Review Article
Hidden Nutritional Risks of Rapid Weight Loss: A Clinical and Nutraceutical Framework for Prevention in Pharmacological and Surgical Obesity Treatments
Bellani D*, Desai A, Anand S, Maldahiar SP, Mishra S and Hegde R
Department of Medical Affairs, IntelliMed Healthcare Solutions Sakhi House 2nd Floor, Corporate Park, VN Purav Marg, Swastik Chamber, Chembur, Mumbai, Maharashtra, India
*Corresponding author:Depti Bellani, Department of Medical Affairs, IntelliMed Healthcare Solutions Sakhi House 2nd Floor, Corporate Park, VN Purav Marg, Swastik Chamber, Chembur, Mumbai, Maharashtra India. E-mail Id: depti.bellani@intellimed.org
Article Information:Submission: 09/04/2026; Accepted: 24/04/2026; Published: 28/04/2026
Copyright: ©2026 Bellani D, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: Obesity management has rapidly expanded with glucagon-like peptide-1 receptor agonists (GLP-1 RAs), dual incretin therapies, alongside established bariatric surgery. Although these interventions induce durable weight loss, emerging evidence indicates, rapid weight reduction carries nutritional and metabolic consequences inadequately addressed in current clinical frameworks.
Objective: To synthesize existing evidence on the mechanisms, prevalence, and clinical impact of nutritional deficiencies associated with GLP-1–based therapies and bariatric surgery, and to propose an integrated, evidence-based approach to nutritional monitoring and supplementation.
Methods: This narrative review integrates data from randomized controlled trials, meta-analyses, observational studies, and international guidelines, including those from the American Diabetes Association (ADA), Endocrine Society, American Heart Association/American College of Cardiology/The Obesity Society (AHA/ACC/TOS), American Society for Metabolic and Bariatric Surgery (ASMBS), International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO), British Obesity and Metabolic Surgery Society (BOMSS), and Indian Council of Medical Research (ICMR).
Results: Pharmacologic and surgical weight-loss modalities converge on pathways of reduced intake, altered gastrointestinal physiology, and systemic metabolic adaptation, resulting in a high protein and micronutrient deficiency risk. Evidence demonstrates that one-fourth of weight loss may derive from fat free mass, with clinically significant deficiencies emerging within the first year of therapy. While bariatric care incorporates structured nutritional surveillance and supplementation, pharmacologic pathways lack comparable protocols despite achieving similar magnitudes of weight loss. This gap is particularly relevant in Indian populations, where high baseline prevalence of micronutrient deficiency and distinct body composition profiles amplify vulnerability.
Conclusion: Rapid weight loss should be reframed as a state of heightened nutritional risk requiring proactive, structured management. Bariatric-style monitoring, individualized supplementation, and body composition assessment must be integrated into GLP-1–based care pathways to preserve metabolic benefits while preventing nutritional compromise. Future research should prioritize trials evaluating supplementation strategies and long-term nutritional outcomes in treated populations.
Objective: To synthesize existing evidence on the mechanisms, prevalence, and clinical impact of nutritional deficiencies associated with GLP-1–based therapies and bariatric surgery, and to propose an integrated, evidence-based approach to nutritional monitoring and supplementation.
Methods: This narrative review integrates data from randomized controlled trials, meta-analyses, observational studies, and international guidelines, including those from the American Diabetes Association (ADA), Endocrine Society, American Heart Association/American College of Cardiology/The Obesity Society (AHA/ACC/TOS), American Society for Metabolic and Bariatric Surgery (ASMBS), International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO), British Obesity and Metabolic Surgery Society (BOMSS), and Indian Council of Medical Research (ICMR).
Results: Pharmacologic and surgical weight-loss modalities converge on pathways of reduced intake, altered gastrointestinal physiology, and systemic metabolic adaptation, resulting in a high protein and micronutrient deficiency risk. Evidence demonstrates that one-fourth of weight loss may derive from fat free mass, with clinically significant deficiencies emerging within the first year of therapy. While bariatric care incorporates structured nutritional surveillance and supplementation, pharmacologic pathways lack comparable protocols despite achieving similar magnitudes of weight loss. This gap is particularly relevant in Indian populations, where high baseline prevalence of micronutrient deficiency and distinct body composition profiles amplify vulnerability.
Conclusion: Rapid weight loss should be reframed as a state of heightened nutritional risk requiring proactive, structured management. Bariatric-style monitoring, individualized supplementation, and body composition assessment must be integrated into GLP-1–based care pathways to preserve metabolic benefits while preventing nutritional compromise. Future research should prioritize trials evaluating supplementation strategies and long-term nutritional outcomes in treated populations.
Keywords:GLP-1 receptor agonists; Dual incretin therapy; Bariatric surgery; Rapid weight loss; Nutritional deficiency; Sarcopenia
Introduction
Rationale for Focusing on GLP-1 Therapies and Bariatric Surgery:
Obesity care has entered an era where we can transform body
weight faster than we are prepared to protect nutritional health.
