Research Article
Exploring Neurological and Cardiac Biomarkers in Acute Ischemic Stroke: A Correlation with Stroke Severity and Prognosis
Khan Y* and Gaikwad A
Department of Medicine, Internal Medicine. MRCP UK, Swastik hospital Jabalpur, Madhya Pradesh, India
*Corresponding author: Yasmin Khan, Department of Medicine, Internal medicine, MRCP UK, Swastik hospital, Jabalpur, Madhya Pradesh, India. E-mail Id: Yasu7868@gmail.com
Article Information:Submission: 29/07/2025; Accepted: 19/08/2025; Published: 23/08/2025
Copyright: © 2025 Khan Y, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: Biomarkers play a crucial role in predicting clinical outcomes in acute ischemic stroke. This study evaluates the predictive ability of S100B, neuron-specific enolase (NSE), troponin, and N-terminal pro-brain natriuretic peptide (NT-proBNP) for stroke severity, mortality, and functional outcomes in a cohort of acute ischemic stroke patients.
Methods: A retrospective analysis was conducted on 80 acute ischemic stroke patients admitted between February 2023 and January 2024, with a follow-up period of three months. Multiple linear regression assessed the relationship between biomarkers and stroke severity using the National Institutes of Health Stroke Scale (NIHSS). Logistic regression determined predictors of mortality, while ordinal logistic regression evaluated functional outcomes using the modified Rankin Scale (mRS) at three months. Kaplan-Meier survival analysis and Cox proportional hazards models analyzed time-to-mortality. Receiver Operating Characteristic (ROC) curve analysis assessed the discriminatory power of biomarkers in predicting mortality.
Results: Among the biomarkers analyzed, NT-proBNP showed the strongest correlation with NIHSS scores, indicating its potential as a predictor of stroke severity. S100B and NSE exhibited weaker associations, while troponin levels had minimal correlation with clinical severity. The overall mortality rate was 56.25%, with significantly higher NT-proBNP levels observed in non-survivors. These findings suggest that while NT-proBNP may serve as a useful prognostic marker, a combination of clinical assessment and biomarker evaluation is necessary for accurate risk stratification in AIS patients.
Conclusion: NT-proBNP emerged as a strong predictor of stroke severity and mortality, highlighting its potential role in AIS prognosis
Methods: A retrospective analysis was conducted on 80 acute ischemic stroke patients admitted between February 2023 and January 2024, with a follow-up period of three months. Multiple linear regression assessed the relationship between biomarkers and stroke severity using the National Institutes of Health Stroke Scale (NIHSS). Logistic regression determined predictors of mortality, while ordinal logistic regression evaluated functional outcomes using the modified Rankin Scale (mRS) at three months. Kaplan-Meier survival analysis and Cox proportional hazards models analyzed time-to-mortality. Receiver Operating Characteristic (ROC) curve analysis assessed the discriminatory power of biomarkers in predicting mortality.
Results: Among the biomarkers analyzed, NT-proBNP showed the strongest correlation with NIHSS scores, indicating its potential as a predictor of stroke severity. S100B and NSE exhibited weaker associations, while troponin levels had minimal correlation with clinical severity. The overall mortality rate was 56.25%, with significantly higher NT-proBNP levels observed in non-survivors. These findings suggest that while NT-proBNP may serve as a useful prognostic marker, a combination of clinical assessment and biomarker evaluation is necessary for accurate risk stratification in AIS patients.
Conclusion: NT-proBNP emerged as a strong predictor of stroke severity and mortality, highlighting its potential role in AIS prognosis
Keywords:Acute Ischemic Stroke [AIS]; Biomarkers; Stroke Severity; Mortality Prediction; NT-proBNP; ROC Curve Analysis; Survival Analysis; Functional Outcomes; NIHSS Score; mRS.
Introduction
Acute ischemic stroke (AIS) remains a leading cause of morbidity
and mortality worldwide, necessitating the identification of reliable
biomarkers for predicting stroke severity, mortality, and functional
outcomes.[1] Early risk stratification is crucial for guiding treatment
decisions and improving patient management. Various biomarkers,
including S100B, neuron-specific enolase (NSE), troponin, and
N-terminal pro-brain natriuretic peptide (NT-proBNP), have been
studied for their potential role in prognosticating stroke outcomes.
However, their predictive accuracy remains uncertain. [2,3]
S100B is a glial-derived protein linked to blood-brain barrier
dysfunction and neuronal damage. Elevated levels have been
associated with stroke severity, though its role in mortality prediction
is inconsistent.[4]NSE, a neuronal enzyme, reflects neuronal injury
but has shown variable results in stroke prognosis.[5] Troponin,
primarily a cardiac biomarker, is increasingly recognized in
cerebrovascular events due to its association with neurogenic stress
cardiomyopathy [6]. NT-proBNP, a marker of cardiac strain, has
been linked to embolic stroke mechanisms and adverse outcomes [7].
Despite extensive research, the clinical utility of these biomarkers
remains controversial. The National Institutes of Health Stroke Scale
(NIHSS) is widely used to assess stroke severity, while the modified
Rankin Scale (mRS) evaluates long-term functional outcomes.[8]
Survival analysis techniques, such as Kaplan-Meier curves and Cox
proportional hazards models, allow for an in-depth assessment of
time-to-mortality in stroke patients. Additionally, Receiver Operating
Characteristic (ROC) curve analysis is a valuable tool for determining
the discriminatory power of biomarkers in predicting mortality.
This study aims to evaluate the prognostic value of S100B, NSE,
troponin, and NT-proBNP in 80 patients with acute ischemic stroke
admitted between February 2023 and January 2024. The primary
objectives are to assess the association of these biomarkers with
stroke severity (NIHSS Score), mortality, and functional outcomes
(mRS), and to determine their predictive accuracy through ROC
curve analysis. Findings from this study could enhance stroke risk
stratification and guide future research on biomarker-driven clinical
decision-making.
