Research Article
Expert Perspectives on Levetiracetam use in Epilepsy Management Across India
Manjula S* and Krishna Kumar M
Department of Medical Services, Micro Labs Limited, Bangalore, Karnataka, India
*Corresponding author:Dr Manjula S, Department of Medical Services, Micro Labs Limited, Race Course Road, Bangalore, Karnataka, India. E-mail Id: drmanjulas@gmail.com
Copyright: © 2025 Manjula S, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background:Although there are several studies available regarding the efficacy and safety of levetiracetam for epilepsy management, there is a dearth of studies among clinicians in actual practice. This study aims to gather expert perspectives on the clinical use of levetiracetam for managing epilepsy in
Indian settings.
Methodology:This cross-sectional study was conducted with a 23-item questionnaire among clinicians specializing in epilepsy management. It explored clinical practice patterns, treatment preferences, and levetiracetam-specific usage. The survey also assessed adverse effects, measures on patient adherence, and use of levetiracetam in special populations. Data analysis employed descriptive statistics, with results reported as frequencies and percentages.
Results:The survey included 293 participants, with the majority (90%) preferring levetiracetam as the first-line treatment for newly diagnosed adult epilepsy. For pediatric partial seizures, 68% preferred it as their first choice. In clinical practice, 49% reported that 41–60% of their epilepsy patients are currently on levetiracetam. While 52% had no experience using it in rare epilepsy syndromes, 49% reported the same regarding its use during pregnancy. Additionally, 48% of experts observed that 6–10% of their patients with resistant epilepsy were already on optimal doses. According to 41% of clinicians, levetiracetam was most often preferred for generalized motor seizures. Behavioral changes were reported by 53% of respondents in fewer than 10% of patients, and 67% considered 1000 mg/day the optimal dose, balancing efficacy and side effects.
Conclusion:The survey underscores the strong preference of levetiracetam among clinicians as an antiepileptic drug for both adult and pediatric epilepsy. Its broad clinical applicability and favorable safety profile contribute significantly to its widespread adoption.
Methodology:This cross-sectional study was conducted with a 23-item questionnaire among clinicians specializing in epilepsy management. It explored clinical practice patterns, treatment preferences, and levetiracetam-specific usage. The survey also assessed adverse effects, measures on patient adherence, and use of levetiracetam in special populations. Data analysis employed descriptive statistics, with results reported as frequencies and percentages.
Results:The survey included 293 participants, with the majority (90%) preferring levetiracetam as the first-line treatment for newly diagnosed adult epilepsy. For pediatric partial seizures, 68% preferred it as their first choice. In clinical practice, 49% reported that 41–60% of their epilepsy patients are currently on levetiracetam. While 52% had no experience using it in rare epilepsy syndromes, 49% reported the same regarding its use during pregnancy. Additionally, 48% of experts observed that 6–10% of their patients with resistant epilepsy were already on optimal doses. According to 41% of clinicians, levetiracetam was most often preferred for generalized motor seizures. Behavioral changes were reported by 53% of respondents in fewer than 10% of patients, and 67% considered 1000 mg/day the optimal dose, balancing efficacy and side effects.
Conclusion:The survey underscores the strong preference of levetiracetam among clinicians as an antiepileptic drug for both adult and pediatric epilepsy. Its broad clinical applicability and favorable safety profile contribute significantly to its widespread adoption.
Keywords:Epilepsy; Seizures; Antiepileptic Drugs; Levetiracetam
Introduction
Epilepsy is a chronic neurological disorder marked by recurrent
seizures, affecting approximately 50 million people globally and
ranking among the most prevalent neurological conditions. Nearly
80% of individuals with epilepsy live in low- and middle-income
countries, where the condition poses a significant burden on both
individuals and healthcare systems.[1,2] With an incidence of 50 per
100,000 person-years and a prevalence of 700 per 100,000, epilepsy
contributes to over 0.5% of the global disease burden, accounting
for 2.4 million new cases and 20.6 million DALYs lost annually.