Targeted metabolic interventions now enable sustained, clinically
meaningful weight loss, marking a new landmark in obesity
management. GLP-1 receptor agonists (GLP-1 RAs) and dual
incretin therapies achieve 15-25% reductions in body weight with
broad metabolic, cardiovascular, and renal benefits [1,2]. Modern
bariatric procedures deliver similar or greater effects, with durable
>20% weight loss and substantial reductions in long-term morbidity
and mortality [3,4].
However, this degree of weight loss reflects systemic metabolic
adaptation rather than selective adiposity loss. Approximately 25%
of total weight reduction arises from loss of fat-free mass [5]. Both
therapeutic modalities constrain nutrient intake and absorption—
GLP-1 agents through appetite suppression, delayed gastric emptying
[6,7], and bariatric surgery via anatomical alteration that impairs
uptake of iron, calcium, vitamin B12, and fat-soluble vitamins [8].
Deficiencies emerge early and are clinically meaningful.
Observational data report up to 12.7% deficiency in six months of
GLP-1 therapy, rising to 22.4% in one year, predominantly involving
protein, vitamin D, and iron, with downstream effects on strength,
bone health, and metabolic resilience [9].
These risks are amplified in Indian populations, characterized by
high baseline micronutrient deficiency and the “thin–fat” phenotype
visceral adiposity with low lean mass at comparatively lower BMI
thresholds [10,11]. Vegetarian dietary patterns further predispose
to vitamin B12, iron, and vitamin D insufficiency, heightening
vulnerability during rapid treatment-induced weight loss.
Despite parallel metabolic efficacy, pharmacologic guidelines,
including those from the American Diabetes Association (ADA), the
Endocrine Society Clinical Practice Guidelines, and the American
Heart Association/American College of Cardiology/The Obesity
Society (AHA/ACC/TOS), rarely mandate structured nutritional
surveillance. This stands in contrast to bariatric protocols from the
American Society for Metabolic and Bariatric Surgery (ASMBS),
the International Federation for the Surgery of Obesity and
Metabolic Disorders (IFSO), and the British Obesity and Metabolic
Surgery Society (BOMSS), which embed lifelong monitoring and
supplementationScope:
This review examines adults undergoing rapid weight loss with
GLP-1 RAs, dual incretin therapies, and bariatric surgery, focusing
on the interplay between metabolic efficacy and nutritional risk.
Particular attention is given to population-specific risk in Indian
cohorts, where baseline micronutrient insufficiency and body composition
patterns modify vulnerability.Methods:
Evidence was synthesized narratively from randomized
controlled trials, meta-analyses, observational cohorts, and major
international and national guidelines, including AHA/ACC/TOS,
Endocrine Society, ADA Standards of Care, ASMBS, IFSO, BOMSS,
and ICMR. Particular emphasis was placed on DEXA-based bodycomposition
studies and longitudinal assessments of micronutrient
status to capture the qualitative as well as quantitative dimensions
of weight loss. Integration prioritized mechanistic coherence, clinical
relevance, and applicability to real-world practice, with explicit
consideration of the Indian context.Pathophysiology of Nutritional Risk:
GLP-1 RAs and bariatric surgery induce weight loss through
distinct but converging biological pathways that establish a sustained
state of negative energy and nutrient balance. In GLP-1–based
therapy, weight reduction is mediated by coordinated central and
peripheral mechanisms. Activation of hypothalamic and brainstem
circuits suppresses appetite and enhances satiety, while modulation
of reward pathways reduces hedonic feeding [12,13]. Peripherally,
delayed gastric emptying prolongs gastric distension and alters meal
patterns, contributing to sustained reductions in intake [6,14].
These effects extend beyond caloric restriction to influence
nutrient exposure and gastrointestinal processing. Reduced gastric
motility alters the timing and distribution of nutrients delivered to
the small intestine, potentially modifying the absorptive environment
for minerals such as iron and calcium [14]. Concurrent hormonal
adaptations, including changes in insulin and glucagon signaling,
further influence substrate utilization and nutrient partitioning
[15,16].
Bariatric procedures achieve weight loss through structural
modification of the gastrointestinal tract, combining restrictive,
malabsorptive, and hormonal mechanisms. Restrictive components
reduce intake, while malabsorptive procedures such as Roux-en-Y
gastric bypass exclude the duodenum and proximal jejunum—primary
sites of absorption for iron, calcium, and several micronutrients [17].
Altered bile acid circulation and reduced pancreatic enzyme mixing
impair fat digestion and absorption, affecting fat-soluble vitamins
[8]. In addition, diminished gastric acid and intrinsic factor secretion
limit vitamin B12 availability [18].
Despite differing mechanisms, both modalities converge on
a shared physiological state characterized by reduced nutrient
availability, altered absorption dynamics, and metabolic adaptation.
Sustained negative nitrogen balance, increased proteolysis, and
attenuated anabolic signaling collectively predispose to loss of lean
tissue when nutrient intake is inadequate [11,19]. Hormonal shifts
involving insulin, leptin, and sex steroids further influence bone
remodeling and metabolic regulation, linking weight loss to skeletal
vulnerability [1,20].