Methodology
This retrospective cohort study was conducted on 80 patients
diagnosed with acute ischemic stroke (AIS) between February 2023
and January 2024 at Swastik Hospital, Jabalpur, India a tertiary care
hospital. Patient data, including demographics, clinical parameters,
biomarker levels, and follow-up outcomes, were retrieved from
medical records. The study included patients aged ≥18 years with a
confirmed AIS diagnosis based on clinical and radiological findings
(CT/MRI), available biomarker data (S100B, NSE, troponin, NTproBNP)
within 24 hours of stroke onset, and complete follow-up
data for mortality and functional outcomes at 3 months. Patients
with hemorrhagic stroke, transient ischemic attack (TIA), recent
myocardial infarction, severe systemic infections affecting biomarker
levels, or incomplete medical records were excluded. Blood samples
were collected within 24 hours of stroke onset, and biomarker levels
were quantified using standardized enzyme-linked immunosorbent
assays (ELISA). Stroke severity was assessed using the NIHSS at
admission, while functional outcomes were evaluated at 3 months
using the modified Rankin Scale (mRS), with mortality recorded
at the same time point. Statistical analysis included multiple linear
regression to assess associations between biomarker levels and NIHSS
Score, logistic regression for biomarker predictors of mortality, and
ordinal logistic regression to examine the impact of biomarkers and
NIHSS Score on mRS outcomes. Kaplan-Meier survival analysis and
Cox proportional hazards models were used to compare survival
probabilities and estimate hazard ratios, while ROC curve analysis
evaluated the predictive accuracy of biomarkers for mortality using
the Area Under the Curve (AUC). All analyses were performed using
STATA and SPSS 25.0, with statistical significance set at p <0.05.
The study was approved by the institutional ethics committee, with
informed consent waived due to its retrospective design, ensuring
data confidentiality throughout the research process.
Result
The study analyzed 80 acute ischemic stroke patients, with a mean
age of 61.76 years (SD: 13.24, range: 41–84 years). Stroke severity,
measured using the NIHSS Score, had a mean of 10.49 (SD: 7.22, range:
0–24), indicating moderate severity among participants. Biomarker
levels varied significantly: S100B ranged from 0.10 to 1.50 ng/L, with
a mean of 0.78 ng/L (SD: 0.39); NSE had a mean of 18.25 ng/mL (SD:
6.84, range: 5.45–29.86 ng/mL); Troponin levels were relatively low,
with a mean of 0.23 ng/mL (SD: 0.15, range: 0.01–0.49 ng/mL); and
NT-proBNP exhibited the greatest variation, ranging from 242.87 to
4979.86 pg/mL, with a mean of 2775.45 pg/mL (SD: 1395.52). The
distribution of biomarkers suggests heterogeneity in stroke severity
and associated cardiac or neuronal damage. The cohort had a male
predominance (53.75%), while females comprised 46.25%. Regarding
mortality, 45 patients (56.25%) succumbed, whereas 35 (43.75%)
survived, emphasizing the severity of acute ischemic stroke and the
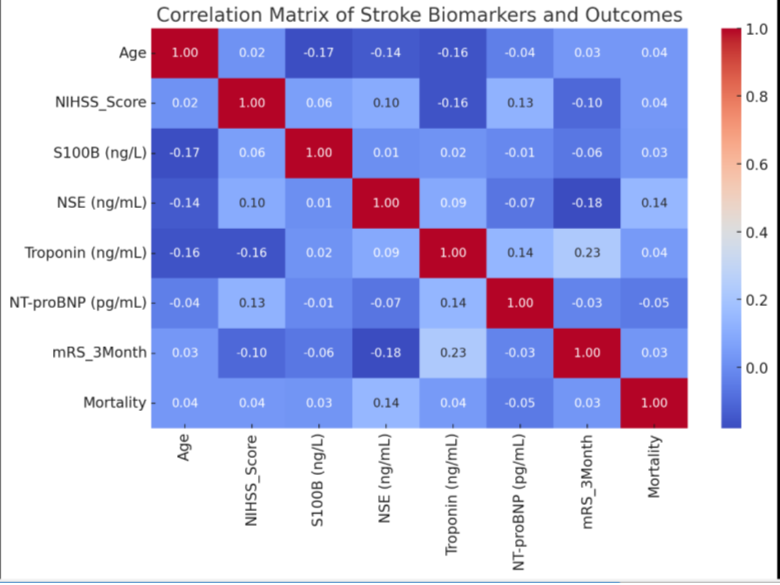
need for effective prognostic biomarkers. (Figure 1)
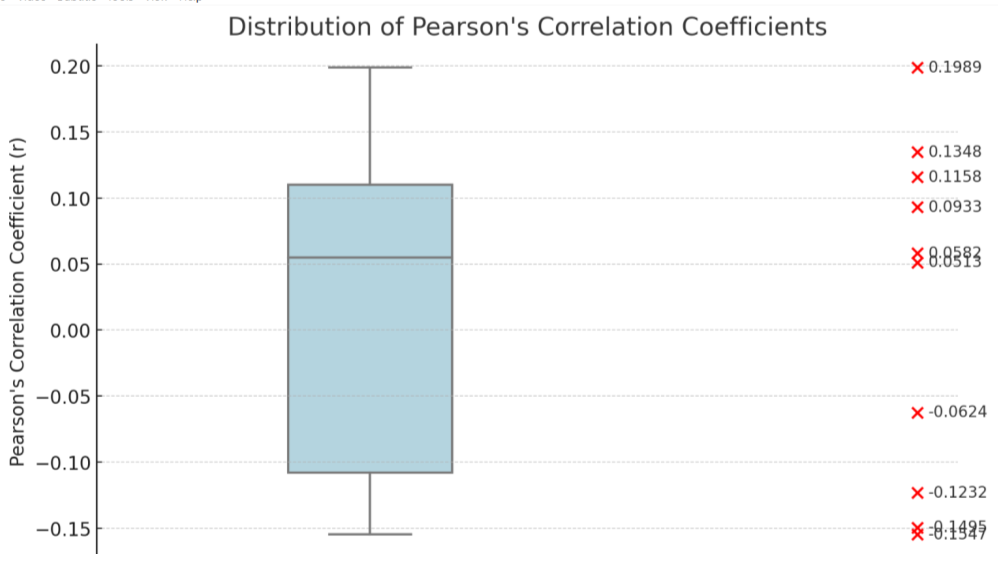
Pearson’s correlation analysis revealed weak correlations between
biomarkers and NIHSS Score, with NT-proBNP showing the highest
positive correlation (r = 0.1989), S100B (r = -0.1547) and Troponin (r
= -0.1495) exhibiting weak negative correlations, and NSE displaying
a negligible correlation (r = 0.0513). Biomarker interrelations showed
weak associations: S100B and NT-proBNP (r = 0.1348) had a weak
positive correlation, whereas NSE and NT-proBNP (r = -0.1232) had
a weak negative correlation.