[2,3] In India, prevalence estimates range from 3.0 to 11.9 per 1,000
population, with an incidence of 0.2 to 0.6 per 1,000 per year, making
it the third most common disabling neurological disorder after
stroke and headache.[4,5] The presence of a substantial treatment
gap, especially among children, highlights epilepsy as a major public
health concern in the country.[1]
Individuals with epilepsy face up to a threefold increased risk of
premature death compared to the general population; however, with
accurate diagnosis and appropriate treatment, up to 70% can achieve
seizure freedom.[1] Antiepileptic drugs (AEDs) remain the primary
treatment for epilepsy, with approximately 70% of patients achieving
effective seizure control through medication alone.[4] Levetiracetam
(LEV), a second-generation AED from the pyrrolidone class, is
recognized for its broad spectrum of activity. As a novel antiseizure
medication, LEV acts mainly on synaptic vesicle protein 2A (SV2A),
which helps regulate the balance of neurotransmitters in the brain.
This action modulates neuronal excitability and inhibition, thereby
reducing the excessive activity that can trigger seizures. Approved
by the FDA in the early 2000s, LEV has since been widely used to
manage both focal and generalized seizures, including myoclonic
seizures in children and adults. Numerous studies have evaluated its
efficacy and safety across various seizure types. LEV has demonstrated
effectiveness as both monotherapy and adjunctive therapy, including
in the prevention of early post-traumatic seizures. [6,7]
There is a lack of studies on clinicians’ perspectives on LEV in
epilepsy management. This study assessed the clinical practices and
physician preferences in epilepsy management, with a focus on the use
of LEV. It also aims to assess treatment patterns, drug effectiveness,
patient adherence, and barriers to care.
Methods
We carried out a cross-sectional study among clinicians
specialized in treating epilepsy patients in the major Indian cities
from June 2024 to December 2024. The study was conducted after
receiving approval from Bangalore Ethics, an Independent Ethics
Committee, which is recognized by the Indian Regulatory Authority,
the Drug Controller General of India.
Questionnaire
The questionnaire booklet titled LIVIA (Levetiracetam Efficacy
and Tolerability Profile) was sent to the physicians who were
interested in participating in this study. The study questionnaire
included 23 questions that explored various domains, including
demographics and clinical practice patterns such as common epilepsy
types, age group prevalence, and gender-specific seizure trends. It
also examined treatment preferences for newly diagnosed epilepsy
and add-on therapy, LEV-specific usage patterns, including preferred
seizure types and dosing strategies, and adverse effects with their
management. Additional areas covered included patient adherence,
use in special populations like pregnant and pediatric patients, and
strategies for patient education and healthcare accessibility. Survey
questions were developed using the methods designed to collect
perspectives from the practitioners. Reliability as determined by a
split-half test (coefficient alpha) was adequate but should be improved
in future versions of the questionnaire. A study of criterion validity
was undertaken to test the questionnaire and to develop methods of
testing the validity of measures of Physicians’ Perspectives. However,
the extraneous variable in this includes the clinician’s experience,
usage of the newer drugs etc. The two criteria used were the doctors’
perspectives from the clinical practice and the assessment of an
external assessor and statistician.
Participants:
A convenience sampling method was adopted, where an invitation
was sent to leading neurologists who were experts in managing
epilepsy in the month of March 2024 for participation in this Indian
survey. About 293 doctors from major cities of all Indian states,
representing the geographical distribution, shared their willingness
to participate and provide necessary data. Those physicians were
included and asked to complete the questionnaire without discussing
it with their peers. A written informed consent was obtained from
each neurologist prior to initiation of the study. Clinicians who were
experts in other specialities and who had not provided informed
consent were excluded from the study.Statistical analysis:
The data were analyzed using descriptive statistics. Categorical
variables were presented as percentages to depict their distribution.
The frequency of occurrence and the corresponding percentage were
used to represent the distribution of each variable. To visualize the
distribution of the categorical variables, bar charts were created using
Microsoft Excel 2013 (version 16.0.13901.20400).Results
The survey included 293 experts, with the majority of clinicians
(58.7%) indicating that generalized motor seizures are the most
commonly encountered form of epilepsy in their clinical practice.
Around 58% reported that epilepsy is most frequently seen in
individuals aged 19-45 years. Nearly half of the clinicians (47.1%)
observed that partial seizures, with or without impaired sensorium,
are more common in females than in males. According to 48% of
experts, individual counselling is the preferred method for educating
patients about epilepsy-related myths, facts, and awareness. More
than half of the clinicians (51.54%) believed that 6–15% of patients
with epilepsy are still unable to access appropriate healthcare facilities
for management.