Collectively, these mechanisms establish the biological substrate
for downstream nutritional deficiency, sarcopenia, and metabolic
adaptation observed during rapid weight loss.Guideline Landscape: Obesity, GLP-1 RAs, and Bariatric Surgery:
The management of obesity is anchored in major international
guidelines that frame obesity as a chronic, multifactorial disease
requiring long-term, staged intervention. The AHA/ACC/TOS and
Endocrine Society recommendations endorse a stepwise escalation
from lifestyle therapy to pharmacotherapy and, when indicated,
bariatric surgery, guided by treatment response and comorbidity
burden [21,22]. ADA Standards of Care position GLP-1 RAs as core
agents for patients with obesity and type 2 diabetes, citing robust
evidence for weight reduction and cardiovascular and renal risk
modification [23,24].
Potent incretin-based therapies, including semaglutide and dual
GLP-1/GIP agonists, now achieve 15–25% sustained weight loss with
broad cardiometabolic benefit, approaching outcomes traditionally
associated with bariatric surgery [2,25]. This convergence in
efficacy extends the relevance of surgical-style risk considerations to
pharmacologic pathways.By contrast, bariatric surgery is embedded within detailed nutritional guidance from ASMBS, IFSO, and BOMSS, which explicitly recognize micronutrient deficiency as an inherent consequence of altered gastrointestinal anatomy [26,27]. These guidelines mandate preoperative nutritional assessment, procedure specific supplementation, and lifelong biochemical surveillance of iron, vitamin B12, calcium, vitamin D, and fat-soluble vitamins, supported by data demonstrating persistent deficiencies and progressive bone loss [3,27,28]. Recommended schedules typically include monitoring at 3–6 month intervals in the first postoperative year and annually thereafter [27].
Pharmacologic guidelines, in contrast, exhibit minimal integration of structured nutritional risk management. ADA, Endocrine Society, and AHA/ACC/TOS documents offer detailed metabolic algorithms but do not require routine micronutrient testing or systematic supplementation for patients treated with GLP-1–based therapies; nutritional advice remains largely generic and non-protocolized [21-23]. This is increasingly misaligned with emerging evidence that GLP-1–induced weight loss is accompanied by reduced intake of protein and micronutrient-rich foods, gastrointestinal intolerance, and quantifiable deficits in vitamin D, iron, and protein status, with deficiency prevalence rising from 12.7% at six months to 22.4% at one year [29-31] Concurrent DEXA data showing 30-40% of total weight loss attributable to fat-free mass loss parallel observations in bariatric cohorts [32,33].
In this context, the absence of structured nutritional frameworks within pharmacologic obesity care pathways represents a critical gap. Bariatric guidelines anticipate and manage nutritional risk through protocolized monitoring and supplementation, whereas pharmacologic pathways remain predominantly focused on glycemic and cardiometabolic endpoints [3,22,27].
The Indian context further accentuates this asymmetry. ICMR and related data highlight the coexistence of obesity with widespread micronutrient deficiency and the “thin-fat” or Asian Indian phenotype—characterized by higher visceral adiposity and lower lean mass at comparatively lower BMI levels [34]. High baseline prevalence of vitamin B12, iron, and vitamin D deficiency amplifies vulnerability to additional nutritional depletion during rapid, GLP-1–mediated guidelines that frame obesity as a chronic, multifactorial disease requiring long-term, staged intervention. The AHA/ACC/TOS and Endocrine Society recommendations endorse a stepwise escalation from lifestyle therapy to pharmacotherapy and, when indicated, bariatric surgery, guided by treatment response and comorbidity burden [21,22]. ADA Standards of Care position GLP-1 RAs as core agents for patients with obesity and type 2 diabetes, citing robust evidence for weight reduction and cardiovascular and renal risk modification [23,24].
Potent incretin-based therapies, including semaglutide and dual GLP-1/GIP agonists, now achieve 15–25% sustained weight loss with broad cardiometabolic benefit, approaching outcomes traditionally associated with bariatric surgery [2,25]. This convergence in efficacy extends the relevance of surgical-style risk considerations to pharmacologic pathways.
By contrast, bariatric surgery is embedded within detailed nutritional guidance from ASMBS, IFSO, and BOMSS, which explicitly recognize micronutrient deficiency as an inherent consequence of altered gastrointestinal anatomy [26,27]. These guidelines mandate preoperative nutritional assessment, procedure specific supplementation, and lifelong biochemical surveillance of iron, vitamin B12, calcium, vitamin D, and fat-soluble vitamins, supported by data demonstrating persistent deficiencies and progressive bone loss [3,27,28]. Recommended schedules typically include monitoring at 3–6 month intervals in the first postoperative year and annually thereafter [27].
