Research Article
Effect of High-Calorie, High-Protein Supplementation on Clinical Outcomes in Intensive Care Unit (ICU) Patients: A Pilot Study
Bathina H1 and Kelkar N2*
1Department of Clinical Dietetics, Apollo Hospitals, Jubilee Hills, Hyderabad, Telangana, India.
2Department of Clinical Nutrition Excellence Academy, Mumbai, Maharashtra, India.
2Department of Clinical Nutrition Excellence Academy, Mumbai, Maharashtra, India.
*Corresponding author:Dr. Nikhil Kelkar, Department of Nutrition Excellence Academy Mumbai, Maharashtra, India. E-mail Id: nikhilkelkar@hexagonnutrition.com
Article Information:Submission: 08/12/2025; Accepted: 03/01/2026; Published: 06/01/2026
Copyright: © 2026 Bathina H, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: The role of high-caloric, high-protein (HCHP) supplementation in the management of critically ill patients in the Intensive Care Unit (ICU) has garnered significant attention. This study explores the effects of a 100% whey protein supplement on nutritional status, clinical outcomes, and recovery in ICU patients.
Materials and Methods: This prospective pilot study examined 30 ICU patients who received HCHP supplementation. Nutritional status was evaluated using the Modified Subjective Global Assessment (SGA), the Sequential Organ Failure Assessment (SOFA), and other anthropometric measurements.
Biochemical parameters, mortality rates, length of ICU stay, and overall health outcomes were assessed. Safety considerations and potential adverse effects, such as redness, itching, bloating, and diarrhea, were also monitored. Results: Several biochemical parameters demonstrated statistical trends toward improvement; however, only random blood sugar met minimal clinically important difference threshold, while albumin did not. Effect sizes (Cohen’s d) indicated small magnitude for albumin and moderate magnitude for RBS.
Conclusion: The study demonstrates that HCHP supplementation in ICU patients can lead to improved handgrip strength and overall recovery. However, safety concerns such as skin irritation and gastrointestinal issues require further investigation. These findings contribute to the ongoing discussion about optimal nutritional strategies in critical care settings and suggest that HCHP supplementation could be a valuable component of ICU patient care. Findings suggest feasibility and potential benefit, although the absence of a control group and small, heterogeneous sample restrict causal inference. These pilot data support the need for a powered randomized controlled trial.
Materials and Methods: This prospective pilot study examined 30 ICU patients who received HCHP supplementation. Nutritional status was evaluated using the Modified Subjective Global Assessment (SGA), the Sequential Organ Failure Assessment (SOFA), and other anthropometric measurements.
Biochemical parameters, mortality rates, length of ICU stay, and overall health outcomes were assessed. Safety considerations and potential adverse effects, such as redness, itching, bloating, and diarrhea, were also monitored. Results: Several biochemical parameters demonstrated statistical trends toward improvement; however, only random blood sugar met minimal clinically important difference threshold, while albumin did not. Effect sizes (Cohen’s d) indicated small magnitude for albumin and moderate magnitude for RBS.
Conclusion: The study demonstrates that HCHP supplementation in ICU patients can lead to improved handgrip strength and overall recovery. However, safety concerns such as skin irritation and gastrointestinal issues require further investigation. These findings contribute to the ongoing discussion about optimal nutritional strategies in critical care settings and suggest that HCHP supplementation could be a valuable component of ICU patient care. Findings suggest feasibility and potential benefit, although the absence of a control group and small, heterogeneous sample restrict causal inference. These pilot data support the need for a powered randomized controlled trial.
Introduction
Patients in the Intensive Care Unit (ICU) often experience
progressive weight loss, typically associated with muscle atrophy and
diminished functional capacity, which can adversely affect clinical
outcomes and the quality of life in the years following discharge.