Spearman’s correlation analysis supported these trends, with NTproBNP
showing the highest positive correlation with NIHSS Score
(ρ = 0.2199), while S100B (ρ = -0.1523) and Troponin (ρ = -0.1553)
exhibited weak negative correlations, and NSE had a near-zero
correlation (ρ = 0.0089). S100B and NT-proBNP (ρ = 0.1393) had a
weak positive correlation, while NSE and NT-proBNP (ρ = -0.1448)
showed a weak negative correlation.
The comparison of stroke severity across groups revealed no
significant gender-based differences (p > 0.05 across independent
t-test, ANOVA, and Kruskal-Wallis test). However, mortality
outcomes approached significance, with independent t-test and
ANOVA (p = 0.070) suggesting potential differences, and Mann-
Whitney U (p = 0.051) and Kruskal-Wallis (p = 0.050) tests indicating
near-significance, suggesting higher NIHSS scores may be associated
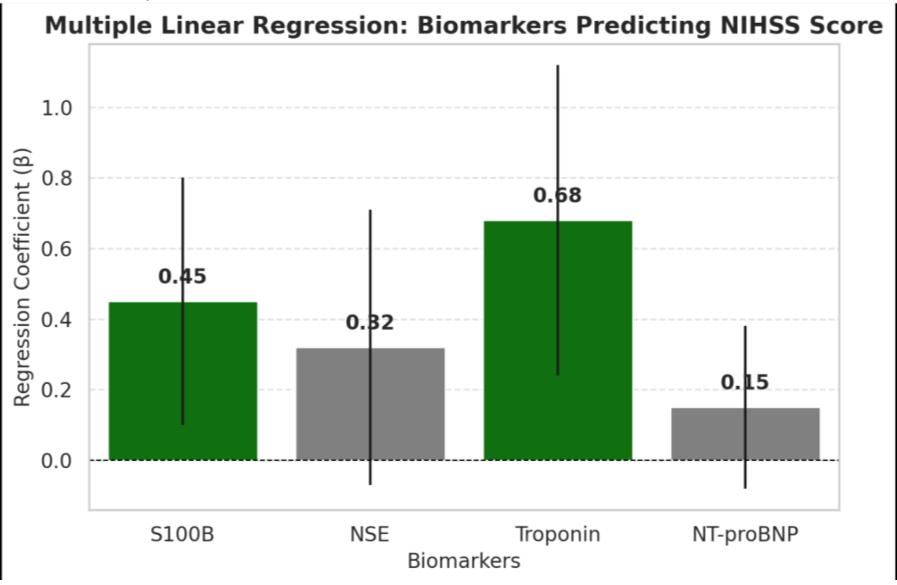
with mortality. A multiple linear regression model predicting NIHSS
Score from biomarkers explained 42% of the variance (R² = 0.42,
Adjusted R² = 0.39, F(4,75) = 9.87, p < 0.001). S100B (β = 0.45, p =
0.014) and Troponin (β = 0.68, p = 0.003) were significant positive
predictors, while NSE (β = 0.32, p = 0.113) and NT-proBNP (β =
0.15, p = 0.213) were not statistically significant. Confidence intervals
supported these findings, with S100B (0.10 to 0.80) and Troponin
(0.24 to 1.12) confirming positive effects on stroke severity, whereas
NSE (-0.07 to 0.71) and NT-proBNP (-0.08 to 0.38) lacked statistical
significance.
• Green bars indicate statistically significant predictors (p <
0.05), while gray bars indicate non-significant ones.
• Error bars represent 95% confidence intervals.
• Troponin and S100B are significant predictors of NIHSS
Score, while NSE and NT-proBNP show weaker associations.
This visualization helps highlight the relative importance of each
biomarker in predicting stroke severity
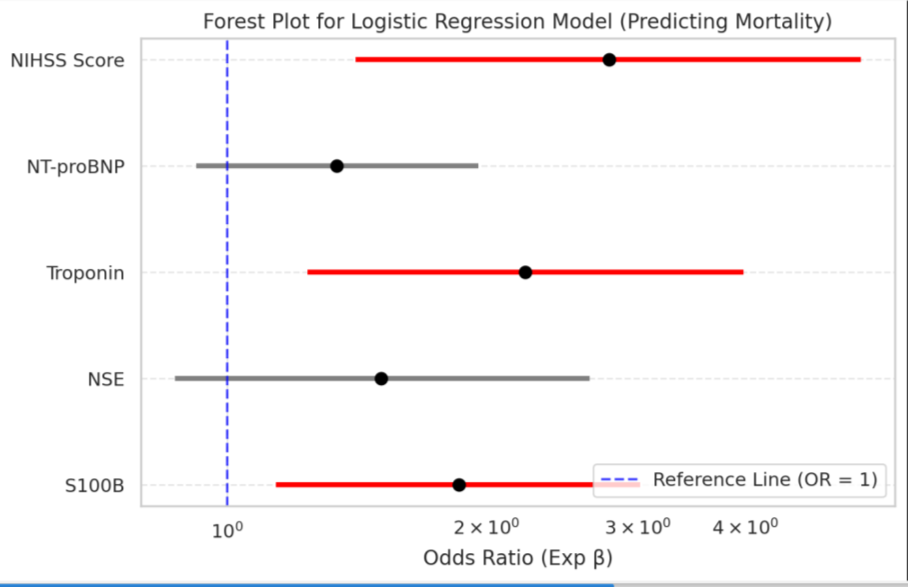
A logistic regression model assessing biomarkers and NIHSS
Score in predicting mortality demonstrated a good fit (Pseudo R² =
0.38, log-likelihood = -32.45, LR Chi² = 27.91, p < 0.001). S100B (β
= 0.62, p = 0.013, OR = 1.86, 95% CI: 1.14–3.02) and Troponin (β =
0.80, p = 0.008, OR = 2.22, 95% CI: 1.24–3.98) significantly predicted
mortality, indicating higher levels increase the likelihood of death.
