Research Article
Clinical Aspects of Iodine and Iodine Deficiency
Barseghyan NA1,2 and Davtyan HH1*
1Yerevan State Medical Universit after Mkhitar Heratsi, Armenia
2Muratsan Hospital Complex, Armenia
2Muratsan Hospital Complex, Armenia
*Corresponding author: HH Davtyan, Yerevan State Medical Universit after Mkhitar Heratsi, Armenia, E-mail Id: hasmikdavtyan179@gmail.com
Article Information:Submission: 31/10/2025; Accepted: 26/11/2025; Published: 29/11/2025
Copyright: © 2025 Barseghyan NA, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Iodine; Iodine Metabolism; Iodine Deficiency Disorders; Free Thyroxine; Iodized Salt
Introduction
Iodine, characterized as a trace element, is essential for the
synthesis of thyroid hormones thyroxine (T4) and triiodothyronine
(T3). These hormones are critical for the functioning of the liver,
kidney, muscle, brain, and central nervous system [5]. Iodine plays
a vital role in regulating overall metabolism and is crucial for fetal
and child neurodevelopment, as well as organ and tissue function
[1]. An average adult typically contains 15-20 mg of iodine in their
body, with 70-80% stored in the thyroid gland [7]. Deficiency during
pregnancy is a leading cause of preventable intellectual disability in
children, underscoring the importance of monitoring iodine status
in pregnant women and women of reproductive age globally [17].
Iodine, the heaviest stable halogen element, primarily exists in nature
as iodide (I-), which is commonly used to produce supplements
and iodized table salt in the form of potassium iodide (KI). Another
naturally occurring form is iodate (IO3-), used to fortify table salt
as potassium iodate (KIO3) [13]. Iodide is naturally present in soil
and seawater, influencing the iodine content of crops. However,
many regions have iodide-depleted surface soils. Since iodide is
found in seawater, it can volatilize into the atmosphere and return
to the soil. In non-coastal regions, this cycle is incomplete, leading
to iodide depletion in plant foods and drinking water. Historically,
iodine deficiency was observed in populations from inland regions
(central Asia and Africa, central and eastern Europe, the central U.S.),
mountainous areas (Alps, Andes, Atlas, Himalayas), and those with
frequent flooding (Southeast Asia) [19]. Early research conducted in
Armenia identified the country as iodine deficient, as evidenced by
the presence of endemic goiter, particularly notable in the southern
regions. Despite efforts in the early 1990s by the major domestic
salt producer to voluntarily iodize food salt, deficiency remained
widespread [11].
Iodine Metabolism:
Iodine is ingested as an inorganic ion or organically bound
compound, but it is absorbed in the form of iodide after the reduction
of iodine compounds in the stomach. Enteric absorption of iodide
occurs primarily in the stomach and duodenum, where the enteric
isoform of the sodium iodide symporter (NIS) is predominantly
expressed. [20].Iodine accumulates predominantly in the thyroid
gland, which contains the largest pool of intracellular iodine in the
human body. However, the most significant amount of iodide is found
in the extracellular fluid, where its concentration is approximately
10–15 μg/L. Circulating iodide is cleared by the kidneys, with a small
portion lost through the skin, intestinal secretions, or expired air
[10].The mammary gland can also accumulate and secrete iodide,
providing an additional source of iodine clearance in lactating
women. Renal clearance of iodide is estimated at 30-50 mL per minute
but largely depends on the individual glomerular filtration rate, with
no evidence of tubular secretion or active transport. Reabsorption
is partial and passive, and renal clearance of iodide is influenced by
overall iodide status. Clearance by the kidney is constant, while uptake
by the thyroid depends on iodine status and intake [19].When iodine
supply is sufficient, uptake by the thyroid may be less than 10%, but it
can exceed 80% due to chronic deficiency. After active transport into
the thyroid, iodide is stored in the thyroglobulin (Tg) protein before
being converted into T3 and T4. These hormones enter circulation
bound to carrier proteins and target tissues, with T3 being the primary
physiologically active form that preferentially binds to its receptors.