Pharmacologic guidelines, in contrast, exhibit minimal integration of structured nutritional risk management. ADA, Endocrine Society, and AHA/ACC/TOS documents offer detailed metabolic algorithms but do not require routine micronutrient testing or systematic supplementation for patients treated with GLP-1–based therapies; nutritional advice remains largely generic and non-protocolized [21-23]. This is increasingly misaligned with emerging evidence that GLP-1–induced weight loss is accompanied by reduced intake of protein and micronutrient-rich foods, gastrointestinal intolerance, and quantifiable deficits in vitamin D, iron, and protein status, with deficiency prevalence rising from 12.7% at six months to 22.4% at one year [29-31] Concurrent DEXA data showing 30-40% of total weight loss attributable to fat-free mass loss parallel observations in bariatric cohorts [32,33].
In this context, the absence of structured nutritional frameworks within pharmacologic obesity care pathways represents a critical gap. Bariatric guidelines anticipate and manage nutritional risk through protocolized monitoring and supplementation, whereas pharmacologic pathways remain predominantly focused on glycemic and cardiometabolic endpoints [3,22,27].
The Indian context further accentuates this asymmetry. ICMR and related data highlight the coexistence of obesity with widespread micronutrient deficiency and the “thin-fat” or Asian Indian phenotype—characterized by higher visceral adiposity and lower lean mass at comparatively lower BMI levels [34]. High baseline prevalence of vitamin B12, iron, and vitamin D deficiency amplifies vulnerability to additional nutritional depletion during rapid, GLP-1–mediated weight loss, yet current Indian frameworks do not embed structured nutritional surveillance within pharmacologic pathways [35].
Overall, the guideline landscape reveals a pronounced asymmetry: bariatric surgery is supported by comprehensive, nutritioncentered protocols, whereas pharmacologic regimens achieving comparable weight loss lack analogous structures. Systematic integration of nutritional monitoring, risk stratification, and targeted supplementation into GLP-1–based care is therefore essential to align practice with evolving evidence and safeguard the long-term benefits of weight reduction.
Nutritional Deficiencies with GLP-1–Based Weight Loss:
Nutritional deficiency during GLP-1 RA –mediated weight
loss reflects the clinical manifestation of sustained reductions in
dietary intake and altered eating patterns. Incretin-based therapies
are associated with significant decreases in total caloric intake,
accompanied by disproportionate reductions in protein consumption
and micronutrient-rich food groups [9,30]. These intake patterns
frequently fall below recommended thresholds required to maintain
physiological function.
Observational data consistently demonstrate inadequate intake
of protein, calcium, and iron, alongside widespread vitamin D
insufficiency, often superimposed on pre-existing deficiency [30].
These deficits are compounded over time, with the prevalence of
clinically significant deficiency increasing from 12.7% at six months
to 22.4% at one year of therapy [9].
The clinical consequences of these deficiencies extend beyond
biochemical abnormalities. Patients frequently exhibit fatigue,
reduced exercise tolerance, and impaired metabolic resilience,
reflecting the systemic impact of inadequate nutrient availability.
Of particular importance is the loss of lean body mass. Dual-energy
x-ray absorptiometry (DXA) body analysis demonstrates that
approximately one-fourth of total weight loss during GLP-1 therapy
is attributable to reductions in fat-free mass [5]. This loss is associated
with diminished strength, reduced resting energy expenditure, and
increased risk of sarcopenia, particularly in older individuals and
those with baseline vulnerability [31,32].
In the Indian context, these risks are amplified by high
baseline prevalence of micronutrient deficiency and dietary
patterns characterized by variable protein intake. Deficiencies in
vitamin B12, iron, and vitamin D are common even in otherwise
healthy individuals, increasing susceptibility to further nutritional
compromise during treatment [35].
Despite this growing body of evidence, current pharmacologic
guidelines provide limited direction on routine nutritional monitoring
or supplementation in GLP-1–treated populations, highlighting
a critical gap between emerging clinical data and established care
pathways.Nutritional Deficiencies after Bariatric Surgery:
Nutritional deficiency after bariatric surgery is a predictable,
multifactorial consequence of profound anatomical and physiological
alterations in the gastrointestinal tract. In contrast to pharmacologic
weight loss, where deficits predominantly reflect reduced intake,
bariatric procedures directly disrupt digestion and absorption,
creating a sustained and often lifelong risk of nutrient depletion.
Pre-existing deficiencies are frequent at baseline, particularly for
vitamin D, iron, and vitamin B12, reflecting dietary inadequacy and
obesity-related metabolic disturbances, with an even higher burden
in Indian populations characterized by pervasive micronutrient
insufficiency and limited dietary diversity [35,36]. Procedure type
critically shapes deficiency profiles: sleeve gastrectomy mainly
restricts intake and lowers gastric acid, while Roux-en-Y gastric
bypass combines restriction with malabsorption via exclusion of
the duodenum and proximal jejunum—key sites for iron, calcium,
and micronutrient absorption; more extensive operations such as
biliopancreatic diversion confer even greater malabsorptive burden
[3,8].
These structural changes disrupt multiple stages of nutrient
assimilation. Bypass of the proximal intestine impairs iron and
calcium uptake; altered bile acid circulation and limited pancreatic
enzyme mixing reduce fat-soluble vitamin absorption; and
diminished gastric acidity compromises liberation and subsequent
intrinsic factor–mediated absorption of vitamin B12 [3]. Deficiencies
evolve over time; postoperative phases are dominated by low
intake and rapid weight loss, whereas long-term deficits reflect
persistent malabsorption and poor adherence to supplementation,
with longitudinal data confirming that abnormalities can persist or
progress, necessitating lifelong surveillance [37].