This significant muscle loss, driven by the hyper-catabolic and
inflammatory nature of critical illness, can be exacerbated by medical
interventions such as corticosteroid use, prolonged immobilization,
and inconsistent nutritional support.[1] Malnutrition affects
38% to 78% of critically ill ICU patients, with nearly one-third
already malnourished at admission and up to two-thirds becoming
malnourished during hospitalization if proper nutrition is not
provided.[2]
Critical illness is often accompanied by severe proteolysis, leading
to muscle loss rates of up to 1 kilogram per day and contributing to
ICU-acquired weakness. Addressing this challenge requires a higher
protein intake to counteract the effects of anabolic resistance that can
arise due to critical illness.[3]
A goal-directed approach to early enteral nutrition aims to meet 100% of both protein and energy requirements from day one or as soon as possible to improve clinical outcomes. The EAT-ICU trial suggested that optimal protein-energy nutrition could positively affect both short-term and long-term physical function in ICU patients.[1]
Protein intake during critical illness should progressively reach 1.2-1.5 g/kg body weight per day as per the European Society for Clinical Nutrition and Metabolism (ESPEN) guidelines, based on studies showing improved nitrogen balance.[3] Recent studies support the importance of adequate protein intake for reducing ICU related complications and mortality. For instance, Weijs et al. found reduced 28-day mortality among patients who received 1.2-1.5 g/ kg/d protein.[4] Similarly, Allingstrup et al. showed a stepwise dose dependent improvement in survival with higher protein delivery [1], while Compher et al. observed a 6.6% reduction in mortality for each 10% increase in protein intake.[5]
Despite these benefits, high caloric and high protein supplementation in the ICU can pose challenges [6], including overfeeding, gastrointestinal intolerance, and metabolic complications. Thus, a nuanced understanding of patient-specific factors is crucial for determining the appropriateness and effectiveness of nutritional interventions.[7] To avoid over-nutrition, energy/ protein goals should be achieved progressively and not before the first 48 hours. This progression should be guided by local protocols to prevent sharp and rapid increases.[8]
A goal-directed approach to early enteral nutrition aims to meet 100% of both protein and energy requirements from day one or as soon as possible to improve clinical outcomes. The EAT-ICU trial suggested that optimal protein-energy nutrition could positively affect both short-term and long-term physical function in ICU patients.[1]
Protein intake during critical illness should progressively reach 1.2-1.5 g/kg body weight per day as per the European Society for Clinical Nutrition and Metabolism (ESPEN) guidelines, based on studies showing improved nitrogen balance.[3] Recent studies support the importance of adequate protein intake for reducing ICU related complications and mortality. For instance, Weijs et al. found reduced 28-day mortality among patients who received 1.2-1.5 g/ kg/d protein.[4] Similarly, Allingstrup et al. showed a stepwise dose dependent improvement in survival with higher protein delivery [1], while Compher et al. observed a 6.6% reduction in mortality for each 10% increase in protein intake.[5]
Despite these benefits, high caloric and high protein supplementation in the ICU can pose challenges [6], including overfeeding, gastrointestinal intolerance, and metabolic complications. Thus, a nuanced understanding of patient-specific factors is crucial for determining the appropriateness and effectiveness of nutritional interventions.[7] To avoid over-nutrition, energy/ protein goals should be achieved progressively and not before the first 48 hours. This progression should be guided by local protocols to prevent sharp and rapid increases.[8]
This study aims to evaluate the impact of goal-directed early
enteral nutrition on short-term clinical outcomes in ICU patients.
The hypothesis is that early enteral nutrition initiated within the
first 24 hours, with 0.8-1 g/kg body weight (BW) protein and isohypocaloric
feeds (20 kcal/kg BW), and progressing to 1.2-1.5 g/kg BW protein and 20-25 kcal/kg BW,
may improve clinical outcomes in critically ill patients.