Both free T3 and T4 are assessed in serum by immunoassay. Iodide
is also produced via intrathyroidal deiodination of iodothyronine
after thyroglobulin hydrolysis. Part of the circulating iodide pool is
re-organified into de novo synthesized iodothyronine, while the rest
enters systemic circulation (iodide leakage). Iodine also originates
from the peripheral degradation of thyroid hormones, entering
circulation, where it can be recycled after subsequent thyroid uptake
or ultimately excreted in urine [20].Recommended Intake:
The recommended daily iodine intake ranges from less than ten
micrograms in areas with extreme iodine deficiency to several hundred
milligrams in patients taking iodine-containing medications. For
adults and the elderly, 150 μg of iodine is generally recommended.
Pregnant or lactating women require at least 200-250 μg daily. In
newborns and children, the iodine requirement per kilogram of body
weight is higher than in adults, corresponding to an absolute iodine
intake requirement of 70-120 μg in children and 40 μg in newborns.
These recommendations are based on factors such as daily thyroid
hormone turnover in healthy individuals, the mean iodine intake
associated with the lowest values of thyroid-stimulating hormone in
the normal range, the smallest thyroid volume, the lowest incidence
of transient hypothyroidism in neonatal screening, and the mean
requirement of levothyroxine to restore euthyroidism in patients
with thyroid agenesis or following thyroidectomy [18].Natural sources of iodine:
Iodine is primarily obtained from food sources, especially
vegetables grown in iodine-rich soil, with the remaining requirement
typically met through drinking water. The oceans serve as the primary
reservoirs of iodide, which, upon conversion to elemental iodine,
follows the water cycle and is delivered to the soil through rainfall.
However, in regions with high precipitation rates, such as mountainous
inland areas, the iodine content of soils and consequently, of food
crops is significantly reduced due to rain leaching. This leads to
iodine-deficient soils and, subsequently, nutritional iodine deficiency,
which is more prevalent in these mountainous inland areas but can
also be found in very rainy coastal zones [3]. Seafood and saltwater
fish are the most important sources of iodine, as marine fauna and
flora accumulate substantial amounts of soluble iodine from seawater.
Freshwater and farmed fish, in comparison to seawater organisms,
contain lower levels of iodine [14]. Therefore, fish sourced from rivers
or lakes typically have lower iodine content [8].Iodine deficiency disorders:
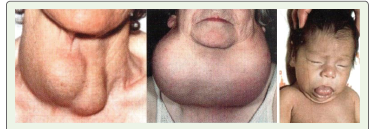
The critical importance of iodine throughout the life cycle is
evident from a group of diseases that arise due to iodine deficiency
(iodine deficiency disorders, IDD) and the subsequent insufficient
production of thyroid hormones. The effects of iodine deficiency
vary depending on the life stage and the severity of the deficiency
[9]. For instance, during pregnancy, severe iodine deficiency
leads to significant mental and growth impairments, including
deaf-mutism and spasticity, which are characteristic features of
cretinism. Conversely, mild-to-moderate iodine deficiency in the
womb is linked to neurodevelopmental issues such as decreased
intelligence quotient, impaired fine motor skills, and difficulties
in verbal and non-verbal communication. Iodine deficiency in
the early stages of life may significantly affect brain development.
Thyroid hormones are necessary for the myelination of the central
nervous system, which takes place before and shortly after birth.
Primary hypothyroidism related to iodine deficiency has been found
to negatively affect cognitive function with potentially irreversible
intellective consequences [19]. Adequate maternal exposure to iodine
in the early stages of pregnancy is essential for the proper intellective
development of the child, irrespective of hypothyroidism.Iodine deficiency has also been associated with increased
miscarriage and stillbirth rates, and congenital disabilities, including
congenital hypothyroidism in the offspring [6]. Congenital
hypothyroidism comprises two classical clinical features with
specific phenotypes: neurological and myxedematous [15].The first is
characterized by intellectual impairment and developmental delays,
and various neurological defects, including the underdevelopment
of the cochlea leading to deafness, defects of the cerebral neocortex
with intellective impairment, and the underdevelopment of the
corpus striatum with motor disorders. Patients do not exhibit signs of
hypothyroidism, and the prevalence of goiter is similar to that observed
in the general population. The hypothyroid phenotype includes
dwarfism with delayed bone and sexual maturation, intellective
impairment, and overt hypothyroidism. Thyroid development is
critically involved, and patients usually exhibit low thyroid volume
or thyroid atrophy. The prevalence and risk factors associated with.