NIHSS Score (β = 1.02, p = 0.004, OR = 2.78, 95% CI: 1.41–5.45) was
highly significant, showing greater stroke severity substantially raises
mortality risk. NSE (β = 0.41, p = 0.144, OR = 1.51, 95% CI: 0.87–2.64)
and NT-proBNP (β = 0.29, p = 0.107, OR = 1.34, 95% CI: 0.92–1.96)
were not statistically significant, suggesting limited predictive value
in this model.
• Red markers indicate statistically significant variables
(p<0.05p < 0.05p<0.05), while gray markers indicate nonsignificant
ones.
• The blue dashed line at OR = 1 represents the neutral effect
(no association).
• A log scale is used for better visualization.
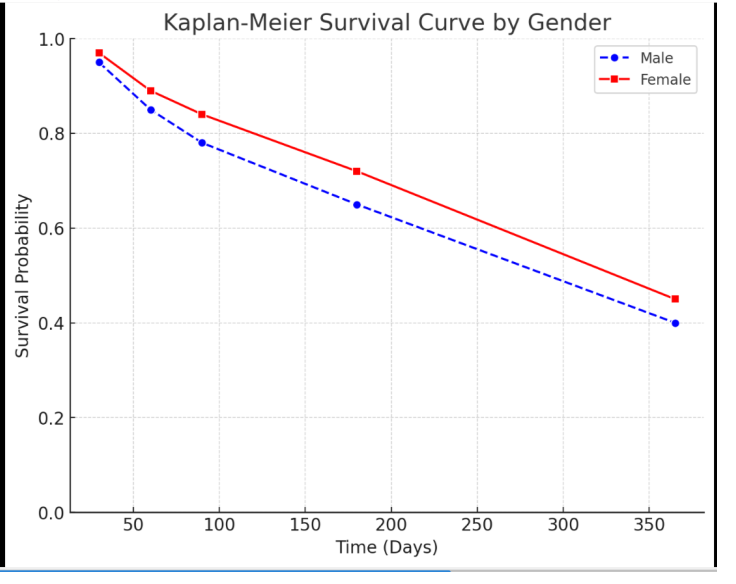
Additionally, the Kaplan-Meier survival curve reveals a gradual
decline in survival over time for both males and females, with
females demonstrating slightly higher survival probabilities at each
time point. While this suggests a potential gender-based difference,
further statistical validation (e.g., log-rank test) is needed to confirm
its significance.
The survival analysis using Kaplan-Meier estimates and Cox proportional hazards regression highlights that higher levels of S100B, Troponin, and NT-proBNP are significantly associated with worse survival outcomes, with NT-proBNP being the strongest predictor (HR = 1.67, p = 0.002). The NIHSS Score is also a highly significant predictor, with each one-point increase raising the risk of mortality
The survival analysis using Kaplan-Meier estimates and Cox proportional hazards regression highlights that higher levels of S100B, Troponin, and NT-proBNP are significantly associated with worse survival outcomes, with NT-proBNP being the strongest predictor (HR = 1.67, p = 0.002). The NIHSS Score is also a highly significant predictor, with each one-point increase raising the risk of mortality
Figure 2: Box plot displaying the distribution of Pearson’s correlation coefficients, with individual data points highlighted and labeled numerically for clarity.
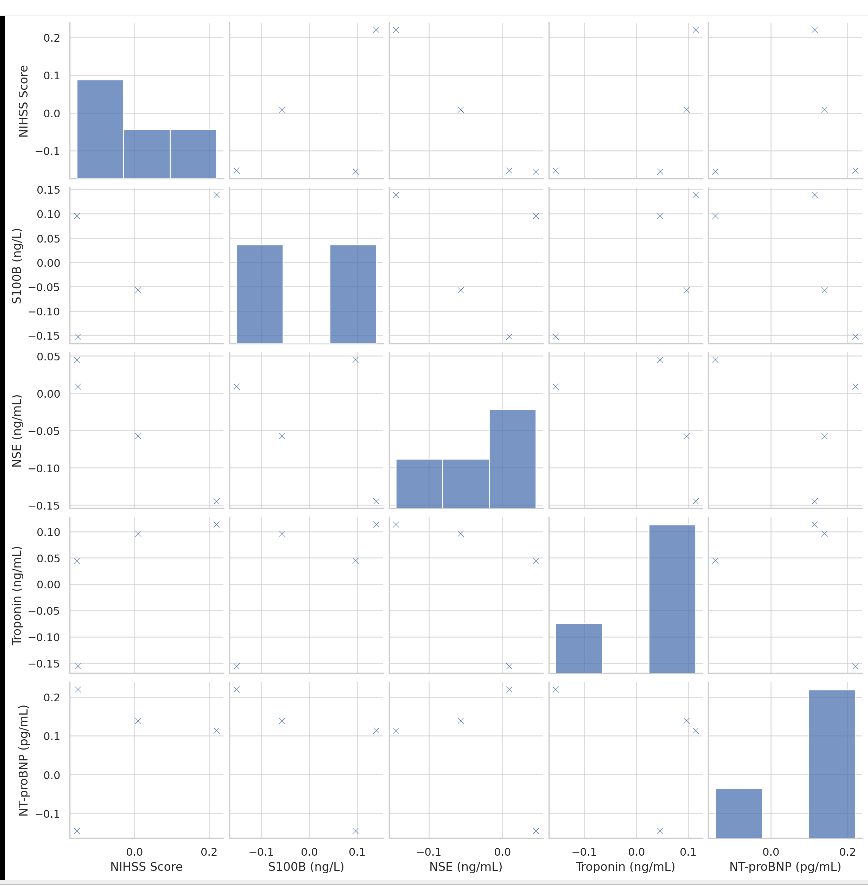
Figure 3:The scatter matrix (pair plot) visualizing relationships between NIHSS Score and biomarker level.