Methods
Study Design:
This was a prospective pilot study incorporating retrospective
baseline review, conducted in the Intensive Care Unit (ICU) of
Apollo Hospital, Hyderabad, India. Baseline demographics and
clinical status were extracted from existing records, while all postintervention
variables (nutrition intake, tolerance, questionnaires,
and follow-up) were collected prospectively consistent with STROBE
recommendations and ensured ethical compliance while minimizing
patient burden. No control group was included because the primary
objective of this pilot was feasibility, safety profiling, and preliminary
effect-size estimation in a real-world ICU setting.Study Participants:
Inclusion Criteria1. Age 18 years or older
2. Acute admission to the ICU
3. Informed consent from the patient or written proxy consent from the next of kin
4. Placement of enteral tube for feeding, if applicable
5. No active heart disease, Multiple Organ Dysfunction Syndrome (MODS), or sepsis
6. Not on dialysis, and no active renal failure or replacement therapy
Exclusion Criteria:
1. Age under 18 years2. Contraindications to enteral or parenteral nutrition
3. Active heart disease, MODS, sepsis, liver failure, traumatic brain injury, burns, or diabetic ketoacidosis
4. Patients on dialysis or with active renal failure/replacement therapy
5. Pregnancy
6. Clinician’s discretion or imminent death
Sample Size:
A total of 30 subjects were included in the pilot study.Intervention and Blinding:
The nutritional intervention strategy was not masked for
research or clinical staff. The study used a high-protein, high-calorie
supplement (PentaSure 2.0) as part of the enteral feeding protocol,
in addition to the oral diet. The goal-directed early enteral nutrition
aimed to meet 100% of the dietary requirements for both protein
and energy within 48 hours, according to the European Society for
Clinical Nutrition and Metabolism (ESPEN) guidelines [3]. The
following points summarize the enteral nutrition approach:1. Initiate protein intake at 0.8–1 g/kg body weight from day 1.
2. Progress to 1.2–1.5 g/kg body weight based on clinical judgment.
3. Caloric goals were set at 20–25 kcal/kg body weight, or as determined by resting energy expenditure (REE = VCO2 x 8.19).
Study Period:
The study was conducted during the patients’ ICU stay, with a
follow-up of up to 90 days.Data Collection:
Data was collected from 30 ICU patients receiving high-calorie,
high-protein feeds through enteral feeding. Nutritional status was
assessed using the Modified Subjective Global Assessment (SGA)
[9]. The Sequential Organ Failure Assessment (SOFA) evaluates
the severity of organ dysfunction in ICU patients [10] and the
Acute Physiology and Chronic Health Evaluation (APACHE) [11],
another severity assessment metric, scores were used to assess illness
severity. Anthropometric measurements, including height, weight,
body mass index (BMI), mid-upper arm circumference (MUAC),
calf circumference, and hand grip strength, were also collected. A
questionnaire was developed to track patients’ calorie and protein
intake, biochemical parameters before and after supplementation,
and the effect of feeding on medical conditions. The length of stay,
nosocomial infections, allergic reactions, and gastrointestinal
complications were documented over an average duration of 14 days
using a questionnaireStatistical Analysis:
Statistical analyses were performed using Microsoft Excel and
SPSS software. Descriptive statistics, such as mean and standard
deviation, were calculated. T-test was used to analyze the level of
significance, and the coefficient of correlation was applied to assess
relationships among variables.Results
The study involved a sample of 30 ICU patients with multiple
serious acute morbidities necessitating intensive care. The mean age
of the patients was 56 ± 16.3 years, with approximately 50% falling
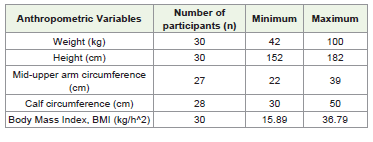
within the 51-70 years age range [Table 1].
According to the Modified SGA scores, 46.7% of the patients were categorized as well-nourished, 40% as moderately malnourished, and 13.3% as severely malnourished.
The anthropometric measurements of the participants are presented in [Table 1]. The mean body mass index was 24.58 ± 5.09, mid-upper arm circumference (MUAC) was 29.6 ± 4.8 cm, and calf circumference was 37.5 ± 5.4 cm. Around 53% (n=16) of the patients had normal BMI, 10% (n=3) were underweight, and 36.66% (n=11) were overweight. MUAC measurements indicated that 73% of patients had values within the normal range, 10% slightly above, and 7% below. Similarly, 69% of patients had normal calf circumference, with the remaining showing values either above or below the normal range.
The SOFA scores showed that 24% had a score below 5, while the remaining 76% had scores above 5. The APACHE scores revealed that 32% of patients scored below 10, while 68% scored above 10, indicating an estimated survival rate of 82%. Among the study subjects, the observed mortality rate was 12%. Additionally, the assessment of their overall condition revealed that 16.7% of them were in good condition, 26.7% were categorized in bad condition, and the majority, 56.7%, were in a poor state
According to the Modified SGA scores, 46.7% of the patients were categorized as well-nourished, 40% as moderately malnourished, and 13.3% as severely malnourished.