Neurological cretinism is related to thyroid hormone deficiency in
the early stages of embryonal development, resulting from a severe
maternal iodine deficiency in a phase when thyroid development is
still incomplete. Myxedematous cretinism is associated with thyroid
insufficiency during late pregnancy or early infancy. Iodine deficiency
remains a significant public health concern, affecting more than one
billion people worldwide and leading to various levels of growth and
developmental abnormalities [2].
Iodine Supplementation:
Thanks to food policies permitting the addition of iodine to food
items, processed foods containing notably elevated levels of iodine
have become more accessible in recent decades. These foods have
been utilized in nationally implemented programs to offer iodine
prophylaxis, mitigating the clinical effects of iodine deficiency. The
global strategy recommended for this purpose is the iodization of
salt for human consumption. Iodine supplementation is an effective
strategy for reducing population iodine deficiency, but care must
be taken to avoid excessive intake. The goal is to increase iodine
intake to the level necessary to prevent IDDs without surpassing
it. The bioavailability of iodine from food varies and is challenging
to determine, and interactions between different foods in the food
matrix are not well understood [14]. The most practical and costeffective
method of providing iodine supplementation to deficient
populations is through the use of iodized salt, as recommended by
organizations such as the World Health Organization (WHO), the
United Nations Children’s Fund (UNICEF), and the International
Council for the Control of Iodine Deficiency Disorders. Other
approaches include the consumption of iodized water, iodized oil,
and iodine tablets [18]. The quantity and type of iodine used for salt
fortification vary by region but typically fall within the range of 20-40
mg iodine/kg salt. The forms used for fortification globally are either
potassium iodate (KIO3), which is more stable, or potassium iodide
(KI), which has a higher iodine content and solubility [7], or sodium
iodide (NaI). Unlike iodization of salt and water, which can reach a
larger proportion of the population, supplementation with iodized oil
or tablets is more suitable for individual use and can rapidly improve
iodine status, especially in regions where salt iodization is not feasible
or cannot be implemented in the short term.Evaluation of Iodine Levels:
Multiple indicators are utilized to assess iodine status, including
urinary iodine concentration (UIC), thyroid volume (TV), serum
thyroid-stimulating hormone (TSH), thyroid hormones, and serum
Tg. Median UIC in spot urine samples is the preferred indicator to
assess iodine status in populations. UIC serves as a reliable marker
of short-term iodine status. Although UIC at the individual level
fluctuates with recent food intake and hydration status, the median
UIC is a valid marker of iodine intake at the group level [21].UIC is
not suitable for determining the proportion of the population with
iodine deficiency or excess. Having two independent spot samples
from a subset of the study population can be used to estimate the
habitual long-term iodine intake and the prevalence of deficiency
and excess [22]. For school-aged children and non-pregnant adults,
iodine intake is considered sufficient when the median UIC in the
population falls within the range of 100–299μg/L. In pregnant women,
iodine intake is considered sufficient when the median UIC ranges
from 150 - 249μg/L. Estimating daily iodine intake for population
estimates can be done by extrapolating from UIC, using estimates of
mean 24-hour urine volume with the equation: UIC (μg/L) × 0.0235
× body weight (kg) = iodine intake (μg/day), assuming 90% excretion
and 1.5 liters urine per 24 hours. Therefore, a median UIC of 100μg/L
in an adult corresponds roughly to an average daily intake of 150μg.