Clinical consequences are systemic and clinically significant:
iron deficiency anemia reduces functional capacity; vitamin B12
deficiency contributes to neurological dysfunction; and calcium and
vitamin D deficiency drive increased bone turnover, osteopenia, and
elevated fracture risk, with documented reductions in bone mineral
density after surgery [37,38]. Additional deficits in fat-soluble
vitamins and trace elements further impair immune competence
and metabolic function. These risks underpin ASMBS and BOMSS
recommendations for routine biochemical monitoring and lifelong
supplementation with multivitamins, iron, calcium, vitamin D,
and vitamin B12, though long-term adherence remains challenging
[26,37].
The contrast with pharmacologic weight-loss pathways is striking:
bariatric care embeds standardized nutritional surveillance and
supplementation, whereas GLP-1–based therapy lacks comparable
frameworks despite overlapping physiological consequences. This
disparity supports extending bariatric principles of structured
monitoring and supplementation to patients undergoing GLP-1–
mediated weight loss.Monitoring Frameworks and Risk Stratification:
The management of rapid weight loss requires structured
monitoring frameworks that extend beyond weight-centric metrics
to include nutritional status, body composition, and functional
reserve. Given the established risk of nutritional compromise across
both pharmacologic and surgical modalities, systematic surveillance
is essential to ensure safe and sustainable outcomes [32].Bariatric surgery provides a well-defined model for such
monitoring. Guidelines from ASMBS and BOMSS mandate
comprehensive biochemical assessment beginning in the preoperative
period and continuing lifelong, reflecting the predictable trajectory
of nutritional risk [26,27]. Monitoring protocols are aligned with
clinically relevant parameters, including iron indices, vitamin B12,
calcium, vitamin D, and, where appropriate, parathyroid hormone,
enabling early identification of deficiency and targeted intervention
[8,20]. Recommended intervals include assessment every three to six
months during the first year and annually thereafter [27].
Conversely, structured monitoring for GLP-1–based therapy
remains insufficiently integrated into clinical practice. Emerging
expert consensus supports baseline nutritional assessment followed
by periodic evaluation of key micronutrients and protein status,
particularly in individuals experiencing significant or sustained
weight loss [39]. Adoption of such protocols represents a necessary
extension of existing pharmacologic care pathways.
Body composition assessment provides an important adjunct
to biochemical monitoring. Techniques such as DEXA enable
quantification of lean and fat mass, offering insight into qualitative
changes in body composition that are not captured by total weight
alone [19]. Incorporation of these measures facilitates early
identification of sarcopenia and informs targeted nutritional and
exercise interventions.
Risk stratification enables tailoring of monitoring intensity. Individuals undergoing malabsorptive bariatric procedures represent the highest-risk group and require intensive surveillance. However, patients receiving GLP-1 therapy may similarly warrant closer monitoring in the presence of rapid weight loss, reduced intake, advanced age, or comorbid conditions [19]. In the Indian context, high baseline prevalence of micronutrient deficiency necessitates routine baseline screening and lower thresholds for ongoing monitoring and supplementation [10,11] . In resource-constrained settings, simplified laboratory panels and integration into primary care pathways may enhance feasibility, while preserving the core principle that nutritional monitoring is integral to obesity management. Collectively, a unified, risk-stratified monitoring approach across both surgical and pharmacologic pathways enables early detection of deficiency, timely intervention, and preservation of the metabolic benefits of weight loss.
Risk stratification enables tailoring of monitoring intensity. Individuals undergoing malabsorptive bariatric procedures represent the highest-risk group and require intensive surveillance. However, patients receiving GLP-1 therapy may similarly warrant closer monitoring in the presence of rapid weight loss, reduced intake, advanced age, or comorbid conditions [19]. In the Indian context, high baseline prevalence of micronutrient deficiency necessitates routine baseline screening and lower thresholds for ongoing monitoring and supplementation [10,11] . In resource-constrained settings, simplified laboratory panels and integration into primary care pathways may enhance feasibility, while preserving the core principle that nutritional monitoring is integral to obesity management. Collectively, a unified, risk-stratified monitoring approach across both surgical and pharmacologic pathways enables early detection of deficiency, timely intervention, and preservation of the metabolic benefits of weight loss.
Evidence-Based Supplementation Strategies: Core Nutrients:
An evidence-based supplementation framework for rapid weight
loss should parallel established bariatric guidance while integrating
emerging data from GLP-1–treated populations. Core principles
include early initiation, risk- and procedure-specific tailoring, and
sustained adherence across the course of weight loss and maintenance.