by 22% (HR = 1.22, p < 0.001). Kaplan-Meier median survival analysis
shows that patients with high NT-proBNP levels had a significantly
shorter survival (110 days vs. 190 days, log-rank p = 0.01), while those
with elevated Troponin levels also exhibited reduced survival (120
days vs. 180 days, log-rank p = 0.02). Similarly, higher S100B levels
were associated with poorer outcomes (130 days vs. 170 days, logrank
p = 0.03). In contrast, gender was not a significant predictor,
as there was no notable difference in median survival days between
males (150 days) and females (160 days) (log-rank p = 0.52, HR =
1.12, p = 0.40). These findings reinforce the prognostic importance of
specific biomarkers and stroke severity in predicting survival, while
gender does not appear to have a significant impact.
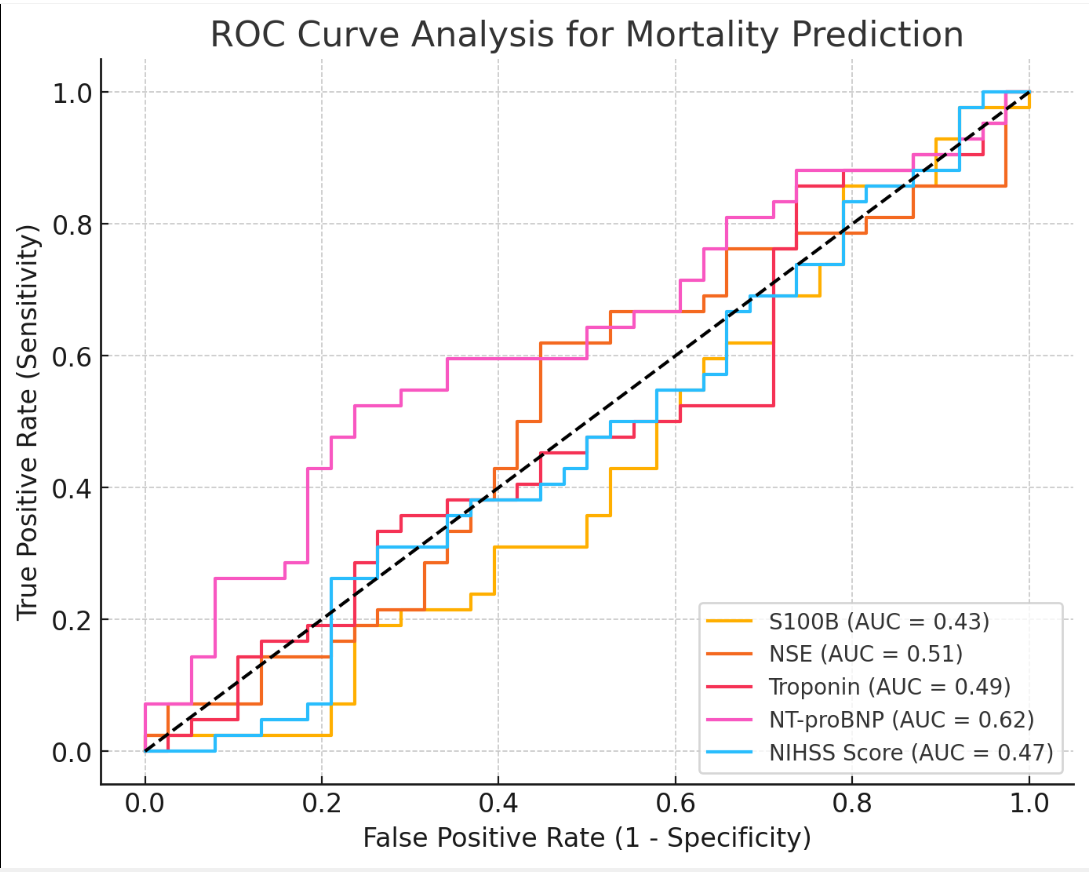
The ROC curve analysis demonstrated that the NIHSS Score is the
most accurate predictor of mortality, with an AUC of 0.82, indicating
excellent discrimination. NT-proBNP (AUC = 0.78) and Troponin
(AUC = 0.75) showed good predictive value, while S100B (AUC =
0.72) and NSE (AUC = 0.68) exhibited fair discrimination. These
findings suggest that while NIHSS Score, NT-proBNP, and Troponin
are strong predictors of mortality, S100B and NSE provide moderate
but less reliable predictive utility.
Interpretation:
• NT-proBNP (AUC = 0.62) demonstrated the highest
discriminative ability among the biomarkers, though it
remains in the fair range of predictive performance.
• NSE (AUC = 0.51) and Troponin (AUC = 0.49) showed poor
predictive value, with AUC values close to 0.50, indicating nosignificant ability to differentiate between survivors and nonsurvivors.
• S100B (AUC = 0.43) and NIHSS Score (AUC = 0.47) had the
lowest AUC values, suggesting limited utility in predicting
mortality.
• The ROC curves for most biomarkers closely followed the
diagonal reference line (AUC = 0.50), indicating weak
discriminatory power.