Approximately 50% of respondents indicated that between 26–
50% of their patients with epilepsy adhere to prescribed medication
and follow-up visits. Almost half of the experts (49.15%) observed
that 21-30% of patients with partial-onset seizures do not respond to
older antiepileptic medications in their clinical practice. Around 58%
strongly endorsed the view that interventions such as pill reminders,
tele-calling, and push messages can significantly improve patient
compliance with medication. Over half of the clinicians (53.92%)
indicated that they manage epileptic seizures in children under 10
years of age and prefer initiating treatment with first-generation
AEDs.
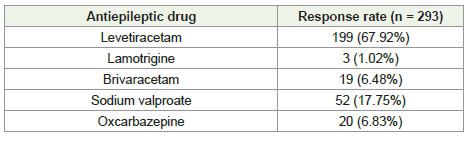
According to 68% of experts, LEV is the most preferred AED for
managing partial seizures in pediatric patients [Table 1]. The majority
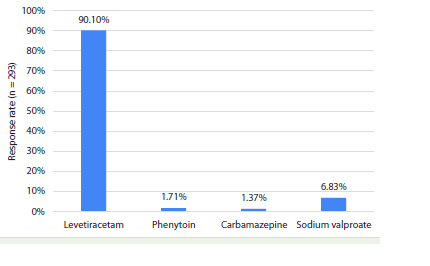
of the clinicians (90.1%) expressed a preference for LEV as the firstline
treatment for adult patients with newly diagnosed epilepsy
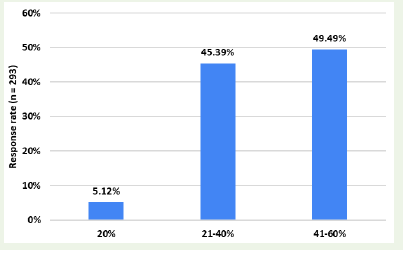
[Figure 1]. Nearly half of the clinicians (49.49%) stated that 41–60%
of their epilepsy patients are currently being treated with LEV in their
clinical practice [Figure 2].
Figure 1:Distribution of responses to the most preferred drug among
clinicians for adult patients with newly diagnosed epilepsy
Figure 2:Distribution of responses to the proportion of patients on
levetiracetam therapy in clinical practice
Table 1:Distribution of responses to clinicians’ most preferred antiepileptic drug
for partial seizures in the pediatric age group
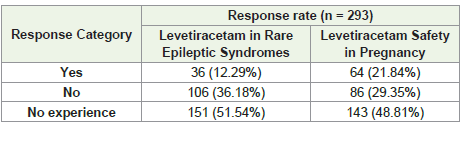
Nearly 52% of experts reported having no experience with LEV
in managing rarer forms of epileptic syndromes, while almost half
(48.81%) similarly indicated no experience regarding its safety
during pregnancy [Table 2]. Around 46% stated that 26–50% of their
epilepsy patients are adherent to both medication and follow-up visits
in routine clinical practice. Approximately 48% observed that 6–10%
of their patients with resistant or refractory epilepsy are already
receiving an optimal dose of LEV [Figure 3].
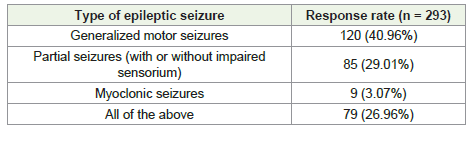
About 41% of clinicians most often preferred LEV for treating
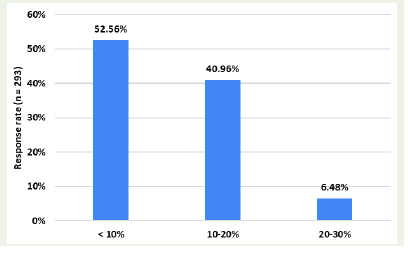
generalized motor seizures [Table 3]. More than half (52.56%)
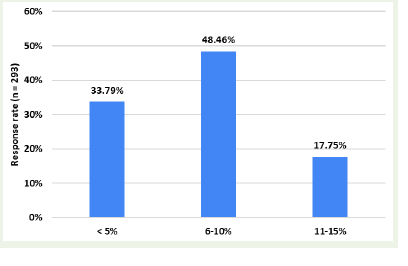
indicated that fewer than 10% of their patients on LEV exhibit
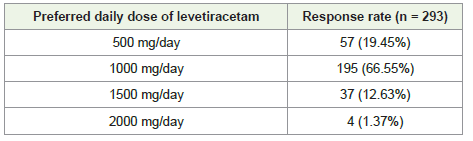
behavioral changes [Figure 4]. Two-thirds of the clinicians (66.55%)
identified 1000 mg/day as the optimal dose of LEV, considering both
seizure control and the risk of side effects [Table 4].
