, including assessment of motor and autonomic function.
• The combination of executive dysfunction and autonomic symptoms in a patient with new-onset psychosis should function as a red flag for underlying neurodegenerative disease. [4-6]
• Psychosis in MSA may occur independently of dopaminergic therapy, indicating intrinsic neuropathological mechanisms. [10,12]
• Antipsychotic selection in this context must carefully consider motor vulnerability: D2-blocking agents carry risk of extrapyramidal exacerbation, and second-generation agents should be used at the lowest effective dose with close monitoring.[8]
• Early and accurate diagnosis prevents prolonged misclassification as primary psychosis, enables family counselling, and guides prognosis.
Conclusion
Psychosis may represent an early, intrinsic manifestation of
MSA–C rather than a treatment-related epiphenomenon. The
co-occurrence of executive dysfunction, autonomic features, and
late-onset psychosis should prompt evaluation for underlying
synucleinopathies. Integration of psychiatric and neurological
assessment is essential for diagnostic accuracy and for guiding safer
antipsychotic management in this vulnerable patient population.
Declarations
Declaration of Patient Consent:
The authors declare that written informed consent was obtained
from the patient for publication of this case report. The patient has
consented to the publication of clinical information. His name will not
be published, and due efforts have been made to conceal identifying
information, although complete anonymity cannot be guaranteed.References
Citation
Kothekar M, Parkar S, Ghodke J, Desai S, Dhar A, et al. Multiple System Atrophy Type C and Its Neuropsychiatric Manifestations: Challenges in Diagnosis and Management-A Case Report. Indian J Neurol. 2026;7(1): 166