The anthropometric measurements of the participants are presented in [Table 1]. The mean body mass index was 24.58 ± 5.09, mid-upper arm circumference (MUAC) was 29.6 ± 4.8 cm, and calf circumference was 37.5 ± 5.4 cm. Around 53% (n=16) of the patients had normal BMI, 10% (n=3) were underweight, and 36.66% (n=11) were overweight. MUAC measurements indicated that 73% of patients had values within the normal range, 10% slightly above, and 7% below. Similarly, 69% of patients had normal calf circumference, with the remaining showing values either above or below the normal range.
The SOFA scores showed that 24% had a score below 5, while the remaining 76% had scores above 5. The APACHE scores revealed that 32% of patients scored below 10, while 68% scored above 10, indicating an estimated survival rate of 82%. Among the study subjects, the observed mortality rate was 12%. Additionally, the assessment of their overall condition revealed that 16.7% of them were in good condition, 26.7% were categorized in bad condition, and the majority, 56.7%, were in a poor state
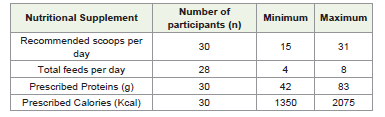
[Table 2] presents the details of the nutritional supplement.
In terms of enteral nutrition, the average number of scoops
recommended for any patient was 24.5 ± 4.1, distributed over 6-7
feeds per day, providing 66.85 ±8.6 gm of protein and 1677 ± 191
kcals to meet the nutritional requirements of the critically ill patients.
The number of scoops and feeds varied as per the patient’s body
weight and health conditions.
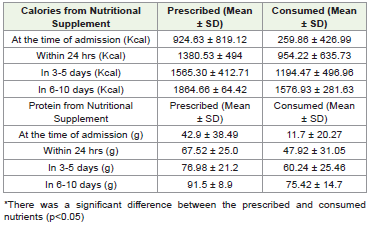
The findings indicate that, initially, the consumption of the
prescribed supplement in ICU patients accounted for only 28% of
the recommended caloric intake and 27% of the prescribed protein
intake. However, subsequent observations showed a significant
improvement, with patients eventually consuming 86% of the
recommended calories and 82% of the prescribed protein (p <0.05)
[Table 3] .
The primary outcome of this study was to evaluate the effect of high-protein supplementation on functional capacity, measured through hand grip strength, in relation to nutritional status as assessed by the Subjective Global Assessment (SGA) score.
At the time of admission, nearly one-third (33.33%) of the patients exhibited normal grip strength in their dominant hand, while the remaining displayed reduced grip strength. The mean handgrip for the dominant hand was 10.32 ± 5.4, and for the nondominant hand was 12.10 ± 3.5. Further, correlation analysis revealed a positive association between the total number of feeds (nutritional supplement) and the dominant hand grip strength (p<0.01).
Regarding the secondary outcomes, the number of days alive without the use of mechanical ventilation ranged from 6 to 12 days, with 34% of patients not requiring a ventilator for 6 days and 64% able to stay off ventilatory support for 12 days. Notably, no new onset of organ failure was reported during the study period. However,
The primary outcome of this study was to evaluate the effect of high-protein supplementation on functional capacity, measured through hand grip strength, in relation to nutritional status as assessed by the Subjective Global Assessment (SGA) score.
At the time of admission, nearly one-third (33.33%) of the patients exhibited normal grip strength in their dominant hand, while the remaining displayed reduced grip strength. The mean handgrip for the dominant hand was 10.32 ± 5.4, and for the nondominant hand was 12.10 ± 3.5. Further, correlation analysis revealed a positive association between the total number of feeds (nutritional supplement) and the dominant hand grip strength (p<0.01).
Regarding the secondary outcomes, the number of days alive without the use of mechanical ventilation ranged from 6 to 12 days, with 34% of patients not requiring a ventilator for 6 days and 64% able to stay off ventilatory support for 12 days. Notably, no new onset of organ failure was reported during the study period. However,
Table 3: Comparison of consumed and prescribed calories and protein from
nutritional supplements at various time points
56.7% of the study population experienced nosocomial infections,
including urinary tract infections, pneumonia, bacterial infections,
and hyperkalemia. Additionally, none of the patients required renal
replacement therapy.