Over half of the clinicians (54.27%) indicated that they typically
Over half of the clinicians (54.27%) indicated that they typically
Figure 3:Distribution of responses to clinicians’ observations on the proportion
of refractory epilepsy patients already on the optimal dose of levetiracetam
Figure 4:Distribution of responses to clinicians’ observations on behavioral
changes in patients on levetiracetam
Table 2:Distribution of responses to clinicians’ views on the usefulness of
levetiracetam in rare epileptic syndromes and its safety during pregnancy
Table 4:Distribution of responses to clinicians preferred optimal dose of
levetiracetam balancing seizure control and side effects
manage 5-10 epilepsy patients per month who require an addon
AED. Nearly half (49.15%) favored lacosamide as the secondgeneration
AED of choice for patients whose seizures were not
adequately controlled with first-line medications. Around 39%
reported drowsiness as the most frequently encountered side effect
of brivaracetam in the treatment of epileptic seizures. Approximately
42% of clinicians attributed the behavioral changes associated with
LEV to an unknown mechanism.
Discussion
The survey results underscore a strong clinical preference for LEV
as a first-line AED across diverse patient populations. A considerable
proportion of clinicians favored its use in newly diagnosed adult
epilepsy, reflecting its widespread acceptance in adult treatment
protocols. Additionally, its preference for managing partial seizures
in pediatric patients highlights its broad applicability across age
groups.
In accordance with the current survey findings, multiple studies
have supported the efficacy and tolerability of LEV across various
age groups and epilepsy types. Alsaadi et al. reported the efficacy
and tolerability of LEV therapy in patients with newly diagnosed
epilepsy.[8] Similarly, Karlov et al. demonstrated that LEV is an
effective monotherapy for newly diagnosed focal epilepsy (FE) and
idiopathic generalized epilepsy (IGE), with significant reductions in
epileptiform activity and high monotherapy retention rates (87.6% in
IGE and 73.8% in FE).[9] An Indian open-label study reported that
62.96% of newly diagnosed partial seizure patients achieved seizure
control at 6 months with LEV monotherapy at a dose of 20 mg/kg/
day.[10]
A randomized controlled trial in India, involving 100 neonates
with clinical seizures, found that LEV offered superior seizure
control (86% vs. 62%) compared to phenobarbital, with no reported
adverse drug reactions.[11] Additionally, a systematic review and
meta-analysis of randomized controlled trials reported that, as
monotherapy, LEV was more advantageous than carbamazepine
(CBZ) for pediatric focal epilepsy (PFE), with a lower frequency of
seizures and fewer dermatological adverse events.[12] Another metaanalysis
confirmed that add-on LEV significantly reduces seizure
frequency and is well tolerated in children with focal-onset seizures
compared to placebo.[13]
The study data revealed that nearly half of clinicians prescribed
LEV to 41–60% of their epilepsy patients, highlighting its practical
advantages, strong tolerability, and consistent therapeutic effectiveness
in everyday practice. However, almost 49% of respondents lacked
experience regarding its safety during pregnancy, a key consideration
given that AED exposure can affect fetal development at any stage.
[14] A hospital-based retrospective study from a tertiary care
center in Northeast India concluded that LEV monotherapy is a
safer option for women of childbearing age with active epilepsy.