However, this approach does not consider iodine uptake in the
thyroid and is less reliable in iodine-deficient situations and during
pregnancy and lactation [18].TSH is a direct marker of thyroid function and can be considered
reflective of iodine status to some extent. It is utilized as part of
newborn screening in many developed countries to detect cases of
congenital hypothyroidism. Neonates have higher iodine turnover in
the thyroid compared to children and adults, and neonatal TSH is
expected to be a sensitive indicator of population iodine deficiency.
However, upon closer examination, it has been concluded that the
increase in TSH observed is not significant enough for it to be a
useful marker [4]. Furthermore, in children and adults, TSH levels
may elevate slightly in cases of iodine deficiency, but they typically
remain within the reference range, making it a relatively insensitive
marker [16].
The primary biologically active thyroid hormones are T4 and
T3, serving as direct indicators of thyroid function. However, they
are ineffective as biomarkers for iodine status at both population
and individual levels. Thyroid hormone levels often fluctuate within
the reference range in response to iodine status, making them
insufficiently sensitive to reliably reflect iodine status except in
severely iodine-deficient regions.
Tg is an iodoglyco protein produced in the follicular cells of
the thyroid gland in response to TSH. It serves as the precursor to
thyroid hormones. Its primary clinical use is as a tumor marker in
patients who have undergone total thyroidectomy for differentiated
thyroid cancer. Tg concentration generally reflects the overall mass
of thyroid cells. Therefore, it has been proposed that it could be used
as an indicator of iodine status in populations living in areas with
endemic goiter. Indeed, studies have shown that tg levels decrease at
a population level following the implementation of a salt iodization
program [12].
Material and Methods
We carried out a study involving pediatric patients (ages 0–17
years) from 2023 to May 2024. All patients were evaluated at
the Division of Pediatric Endocrinology at “Muratsan” Hospital
Complex. Blood samples were collected in the morning. The thyroid
hormone levels are expressed in the following units: TSH in μU/mL,
T4 in ng/dL. The normal values (nv) for T4 are 0.8–1.8 ng/dL. The
immunological tests were conducted using clinical routine analysis
instruments at “Muratsan” Hospital Complex, employing accredited
techniques. The immunological analyses were performed using the
Cobas analyzer.
Results and Discussion
T4 circulates in the bloodstream as an equilibrium mixture
of free and serum bound hormone. Free T4 is the unbound and
biologically active form, which represents only 0.03 % of the total T4.
The remaining T4 is inactive and bound to serum proteins such as
T4 binding globulin (75%), pre-albumin (15%), and albumin (10%).
Therefore, free T4 is a useful tool in clinical routine diagnostics for
the assessment of the thyroid status. It should be measured together
with TSH if thyroid disorders are suspected and is also suitable for
monitoring thyrosuppressive therapy [22].
The Diagnostic Laboratory of the “Muratsan” Hospital Complex at the Yerevan State Medical University named after M. Heratsi conducts modern complex studies for children and adolescents with acute and subacute diseases, both inpatient and outpatient. The investigations of the thyroid hormone T4 are of key importance in Armenia. Laboratory diagnostic tests for T4 allow for the early detection of thyroid pathologies in children and adolescents. In 2023 alone, the laboratory conducted 1,114 free T4 tests among children, and during the period from January to May 2024, more than 1,025 children were tested. The number of patients is trending upward, which is due to several factors. Free T4 screening is among the strategically important issues for Armenia. Diagnostic laboratory measures among socially vulnerable patients allow for the regulation of tissue growth in children through early endocrinological interventions, normalizing the metabolism of proteins, carbohydrates, and lipids.
The Diagnostic Laboratory of the “Muratsan” Hospital Complex at the Yerevan State Medical University named after M. Heratsi conducts modern complex studies for children and adolescents with acute and subacute diseases, both inpatient and outpatient. The investigations of the thyroid hormone T4 are of key importance in Armenia. Laboratory diagnostic tests for T4 allow for the early detection of thyroid pathologies in children and adolescents. In 2023 alone, the laboratory conducted 1,114 free T4 tests among children, and during the period from January to May 2024, more than 1,025 children were tested. The number of patients is trending upward, which is due to several factors. Free T4 screening is among the strategically important issues for Armenia. Diagnostic laboratory measures among socially vulnerable patients allow for the regulation of tissue growth in children through early endocrinological interventions, normalizing the metabolism of proteins, carbohydrates, and lipids.