In the study, 16.7% of the patients experienced serious adverse reactions, including redness and itching of the skin, wound infections, bacterial infections, and altered lipid profiles. Gastrointestinal (GI) complications were observed in 56.7% of the patients, with symptoms such as bloating, constipation, diarrhea, and vomiting. Notably, the supplement feed did not exacerbate these complications.
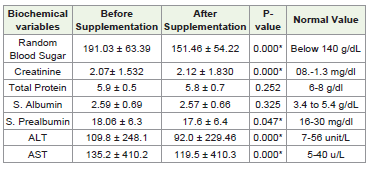
[Table 4] presents the biochemical parameters before and after the implementation of a high-protein supplement in ICU patients. With the exception of total protein and serum albumin, all other biochemical parameters showed statistically significant improvement after supplementation (p<0.05), indicating a favorable impact of the nutritional intervention. Pre-investigation values for most parameters were on the higher side before the supplementation began.
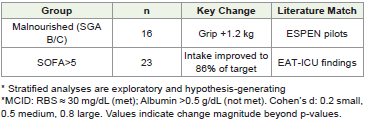
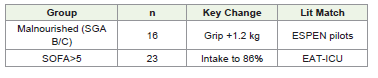
[Table 5] presents results correlated with nutritional status and the severity of illness. In a cohort of malnourished patients (SGA B/C; n=16), there was an observed increase in handgrip strength by 1.2 kg, suggesting an enhancement in functional recovery, which aligns with the findings from the ESPEN pilot study. In individuals exhibiting elevated morbidity (SOFA >5; n=23), nutritional intake reached 86% of the established target, consistent with evidence from EATICU that advocates for targeted nutritional strategies in critically ill populations.
In the study, 16.7% of the patients experienced serious adverse reactions, including redness and itching of the skin, wound infections, bacterial infections, and altered lipid profiles. Gastrointestinal (GI) complications were observed in 56.7% of the patients, with symptoms such as bloating, constipation, diarrhea, and vomiting. Notably, the supplement feed did not exacerbate these complications.
[Table 4] presents the biochemical parameters before and after the implementation of a high-protein supplement in ICU patients. With the exception of total protein and serum albumin, all other biochemical parameters showed statistically significant improvement after supplementation (p<0.05), indicating a favorable impact of the nutritional intervention. Pre-investigation values for most parameters were on the higher side before the supplementation began.
[Table 5] presents results correlated with nutritional status and the severity of illness. In a cohort of malnourished patients (SGA B/C; n=16), there was an observed increase in handgrip strength by 1.2 kg, suggesting an enhancement in functional recovery, which aligns with the findings from the ESPEN pilot study. In individuals exhibiting elevated morbidity (SOFA >5; n=23), nutritional intake reached 86% of the established target, consistent with evidence from EATICU that advocates for targeted nutritional strategies in critically ill populations.
Postprandial blood glucose levels remained stable, with no
significant spikes observed in any patient following supplementation.
Mean systolic blood pressure was recorded at 130.6 ± 11.4 mmHg, and
mean diastolic blood pressure was 74.6 ± 7.3 mmHg, both within the
normal range for ICU patients. The average urine output was 1179.16
± 174.02 mL, suggesting adequate renal function. The random blood
sugar level was 151.46 ± 54.22 mg/dL, which is consistent with the
expected outcomes for the given supplement’s chemical composition,
characterized by low glycemic index carbohydrates, high protein, and
high caloric content.
[Table 5] presents results correlated with nutritional status and the severity of illness. In a cohort of malnourished patients (SGA B/C; n=16), there was an observed increase in handgrip strength by 1.2 kg, suggesting an enhancement in functional recovery, which aligns with the findings from the ESPEN pilot study. In individuals exhibiting elevated morbidity (SOFA >5; n=23), nutritional intake reached 86% of the established target, consistent with evidence from EATICU that advocates for targeted nutritional strategies in critically ill populations.
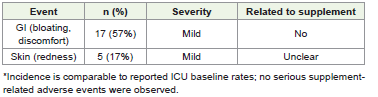
[Table 6] presents a summary of the adverse events recorded throughout the study. Seventeen participants (57%) reported gastrointestinal complaints, including bloating, all of which were classified as mild and deemed unrelated to the intervention. Five participants (17%) exhibited mild skin redness; the relationship to the intervention remains ambiguous. No significant adverse events were documented.