Protein is foundational for preserving lean mass in the context
of rapid, catabolic weight loss. GLP-1– and surgery-induced weight
reduction is consistently associated with substantial loss of fat-free
mass, driven by increased proteolysis and reduced muscle protein
synthesis [19]. Bariatric and nutrition guidelines support protein
intakes of 1.0–1.5 g/kg/day, with higher targets of 1.2–2.0 g/kg/day
increasingly advocated in GLP-1–treated and high-risk patients
to mitigate sarcopenia and maintain function[40]. Given frequent
shortfalls due to appetite suppression and gastrointestinal intolerance,
high-quality protein supplements (e.g., whey, casein, or equivalent
alternatives) are often required to meet these thresholds [41].
Targeted anabolic support can further optimise muscle
preservation. Leucine, via mTOR activation, and its metabolite
β-hydroxy β-methylbutyrate (HMB) have demonstrated attenuation
of muscle protein breakdown and improved lean mass retention
in catabolic settings, supporting their use as adjuncts during rapid
weight loss[42]. Creatine, through augmentation of intramuscular
phosphocreatine stores, enhances ATP availability, strength, and
lean mass, particularly when combined with resistance training—
an approach that may be especially valuable in GLP-1–treated
individuals at risk of functional decline [43].
Micronutrient supplementation remains a cornerstone of
bariatric care and is directly applicable to pharmacologic weight
loss. Routine use of a complete multivitamin–mineral preparation,
together with targeted vitamin D, calcium, iron, and vitamin B12, is
strongly recommended in bariatric guidelines due to high prevalence
and clinical impact of deficiencies [8,26]. Vitamin D deficiency is
ubiquitous and exacerbated by reduced intake and pre-existing
insufficiency; supplementation supports bone and muscle health but
may not fully prevent bone mineral density loss. Calcium is essential
to counteract negative calcium balance and bone resorption,
particularly in malabsorptive states [28].
Iron deficiency is frequent across both bariatric and GLP-
1 pathways, driven by reduced intake, impaired absorption, and
increased physiological demands; declines in ferritin and hemoglobin
in GLP-1 cohorts emphasize the need for individualized oral or
parenteral iron strategies [31]. Vitamin B12 deficiency, arising
from reduced gastric acid, intrinsic factor–dependent absorption,
and suboptimal intake, warrants routine monitoring and oral
or parenteral replacement, especially in vegetarian and Indian
populations with high baseline risk [44]. Additional micronutrients
such as zinc, selenium, and fat-soluble vitamins contribute to
immune and metabolic function and are appropriately covered
within comprehensive multivitamin regimens recommended by
bariatric societies [27].
Overall, supplementation should begin early, be guided by
baseline status and anticipated risk, and be adapted to treatment
modality and patient characteristics. Extending bariatric-style,
protocolized supplementation strategies to GLP-1–treated patients
represents a necessary evolution to safeguard metabolic benefits while
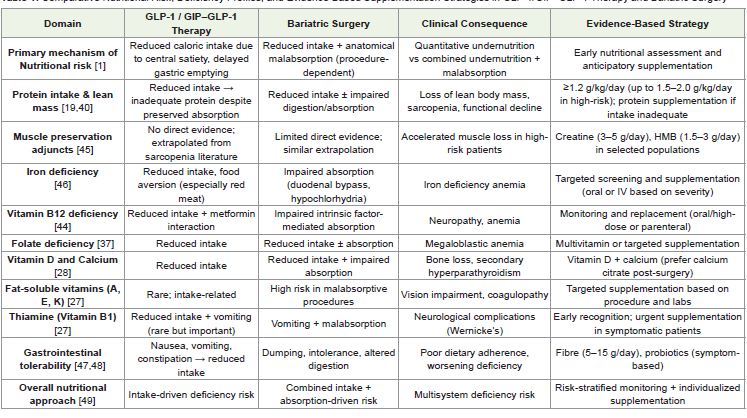
preventing avoidable nutritional compromise [8]. A consolidated
comparison of nutritional risk, deficiency profiles, and evidence based
supplementation strategies across GLP-1–based therapies and
bariatric surgery is summarized in [Table 1].Targeted Nutraceuticals and Adjunctive Therapies:
Omega-3 Fatty AcidsOmega-3 fatty acids provide complementary metabolic and antiinflammatory benefits. Meta-analytic data show dose-dependent reductions in triglycerides, supporting their role in cardiovascular risk modification during weight loss. Additional effects on inflammation and lipid metabolism further justify omega-3 supplementation in patients with elevated cardiometabolic risk [50,51].
Fiber Supplements and Probiotics:
Rapid weight loss often reduces fiber intake due to lower food
volume and tolerance, contributing to constipation and microbiota
perturbation. Soluble fiber supplementation improves bowel
regularity and may enhance satiety and glycemic control [47].