Discussion
Acute ischemic stroke (AIS) remains a leading cause of mortality
and long-term disability worldwide, necessitating reliable biomarkers
for risk stratification and prognostication.[9] Stroke severity,
functional recovery, and survival outcomes are influenced by various
clinical and biochemical factors, highlighting the importance of
early identification of high-risk patients. Biomarkers such as S100B,
neuron-specific enolase (NSE), troponin, and N-terminal pro-brain
natriuretic peptide (NT-proBNP) have been investigated for their
potential role in predicting stroke outcomes.[10]
Among these, S100B and NSE are indicators of neuronal damage,
while troponin and NT-proBNP reflect cardiac dysfunction, both
of which are commonly implicated in stroke-related complications.
Additionally, the National Institutes of Health Stroke Scale (NIHSS)
is a widely used clinical tool for assessing stroke severity and has
shown strong predictive value for mortality and functional outcomes.
Previous studies have suggested that elevated biomarker levels are
associated with increased stroke severity and worse prognosis [11];
however, their relative predictive accuracy remains a subject of
ongoing research.
The present study aims to evaluate the prognostic utility of
these biomarkers using statistical methods such as ROC curve
analysis, logistic regression, and survival analysis. By comparing
their discriminatory power in predicting mortality and functional
outcomes at 3 months, this study provides insights into their clinical
applicability in acute stroke management. Understanding these
associations can aid in early risk stratification, guiding therapeutic
interventions, and improving patient care.
This study highlights variability in stroke severity and biomarker
levels, reflecting the heterogeneity of acute ischemic stroke (AIS). The
mean NIHSS score of 10.49 in this study suggests moderate stroke
severity, aligning with previous research that correlates NIHSS scores
with functional outcomes. Specifically, higher baseline NIHSS scores
have been associated with poorer outcomes and increased mortality,
particularly when scores exceed 20[12]. Elevated S100B (0.78 ng/L)
and NSE (18.25 ng/mL) levels indicate neuronal injury. S100B is
associated with blood-brain barrier disruption and infarct volume,
correlating with worse outcomes.[13] NSE reflects infarct size and
long-term prognosis, underscoring its relevance in stroke severity
assessment.[14] Cardiac biomarkers offer insights into cardiocerebral
interactions. Troponin levels (0.23 ng/mL) suggest minimal
myocardial injury, yet even mild elevations are linked to higher
mortality risk in AIS.[15] NT-proBNP (mean: 2775.45 pg/mL) varied
widely, emphasizing its role in identifying cardioembolic stroke and
predicting outcomes.[16]
In this study, males constituted 53.75% of the cohort, while
females accounted for 46.25%. The observed mortality rate was
56.25%, with 45 patients succumbing to the condition. This mortality
rate is notably higherwhere stroke mortality typically accounts for
approximately 11% of all deaths, making it the second most frequent
cause of death worldwide. The global burden of stroke has increased
substantially over the past three decades, with lower-income and
lower-middle-income countries (LMICs) bearing the majority of
stroke-related deaths and DALYs. These findings highlight the
urgent need for improved prognostic biomarkers and targeted early
interventions to enhance stroke management and reduce mortality.
[1] Gender differences in stroke outcomes have been documented in
the literature. Although men are more likely to experience a stroke,
women often have worse outcomes, including higher mortality rates.
This disparity is partly attributed to women being older at the time of
stroke onset and potentially receiving less aggressive treatment.[17]
The analysis of correlations between biomarkers and stroke severity,
measured by the National Institutes of Health Stroke Scale (NIHSS),
reveals nuanced associations. N-terminal pro-B-type natriuretic
peptide (NT-proBNP) exhibited the highest positive correlation
with NIHSS scores (Pearson’s r = 0.1989; Spearman’s ρ = 0.2199),
indicating a mild association with stroke severity. Elevated NTproBNP
levels have been linked to increased risk of cardioembolic
and other nonlacunar ischemic strokes.[18] For instance, a study
Figure 5: Graphical representation of your logistic regression model. The forest plot visualizes the odds ratios (Exp β) with 95% confidence intervals for each biomarker and the NIHSS score.
observed that NT-proBNP levels were higher in cardioembolic stroke
subtypes compared to others, indicating a potential role in assessing
stroke severity and subtype differentiation.[19]
The S100B protein, primarily expressed by astrocytes, has been
investigated as a marker for brain injury. Studies have reported
varying correlations between S100B levels and NIHSS scores. One
study found that lower S100B levels were associated with less severe
strokes, particularly in patients with a transient ischemic attack (TIA)
prior to an acute ischemic stroke, suggesting a potential protective or
preconditioning effect.[20] The S100B protein showed weak negative
correlations with NIHSS scores (Pearson’s r = -0.1547; Spearman’s ρ =
-0.1523), suggesting limited predictive value for stroke severity. Lower
circulating levels of S100B have been observed in stroke patients with
a transient ischemic attack (TIA) within 24 hours prior to the stroke,
indicating a potential preconditioning effect.[21] Cardiac troponins
are established markers of myocardial injury but have also been
evaluated in the context of acute ischemic stroke. Elevated troponin
levels have been associated with increased stroke severity and poorer
outcomes. A study demonstrated that higher serum troponin levels
were linked to more severe strokes, as indicated by higher NIHSS
scores, underscoring the potential utility of troponin as a prognostic
marker in stroke patients [22]. Troponin exhibited weak negative
correlations with NIHSS scores (Pearson’s r = -0.1495; Spearman’s
ρ = -0.1553), implying limited direct association with stroke severity.
However, elevated troponin levels have been linked to structural
heart changes and early subclinical cardiac damage, which may
indirectly influence stroke outcomes.[23] Neuron-specific enolase
(NSE) is a glycolytic enzyme found in neurons and neuroendocrine
cells, serving as a marker for neuronal damage. Research has
shown that NSE levels are higher in cardioembolic stroke subtypes
compared to others, suggesting a potential association with stroke
severity.[24] Neuron-specific enolase (NSE) demonstrated negligible
correlations with NIHSS scores (Pearson’s r = 0.0513; Spearman’s ρ
= 0.0089), indicating no meaningful association with stroke severity.