[Table 5] presents results correlated with nutritional status and the severity of illness. In a cohort of malnourished patients (SGA B/C; n=16), there was an observed increase in handgrip strength by 1.2 kg, suggesting an enhancement in functional recovery, which aligns with the findings from the ESPEN pilot study. In individuals exhibiting elevated morbidity (SOFA >5; n=23), nutritional intake reached 86% of the established target, consistent with evidence from EATICU that advocates for targeted nutritional strategies in critically ill populations.
[Table 6] presents a summary of the adverse events recorded throughout the study. Seventeen participants (57%) reported gastrointestinal complaints, including bloating, all of which were classified as mild and deemed unrelated to the intervention. Five participants (17%) exhibited mild skin redness; the relationship to the intervention remains ambiguous. No significant adverse events were documented.
Discussion
Although termed retrospective in initially, was conducted
prospectively with retrospective baseline review. The single-arm
design limits attribution of outcomes solely to supplementation;
however, pilot trials appropriately emphasize feasibility and effect size
estimation. Mixed ICU case mix and small sample size further
support cautious interpretation and need for a randomized controlled
trail.
The significant improvement in the calorie and protein intake of patients to 86% and 82% of the prescribed intake, respectively, in comparison to the initial intake of only 28% and 27% of calories and protein prescribed, suggests an increasing adherence to nutritional support over time, underlining the potential for improved nutritional management in critical care settings. This trend underscores the importance of closely monitoring and adjusting nutritional support to meet patients’ evolving needs in the ICU.
Furthermore, the findings indicate a meaningful improvement in hand grip strength with an increased number of recommended feeds. Thereby, suggesting that nutritional interventions can positively impact muscle strength and functional capacity in critically
The significant improvement in the calorie and protein intake of patients to 86% and 82% of the prescribed intake, respectively, in comparison to the initial intake of only 28% and 27% of calories and protein prescribed, suggests an increasing adherence to nutritional support over time, underlining the potential for improved nutritional management in critical care settings. This trend underscores the importance of closely monitoring and adjusting nutritional support to meet patients’ evolving needs in the ICU.
Furthermore, the findings indicate a meaningful improvement in hand grip strength with an increased number of recommended feeds. Thereby, suggesting that nutritional interventions can positively impact muscle strength and functional capacity in critically
ill patients, and highlight the potential benefits of consistent highprotein
supplementation in the ICU setting. The observed correlation
between the number of feeds and hand grip strength reinforces
the need for ongoing nutritional support as part of comprehensive
critical care. Matheson et al. reported similar findings of improved
hand grip strength with specialized ONS. [13]
The nutritional status at the time of discharge was good for 86.7 % of the patients as per the standard classification of the SGA scores. The mean value of the SOFA score, which is indicative of determining overall mortality, was found to be 5 ±1.7, which suggests a positive prognosis for the patients.[14] Similarly, the APACHE score, which is conventionally used to determine the mortality rate among patients admitted to the ICU, was found to be 12 ± 4, which corresponds to 85% of survival. [15,16]
While serious adverse reactions and GI complications are common among ICU patients [17], the nutritional supplement regimen did not seem to worsen these issues in the current study. Careful monitoring and individualized adjustments in feeding protocols may mitigate potential adverse effects.
The high-protein supplementation had a generally positive impact on key biochemical parameters without causing detrimental effects on blood pressure, glucose levels, or renal function. This indicates that the nutritional intervention was well-tolerated and did not lead to adverse metabolic responses among the critically ill patients in the ICU. Further research might explore the mechanisms behind these effects and the potential for similar supplementation strategies in broader ICU populations.
Additionally, the results suggest variability in the duration of mechanical ventilation needs among ICU patients, indicating that a significant proportion could maintain respiratory function without mechanical support for extended periods. The relatively high rate of nosocomial infections underscores the importance of infection control measures in critical care settings. Despite these complications, the absence of organ failure onset and the lack of renal replacement therapy requirements indicate that the overall severity of illness remained manageable. Further analysis of these outcomes could guide infection prevention strategies and ventilatory support protocols in the ICU. The adverse effects could have been influenced by various factors during the course of treatment, not solely by the nutritional supplement.