Probiotics can favourably modulate gut microbial composition, with
emerging evidence for benefits in metabolic regulation and immune
function [48].Integrating GLP-1 and Bariatric Frameworks: A Practical Algorithm:
The evolving obesity-treatment landscape supports a shift from
modality-specific pathways to an integrated metabolic framework
in which pharmacologic and surgical strategies lie on a single
continuum. Both GLP-1 RAs and bariatric surgery converge on
shared mechanisms—reduced intake, altered gastrointestinal
physiology, and systemic metabolic adaptation—yet only bariatric
care is underpinned by structured nutritional protocols [52]. Bariatric
guidelines from ASMBS and BOMSS provide comprehensive
models of baseline assessment, scheduled monitoring, and lifelong
supplementation [26,27], whereas ADA, Endocrine Society, and
AHA/ACC/TOS guidance largely prioritizes metabolic outcomes
and under-addresses nutritional surveillance, despite rising evidence
of deficiency in GLP-1–treated cohorts [21-23]. This divergence is
increasingly incongruent with data showing comparable weight-loss
magnitude and physiological impact between pharmacologic and
surgical approaches [2,3].
An integrated algorithm should begin with a comprehensive
baseline assessment, including diet history, micronutrient
biochemistry, and, where feasible, body composition analysis.
Baseline deficiencies are common across both groups and are
particularly prevalent in Indian populations, where vitamin B12,
iron, and vitamin D deficiency are highly prevalent [35]. Subsequent
risk stratification then guides monitoring intensity: malabsorptive
bariatric procedures occupy the highest-risk tier [27], but GLP-1–
treated patients may transition into higher-risk strata with rapid or
sustained weight loss, marked intake reduction, advanced age, frailty,
or chronic disease [32].
Monitoring intervals can reasonably mirror bariatric protocols,
three to six monthly reviews in the first year, followed by annual
surveillance[27] with emerging consensus supporting similar
schedules in GLP-1–treated individuals experiencing substantial or
prolonged weight loss [49]. Supplementation should be proactive and
individualized: core use of multivitamins, vitamin D, calcium, iron,
and vitamin B12 in high-risk patients, titrated against biochemical
results, with lower thresholds for initiation in GLP-1 cohorts
given the rapid emergence of deficiencies [29]. Integration of body
composition metrics is essential, as DEXA data show a substantial
proportion of GLP-1–mediated weight loss derives from fat-free
Table 1:Comparative Nutritional Risk, Deficiency Profiles, and Evidence-Based Supplementation Strategies in GLP-1/GIP–GLP-1 Therapy and Bariatric Surgery
mass, enabling early recognition of sarcopenia and timely nutritional
and exercise intervention [5,32].
Contextual adaptation is crucial in India, where high baseline micronutrient deficiency and variable protein intake amplify risk [35,53]. Here, baseline screening carries heightened priority, empiric supplementation may be appropriate even without overt biochemical deficiency, and simplified monitoring panels integrated into routine practice can enhance feasibility. A unified, risk-stratified algorithm spanning GLP-1 therapy and bariatric surgery thus aligns care with emerging evidence, embedding structured monitoring, early supplementation, and body composition assessment to preserve the metabolic benefits of weight loss while minimizing nutritional harm.
Contextual adaptation is crucial in India, where high baseline micronutrient deficiency and variable protein intake amplify risk [35,53]. Here, baseline screening carries heightened priority, empiric supplementation may be appropriate even without overt biochemical deficiency, and simplified monitoring panels integrated into routine practice can enhance feasibility. A unified, risk-stratified algorithm spanning GLP-1 therapy and bariatric surgery thus aligns care with emerging evidence, embedding structured monitoring, early supplementation, and body composition assessment to preserve the metabolic benefits of weight loss while minimizing nutritional harm.
Special Clinical Scenarios:
The use of weight-loss therapies in heterogeneous clinical
populations introduces additional complexity, as comorbidities,
physiological states, and demographic factors modify both efficacy
and safety. In these settings, nutritional management assumes
heightened importance to preserve physiological reserve alongside
metabolic benefit.In heart failure, GLP-1 RAs may improve metabolic and cardiovascular profiles but operate within a complex pathophysiological milieu characterized by altered energy metabolism and, in advanced stages, cardiac cachexia with skeletal muscle wasting [54,55]. Rapid weight loss in this setting may further exacerbate loss of lean body mass unless protein intake, anabolic support, and resistance exercise are optimized, with concurrent attention to fluid balance and electrolyte homeostasis, particularly in patients receiving diuretic therapy.
Frailty represents a high-risk state in which low physiological
reserve, baseline sarcopenia, and micronutrient deficits converge. In
older adults, rapid weight loss can precipitate functional decline unless
protein intake, vitamin D status, and structured resistance training
are actively intensified [18].Women of child-bearing potential
require additional precaution: bariatric guidance recommends
deferring pregnancy until weight stabilizes and nutritional adequacy
is confirmed, and analogous caution is warranted with GLP-1 therapy
given the potential impact of folate, iron, and vitamin B12 deficiency
on fetal development. Preconception counseling and targeted
supplementation are essential [56,57].
In the Indian context, vegetarian dietary patterns, variable protein intake, and high baseline prevalence of vitamin B12, iron, and vitamin D deficiency magnify nutritional risk during GLP- 1–induced appetite suppression or post-bariatric restriction [35]. Socioeconomic constraints and variable access to nutrient-dense foods further reinforce the need for proactive supplementation and culturally tailored dietary counseling.