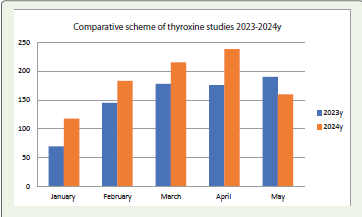
The diagram schematically presents the effectiveness of the studies,
which has almost doubled over the course of 1.5 years. Laboratory
diagnostic tests allow for the early detection of hyperthyroidism or
hypothyroidism in children, enabling targeted treatment for children
and adolescents [Figure 1].
The diagram clearly shows a doubling in the number of patient visits in January. The growth trend continues in the following months as well [Figure 2].
Thyroid function disorders in children lead to either hyperthyroidism or hypothyroidism. According to the analysis of screening test results, in cases of hyperthyroidism, children show an increase in T4 concentration in the blood, a sharp decrease in calcium (Ca) levels (while the amount of calcitonin remains normal), presence of hyperglycemia and glycosuria, and low cholesterol concentration in the blood serum.
Primary hypothyroidism is caused by a thyroid gland dysfunction, while secondary hypothyroidism is due to a pituitary gland functional disorder. Primary hypothyroidism is diagnosed in newborns and is a result of fetal intrauterine developmental disturbances. The widespread occurrence of hypothyroidism in neonatal-age children is dangerous because it has no clinical manifestations in the first days of life. Hypothyroidism in neonatal-age children can only be detected through laboratory diagnostic tests. Due to this, screening tests for the detection of congenital hypothyroidism are conducted in several leading countries and now also in Armenia. T4 screening
The diagram clearly shows a doubling in the number of patient visits in January. The growth trend continues in the following months as well [Figure 2].
Thyroid function disorders in children lead to either hyperthyroidism or hypothyroidism. According to the analysis of screening test results, in cases of hyperthyroidism, children show an increase in T4 concentration in the blood, a sharp decrease in calcium (Ca) levels (while the amount of calcitonin remains normal), presence of hyperglycemia and glycosuria, and low cholesterol concentration in the blood serum.
Primary hypothyroidism is caused by a thyroid gland dysfunction, while secondary hypothyroidism is due to a pituitary gland functional disorder. Primary hypothyroidism is diagnosed in newborns and is a result of fetal intrauterine developmental disturbances. The widespread occurrence of hypothyroidism in neonatal-age children is dangerous because it has no clinical manifestations in the first days of life. Hypothyroidism in neonatal-age children can only be detected through laboratory diagnostic tests. Due to this, screening tests for the detection of congenital hypothyroidism are conducted in several leading countries and now also in Armenia. T4 screening
reveals the presence of the disease in the preclinical stage, allowing
for therapeutic interventions. Hypothyroidism is accompanied by
high concentrations of cholesterol, triglycerides, and T4 in the blood
serum.
Ensuring adequate iodine intake is essential for maintaining optimal health, particularly in areas where iodine deficiency is common. Therefore, supplementation programs, such as the use of iodized salt or iodine supplements, are effective strategies for preventing and addressing iodine deficiency, leading to better health outcomes and a reduced risk of related health issues.
Ensuring adequate iodine intake is essential for maintaining optimal health, particularly in areas where iodine deficiency is common. Therefore, supplementation programs, such as the use of iodized salt or iodine supplements, are effective strategies for preventing and addressing iodine deficiency, leading to better health outcomes and a reduced risk of related health issues.
Conclusion
Ensuring adequate iodine intake is essential for maintaining
optimal health, particularly in areas where iodine deficiency is
common. Continued efforts to address iodine deficiency through
supplementation programs and public health initiatives are essential
to reduce the global burden of iodine deficiency disorders and
improve health outcomes for populations at risk.
References
Citation
Barseghyan NA, Davtyan HH. Clinical Aspects of Iodine and Iodine Deficiency. Indian J Nutri. 2025;12(2): 333.