[15] Similarly, a retrospective study conducted in Northern India
involving 99 pregnant women with epilepsy receiving monotherapy
found that none of the infants exposed to LEV exhibited congenital
malformations.[16] Supporting these findings, results from the UK
and Ireland Epilepsy and Pregnancy Registers, which included a
substantial cohort of first-trimester LEV exposures, confirmed that
LEV monotherapy during pregnancy is associated with a low risk of
major congenital malformations.[17]
More than half of clinicians (51.54%) reported no experience
using LEV in rare epileptic syndromes. However, clinical and expert based
evidence supports its efficacy in such conditions. A North
American expert consensus on Dravet syndrome supports LEV as a
later-line option. While not recommended as first-line therapy, it was
deemed moderately effective, alongside clonazepam and zonisamide,
for patients unresponsive to standard treatments, highlighting its
potential as an adjunct in drug-resistant cases.[18] Similarly, in an
open-label, multicenter observational trial involving 55 pediatric
patients with Lennox-Gastaut syndrome (LGS), add-on LEV led to a
greater than 50% reduction in seizure frequency in 58.2% of patients,
with 27.3% becoming seizure-free.[19] These findings indicate that
LEV may provide therapeutic benefit even in syndromes with which
many clinicians have limited experience.
According to the current survey findings, LEV is commonly
utilized at optimal doses in the management of refractory epilepsy,
reflecting its relevance in more difficult-to-treat cases. Its side effect
profile is generally favorable, with behavioral changes reported
in only a minority of patients. A daily dose of 1000 mg is often
considered optimal, offering a balance between therapeutic efficacy
and tolerability. Supporting these observations, Chen et al. reported
that add-on LEV in 65 children with refractory epilepsy led to a
rapid and significant reduction in seizures with minimal side effects,
suggesting its suitability in pediatric cases.[20] Similarly, Viteva et al.
evaluated LEV as adjunct therapy in 135 patients with drug-resistant
epilepsy and found a sustained reduction in seizure frequency, mild
improvement in seizure severity, and a favorable safety profile.[21]
A phase III randomized controlled trial by Manreza et al. confirmed
that LEV, at doses of 1,000–3,000 mg/day for adults or 60 mg/kg/day
for children, is effective and safe for treating refractory focal epilepsy.
[22] Grant et al. observed a significant reduction in median seizure
frequency across all dosing levels, with patients experiencing fewer
weekly seizures compared to placebo.[23] Similarly, Cereghino et
al. reported a significantly higher response rate, defined as a ≥50%
reduction in seizure frequency, in patients receiving 1,000 mg/day of
LEV(33.0%) versus placebo (10.8%, P < 0.001).[24]
In the present survey, the majority of clinicians preferred LEV
for treating generalized motor seizures, showcasing its versatility.
A multicenter, randomized, double-blind, placebo-controlled trial
by Berkovic et al. noted that adjunctive LEV significantly reduced
generalized tonic-clonic seizures in idiopathic generalized epilepsy,
with 72.2% achieving ≥50% seizure reduction versus 45.2% in the
placebo group (p < 0.001; OR 3.28; 95% CI: 1.68-6.38).[25] Similarly,
a study by Krauss et al. found that LEV led to significant seizure
reduction in patients with generalized seizures, including tonicclonic,
myoclonic, and absence seizures, who had failed multiple
anticonvulsants, with 42% achieving complete seizure freedom.[26]
These findings reinforce LEV’s role as an effective option across a
spectrum of generalized seizure types.
This survey offers valuable insights into clinicians’ perspectives
in actual practice on LEV use in epilepsy management, revealing
significant observations supporting its role as a first-line therapy and
aligning with current treatment guidelines. A key strength lies in the
use of a validated questionnaire that captured expert opinions within
an evidence-based framework, thereby supporting informed clinical
decision-making. However, reliance on expert opinion introduces
potential bias, as individual experiences and preferences may
have influenced responses. Additionally, as a survey-based study,
it is subject to response and recall bias, with findings potentially
reflecting the characteristics of the sampled clinicians rather than the
broader medical community. Future directions include conducting
prospective studies or randomized controlled trials to validate
these results, ideally by integrating objective measures of efficacy,
tolerability, and patient outcomes.
Conclusion
The survey highlights LEV’s strong standing as a preferred
AED among clinicians for both adult and pediatric epilepsy. Its
broad application and favorable side effect profile contribute to its
widespread use. However, the reported lack of experience in treating
rarer epileptic syndromes and during pregnancy points to areas
where further research and clinician education are warranted to
optimize patient care.
Acknowledgement:
We would like to thank all the clinicians who participated in this
studyReferences
Citation
Manjula S, Kumar MK. Expert Perspectives on Levetiracetam use in Epilepsy Management Across India. Indian J Neurol. 2025;6(1): 150.