In the Indian context, vegetarian dietary patterns, variable protein intake, and high baseline prevalence of vitamin B12, iron, and vitamin D deficiency magnify nutritional risk during GLP- 1–induced appetite suppression or post-bariatric restriction [35]. Socioeconomic constraints and variable access to nutrient-dense foods further reinforce the need for proactive supplementation and culturally tailored dietary counseling.
Implementation in Multidisciplinary Care:
Effective implementation of nutritional strategies in rapid weight
loss requires a multidisciplinary model that integrates medical,
nutritional, and behavioral expertise. The complexity of nutritional
risk across GLP-1–based therapy and bariatric surgery exceed the
scope of single-discipline care, necessitating coordinated involvement
of physicians, dietitians, nurses, and allied health professionals.Clinicians are responsible not only for initiating pharmacologic or surgical treatment but also for longitudinal oversight of nutritional status, including baseline assessment, interpretation of biochemical results, and dynamic adjustment of supplementation. Dietitians operate these strategies by translating guidance into individualized meal plans, troubleshooting intake barriers, and reinforcing adherence through structured follow-up. Behavioral support is integral. Appetite suppression, gastrointestinal symptoms, and lifestyle constraints frequently undermine adherence to diet and supplementation. Targeted interventions such as counseling, motivational interviewing, and digital tools help sustain behavioral change and optimize long-term adherence. In the Indian context, implementation must account for variable access to specialists, resource limitations, and heterogeneous care pathways. Pragmatic solutions include embedding nutritional care in primary practice, leveraging telemedicine, and using communitybased programs to extend reach and continuity. Patient education remains pivotal. Individuals undergoing rapid weight loss should clearly understand their risk of nutritional deficiency and the rationale for monitoring and supplementation. Informed, engaged patients are more likely to participate proactively and achieve durable clinical benefits.
Knowledge Gaps and Future Research Agenda:
Despite major advances in obesity therapeutics, the evidence base
for nutritional management remains incomplete, particularly for
GLP-1–based weight loss. Bariatric surgery is supported by extensive
data on mechanisms, prevalence, and treatment of deficiencies,
whereas evidence for GLP-1 therapies is comparatively nascent.
A key gap is the absence of randomized controlled trials testing
structured supplementation in GLP-1–treated populations. Current
practice largely extrapolates from bariatric cohorts or observational
data, limiting the precision of guidance. Future trials should evaluate
protein supplementation, micronutrient replacement, and targeted
nutraceuticals with endpoints that include lean mass preservation,
functional capacity, and quality of life, not only weight change.
Long-term nutritional trajectories under prolonged GLP-1
therapy are also poorly defined. Although deficiencies within the first
year have been documented [9], their persistence, progression, and
clinical consequences over multi-year treatment require clarification.
Integration of body composition outcomes, particularly DEXAderived
lean and fat mass changes, is essential to capture qualitative
aspects of weight loss beyond total weight alone [19].
In India, population-specific research is a priority. Heterogeneity
in dietary patterns, high baseline micronutrient deficiency, and sociocultural
determinants of diet demand locally generated evidence to
inform context-appropriate guidelines and algorithms.
Finally, translational and implementation research is needed to
bridge evidence and practice. The effectiveness of multidisciplinary
care models, digital health solutions, and community-based strategies
in improving nutritional monitoring, adherence, and outcomes
remains underexplored and should form a core component of the
future research agenda.Conclusion
GLP-1–based pharmacotherapy and bariatric surgery have
irrevocably raised the ceiling of what is achievable in obesity care,
shifting the field from incremental weight reduction to profound,
disease-modifying benefit. However, their true success cannot
be measured in kilograms alone. The same interventions that
normalize glycemia and reduce cardiovascular risk also reshape body
composition and nutrient reserves, with consequences that extend to
strength, resilience, and long-term functional independence.
What emerges from the current evidence is a simple but
transformative insight: rapid weight loss is not merely a metabolic
triumph; it is a nutritionally vulnerable state. Surgical care has
already internalized this reality through mandatory monitoring
and lifelong supplementation; pharmacologic pathways must now
catch up. Treating GLP-1–induced weight loss with the same rigor
accorded to bariatric surgery through structured surveillance,
early supplementation, and protection of lean mass redefines “best
practice” for the next era of obesity medicine.
Nowhere is this shift more urgent than in settings with high
baseline micronutrient deficiency and low lean mass, where unguarded
weight loss risks exchanging cardiometabolic disease for sarcopenia,
frailty, and fracture. The imperative is clear: obesity therapies must be
judged not only by the magnitude of weight they remove, but by the
strength, function, and quality of life they preserve.
A future-ready obesity paradigm will therefore be one in which
every prescription for weight loss is implicitly a prescription for
nutritional stewardship where metabolic success and nutritional
integrity are pursued as inseparable, co-equal goals.
References
Citation
Bellani D, Desai A, Anand S, Maldahiar SP, Mishra S, et al. Hidden Nutritional Risks of Rapid Weight Loss: A Clinical and Nutraceutical Framework for Prevention in Pharmacological and Surgical Obesity Treatments. Indian J Nutri. 2026;13(1): 341.