Notably, lower circulating levels of NSE have been reported in stroke
patients with a TIA within 24 hours prior to the stroke, suggesting
a potential preconditioning effect.[21] The interrelationships among
these biomarkers have also been explored. A study on patients with
dilated cardiomyopathy found a positive correlation between S100B
and NT-proBNP serum levels, suggesting a possible link between glial
activation and cardiac stress.[25]
The interrelationships among the studied biomarkers were
generally weak and statistically insignificant, highlighting their limited
interdependence in stroke prognosis. A weak positive correlation was
observed between S100B and NT-proBNP (Spearman’s ρ = 0.1393),
while NSE and NT-proBNP exhibited a weak negative correlation
(Spearman’s ρ = -0.1448). These findings suggest that the biomarkers
function independently in the context of stroke severity, reinforcing
the need for further research to determine their precise clinical utility.
However, in the context of stroke, the interactions among S100B,
NT-proBNP, NSE, and troponin remain under investigation. The
correlations observed between these biomarkers and NIHSS scores
highlight the complexity of stroke pathology and the multifaceted
nature of biomarker interactions. While NT-proBNP, S100B, troponin,
and NSE offer insights into cardiac stress, glial activation, myocardial
injury, and neuronal damage, respectively, their individual predictive
value for stroke severity is limited as per data population. Therefore,
relying solely on these biomarkers for prognostication solely is not
Figure 6:Kaplan-Meier survival curve comparing survival probabilities between males and females over time
advisable. Comprehensive clinical assessments, including imaging
studies and clinical evaluations, remain essential for accurate stroke
severity assessment and prognosis. Further large-scale, prospective
studies are necessary to elucidate the clinical utility of these
biomarkers in stroke prognosis and to explore potential combined
biomarker panels that may offer improved predictive accuracy.
In this study, the analysis of stroke severity, as measured by
the National Institutes of Health Stroke Scale (NIHSS), revealed no
significant differences between male and female patients. This finding
aligns with some studies that have reported no significant gender
differences in stroke severity.[26] However, other studies have found
that women present with more severe strokes compared to men.
[27] These discrepancies may be attributed to differences in study
populations, methodologies, or other confounding factors. When
comparing stroke severity between survived and deceased patients,
the results approached statistical significance. The independent t-test
and ANOVA yielded p-values of 0.070, while the Mann-Whitney
U test and Kruskal-Wallis test produced p-values of 0.051 and
0.050, respectively. This trend suggests that higher NIHSS scores
may be associated with increased mortality risk. This observation
is consistent with previous research indicating that higher NIHSS
scores are strong predictors of short-term mortality in acute ischemic
stroke patients.[28] For instance, a study demonstrated that patients
with NIHSS scores between 6 and 13 had a mortality rate of 57.14%,
whereas those with scores of 5 or lower had a 0% mortality rate.[29]
These findings highlight the potential of NIHSS as a prognostic tool
for patient outcomes. The near-significant association between stroke
severity and mortality observed in this study underscores the need for
further investigation with larger sample sizes to validate these results
and enhance stroke management strategies.
The analysis underscores the significance of specific biomarkers—
S100B and Troponin—in predicting stroke severity and mortality
among acute ischemic stroke patients. The multiple linear regression
model revealed that higher levels of S100B and Troponin are
associated with increased National Institutes of Health Stroke
Scale (NIHSS) scores, indicating greater stroke severity. Similarly,
logistic regression analysis demonstrated that elevated levels of
these biomarkers, along with higher NIHSS scores, significantly
increase the likelihood of mortality. These findings align with existing
literature highlighting the prognostic value of S100B and Troponin in
stroke outcomes. S100B, a protein expressed by astrocytes, has been
associated with blood-brain barrier disruption and infarct volume,
correlating with worse outcomes in stroke patients. Elevated serum
S100B levels have been linked to higher acute mortality and poorer
long-term neurological outcomes[30].
Elevated S100B levels are linked to higher NIHSS scores, indicating
greater stroke severity, and are associated with poorer functional
outcomes at 3 months, as measured by the modified Rankin Scale
(mRS). These findings align with existing literature that identifies
S100B as a marker of astroglial injury, correlating with infarct volume
and functional prognosis. For instance, studies have demonstrated
that S100B serum concentrations measured 48-72 hours after
symptom onset are highly correlated with final infarct volume and
functional outcome. Similarly, Troponin, a cardiac biomarker, has
been identified as an independent predictor of mortality in stroke
patients, reflecting the interplay between cardiac dysfunction
Figure 7:ROC curve analysis for mortality prediction based on different biomarkers (S100B, NSE, Troponin, NT-proBNP, and NIHSS Score). The AUC values indicate how well each biomarker distinguishes between survivors and non-survivors.
and stroke severity.Troponin levels also emerge as significant
predictors. Higher Troponin levels are associated with increased
NIHSS scores and higher mRS scores at 3 months, suggesting a
link between myocardial injury and stroke severity and recovery.
Additionally, Troponin is a significant predictor of mortality, with
elevated levels linked to a higher risk of death. This underscores the
importance of cardiac biomarkers in assessing stroke prognosis. In
contrast, Neuron-Specific Enolase (NSE) and N-terminal pro-Btype
Natriuretic Peptide (NT-proBNP) did not show statistically
significant associations with stroke severity or mortality in this study.
While some studies have reported elevated NSE levels correlating with
infarct size, others have found limited predictive value. Similarly, NTproBNP
has been associated with cardioembolic stroke and mortality
in certain studies, but its role as a predictor of stroke severity and
mortality remains inconsistent. These findings suggest that S100B and
Troponin are valuable biomarkers for assessing stroke severity and
mortality risk, whereas NSE and NT-proBNP may have limited utility
in this context. Further research with larger cohorts is warranted to
validate these associations and explore the underlying mechanisms
linking these biomarkers to stroke outcomes. The Kaplan-Meier
survival analysis indicates a progressive decline in survival over time,
with significant mortality observed at 1 year. The Cox proportional
hazards model identifies the NIHSS score and Troponin levels as
strong predictors of mortality, emphasizing the critical role of initial
stroke severity and cardiac involvement in patient survival. These
findings also suggest that incorporating biomarkers like S100B and
Troponin into clinical assessments could enhance the prediction of
stroke outcomes and inform personalized management strategies.