The nutritional status at the time of discharge was good for 86.7 % of the patients as per the standard classification of the SGA scores. The mean value of the SOFA score, which is indicative of determining overall mortality, was found to be 5 ±1.7, which suggests a positive prognosis for the patients.[14] Similarly, the APACHE score, which is conventionally used to determine the mortality rate among patients admitted to the ICU, was found to be 12 ± 4, which corresponds to 85% of survival. [15,16]
While serious adverse reactions and GI complications are common among ICU patients [17], the nutritional supplement regimen did not seem to worsen these issues in the current study. Careful monitoring and individualized adjustments in feeding protocols may mitigate potential adverse effects.
The high-protein supplementation had a generally positive impact on key biochemical parameters without causing detrimental effects on blood pressure, glucose levels, or renal function. This indicates that the nutritional intervention was well-tolerated and did not lead to adverse metabolic responses among the critically ill patients in the ICU. Further research might explore the mechanisms behind these effects and the potential for similar supplementation strategies in broader ICU populations.
Additionally, the results suggest variability in the duration of mechanical ventilation needs among ICU patients, indicating that a significant proportion could maintain respiratory function without mechanical support for extended periods. The relatively high rate of nosocomial infections underscores the importance of infection control measures in critical care settings. Despite these complications, the absence of organ failure onset and the lack of renal replacement therapy requirements indicate that the overall severity of illness remained manageable. Further analysis of these outcomes could guide infection prevention strategies and ventilatory support protocols in the ICU. The adverse effects could have been influenced by various factors during the course of treatment, not solely by the nutritional supplement.
Conclusion
The high caloric and high protein supplementation in ICU
patients demonstrated a capacity to improve hand grip strength,
stabilize blood glucose levels, and positively affect other biochemical
parameters, suggesting favorable outcomes in terms of patient
tolerance among critically ill patients.
Tailored nutritional support can lead to improved outcomes such as lower mortality rates and reduced ICU stay durations. However, careful consideration of individual patient needs is essential to mitigate risks like overfeeding and metabolic disturbances.
Tailored nutritional support can lead to improved outcomes such as lower mortality rates and reduced ICU stay durations. However, careful consideration of individual patient needs is essential to mitigate risks like overfeeding and metabolic disturbances.
Clinical Significance:
High caloric and high protein supplementation in ICU patients
can lead to improved outcomes, such as increased hand grip strength,
stable blood glucose levels, and overall better biochemical responses.
These benefits may translate to reduced mortality rates and shorter
ICU stays. The study also highlights the need for personalized
nutritional approaches to avoid complications like overfeeding and
metabolic disturbances.
This study adds to the evolving understanding of critical
care nutrition and underscores the importance of personalized
approaches. There is an ongoing need for research to optimize
nutritional protocols, the key to shaping the future of critical care
nutrition, ultimately enhancing patient outcomes and quality of care
in ICU settings.Funding Statement:
No funding was received. Oral nutritional supplements were
procured independently through the hospital pharmacy as part of
routine care. The manufacturer had no role in study design, data
collection, analysis, interpretation, manuscript writing, or publication
decision.Conflict of Interest:
The authors declare no financial or personal conflicts. Dr. Nikhil
Kelkar’s affiliation was fully disclosed; he had no role in patient care,
recruitment, data collection, analysis, or interpretation.Authors Contribution:
The corresponding author provided manuscript oversight and
handled journal correspondence. Clinical care and data collection
were performed at Apollo Hospital by Dt. Haritha Bathina and the
clinical teamAcknowledgements:
The authors acknowledge the Clinical Nutrition Team of Apollo
Hospitals, Jubilee Hills, Hyderabad for meticulous monitoring and
documentation.Bathina H: 0009-0000-7890-9423
Dr. Nikhil Kelkar: 0000-0001-6607-5580
References
Citation
Bathina H, Kelkar N. Effect of High-Calorie, High-Protein Supplementation on Clinical Outcomes in Intensive Care Unit (ICU) Patients: A Pilot Study. Indian J Nutri. 2026;13(1): 334.