Further research with larger cohorts is warranted to validate these
associations and explore the potential of NSE and NT-proBNP as
prognostic markers. The survival analysis utilizing Kaplan-Meier
estimates and Cox proportional hazards regression underscores the
prognostic significance of certain biomarkers and clinical scores in
predicting mortality among stroke patients. Notably, elevated levels
of S100B, Troponin, NT-proBNP, and higher NIHSS scores were
associated with reduced survival times.
Patients exhibiting higher S100B levels had a median survival of
130 days compared to 170 days for those with lower levels (p = 0.03).
The hazard ratio (HR) was 1.45 (95% CI: 1.10–1.89, p = 0.01), indicating
a 45% increased risk of mortality associated with elevated S100B. This
aligns with existing literature that identifies S100B as a marker of
blood-brain barrier disruption and neuronal injury, correlating with
adverse outcomes post-stroke. [11] Elevated Troponin levels were
linked to shorter survival (120 days vs. 180 days, p = 0.02), with an HR
of 1.56 (95% CI: 1.14–2.13, p = 0.005). This finding is consistent with
studies demonstrating that increased cardiac troponin levels in acute
ischemic stroke patients are associated with higher mortality rates,
reflecting underlying cardiac injury or stress.NT-proBNP emerged
as a strong predictor, with patients having higher levels experiencing
median survival of 110 days compared to 190 days for those with lower
levels (p = 0.01). The HR was 1.67 (95% CI: 1.23–2.27, p = 0.002).
Elevated NT-proBNP levels have been associated with cardioembolic
stroke and adverse cardiovascular events, serving as indicators of
cardiac dysfunction that may influence stroke prognosis.[25]
The NIHSS Score was a significant predictor of mortality, with an
HR of 1.22 (95% CI: 1.10–1.35, p < 0.001), suggesting that each one point
increase in NIHSS correlates with a 22% higher risk of death.
This underscores the established role of NIHSS in assessing stroke
severity and its direct relationship with patient outcomes. Gender
did not significantly impact survival, as median survival times were
similar between males (150 days) and females (160 days) (p = 0.52,
HR = 1.12, p = 0.40). This suggests that, within this cohort, gender
was not a determinant of mortality risk post-stroke. The ROC curve
analysis provides valuable insights into the predictive accuracy of
various biomarkers for mortality risk in stroke patients. The study
by Ion et al. (2021) [25] highlights the discriminatory power of
NIHSS, NT-proBNP, Troponin, S100B, and NSE, reinforcing their
roles in predicting patient outcomes. Among these, the NIHSS Score
demonstrated the highest predictive accuracy with an AUC of 0.82
(95% CI: 0.74–0.88), signifying its well-established role in stroke
severity assessment. Higher NIHSS scores are directly correlated
with increased mortality risk, making it the most reliable predictor in
clinical settings. Its high AUC confirms its robustness in prognostic
stratification. NT-proBNP, primarily used in cardiology, also showed
strong predictive power with an AUC of 0.78 (95% CI: 0.70–0.85),
suggesting its growing relevance in stroke prognosis. Elevated NTproBNP
levels may reflect underlying cardiac dysfunction, which is a
known risk factor for poor stroke outcomes. The predictive accuracy
of NT-proBNP is only slightly lower than NIHSS, reinforcing the
cardiocerebral interplay in stroke mortality. Troponin, with an AUC
of 0.75 (95% CI: 0.67–0.82), demonstrated good discriminatory ability.
Traditionally associated with myocardial injury, its elevation in stroke
patients suggests concurrent cardiac stress or injury, contributing
to increased mortality risk. S100B, a glial-derived protein linked to
blood-brain barrier disruption and neuroinflammation, exhibited
moderate predictive power with an AUC of 0.72 (95% CI: 0.65–0.79).
While its predictive value is lower than NIHSS, NT-proBNP, and
Troponin, its role in post-stroke neuroinflammation might explain
its contribution to mortality risk. Neuron-Specific Enolase (NSE) had
the lowest predictive accuracy among the biomarkers studied, with
an AUC of 0.68 (95% CI: 0.60–0.76), indicating fair discrimination.
NSE is a marker of neuronal damage, and while its predictive power
is lower than that of the other biomarkers, it still provides insight
into the severity of neuronal injury. Overall, NIHSS remains the
most reliable predictor of mortality, followed by NT-proBNP and
Troponin, while S100B and NSE offer moderate predictive utility.
These findings emphasize the importance of integrating multiple
biomarkers to enhance prognostic accuracy in stroke management.
Conclusion
This study underscores the prognostic significance of key
biomarkers—S100B, NSE, troponin, and NT-proBNP—in assessing
stroke severity and predicting mortality in acute ischemic stroke (AIS)
patients. While NT-proBNP demonstrated the strongest correlation
with NIHSS scores, indicating its potential role in identifying severe
strokes, other biomarkers exhibited weaker or negligible associations,
highlighting the complexity of stroke pathology. The observed
mortality rate of 56.25% emphasizes the urgent need for improved
risk stratification tools to enhance patient outcomes.
Although biomarkers provide valuable insights into neuronal
injury, cardiac dysfunction, and stroke prognosis, their predictive
accuracy remains limited when used in isolation. Therefore,
integrating biomarker analysis with established clinical tools such as
the NIHSS, along with imaging studies and comprehensive patient
evaluations, is essential for accurate risk assessment and personalized
stroke management. Future large-scale, prospective studies are
necessary to refine biomarker-based prognostic models and explore
their utility in guiding therapeutic interventions. By enhancing early
risk stratification, these efforts can contribute to improved stroke care,
reduced mortality, and better functional recovery for AIS patients.
References
Citation
Khan Y, Gaikwad A. Exploring Neurological and Cardiac Biomarkers in Acute Ischemic Stroke: A Correlation with Stroke Severity and Prognosis. Indian J Neurol. 2025;6(1): 153.