Case Report
The Impact of Gut Microbiota Modulation on Bone Health and Rheumatoid Arthritis: A Case Report and Literature Review
Harpreet Kour1* and Shrihari L Kulkarni2
1Department of Physiology, Jawaharlal Nehru Medical College, KLE Academy of Higher Education and Research, Belagavi, Karnataka, India.
2Department of Orthopaedics, SDM College of Medical Sciences and Hospital, Shri Dharmasthala Manjunatheshwara University, Dharwad, Karnataka, India.
2Department of Orthopaedics, SDM College of Medical Sciences and Hospital, Shri Dharmasthala Manjunatheshwara University, Dharwad, Karnataka, India.
*Corresponding author:Harpreet Kour, Department of Physiology, Jawaharlal Nehru Medical College, KLE Academy of Higher Education and Research, Belagavi, Karnataka, India. E-mail Id: harpreetkour.kour@gmail.com
Article Information:Submission: 18/10/2025; Accepted: 12/11/2025; Published: 14/11/2025
Copyright: © 2025 Kour H, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: The gut-bone axis represents a novel paradigm in which gut microbiota influences bone metabolism and systemic inflammation. Dysbiosis has been implicated in the pathogenesis of rheumatoid arthritis (RA), suggesting that microbiome-targeted therapies could offer adjunct benefits.
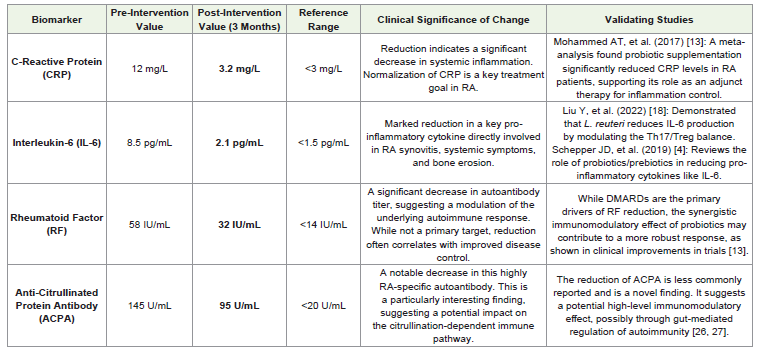
Case Presentation: A 52-year-old female presented with seropositive RA and concomitant irritable bowel syndrome (IBS). Stool analysis confirmed gut dysbiosis, characterized by an overgrowth of Prevotella copri and reduced commensal Lactobacillus and Bifidobacterium. In addition to initiating standard pharmacological therapy (methotrexate and adalimumab), a targeted microbiome modulation regimen was introduced, comprising a daily probiotic (Lactobacillus reuteri, 1×10⁹ CFU) and prebiotic (inulin, 10g). After three months of combined therapy, the patient reported significant improvements in joint pain, stiffness, and gastrointestinal symptoms. Objective biomarkers showed marked reduction: CRP decreased from 12 mg/L to 3.2 mg/L, IL-6 from 8.5 pg/ mL to 2.1 pg/mL, rheumatoid factor (RF) decreased from 58 IU/mL to 32 IU/mL, and anti-citrullinated protein antibody (ACPA) levels decreased from 145 U/ mL to 95 U/mL.
Conclusion: This case demonstrates significant clinical and serological improvement in a patient with RA following a combined pharmacological and microbiome-targeted intervention. It provides supportive evidence for the role of the gut-joint axis in RA and suggests that probiotics and prebiotics may be a beneficial adjunct to conventional therapy, warranting further investigation in controlled trials.
Case Presentation: A 52-year-old female presented with seropositive RA and concomitant irritable bowel syndrome (IBS). Stool analysis confirmed gut dysbiosis, characterized by an overgrowth of Prevotella copri and reduced commensal Lactobacillus and Bifidobacterium. In addition to initiating standard pharmacological therapy (methotrexate and adalimumab), a targeted microbiome modulation regimen was introduced, comprising a daily probiotic (Lactobacillus reuteri, 1×10⁹ CFU) and prebiotic (inulin, 10g). After three months of combined therapy, the patient reported significant improvements in joint pain, stiffness, and gastrointestinal symptoms. Objective biomarkers showed marked reduction: CRP decreased from 12 mg/L to 3.2 mg/L, IL-6 from 8.5 pg/ mL to 2.1 pg/mL, rheumatoid factor (RF) decreased from 58 IU/mL to 32 IU/mL, and anti-citrullinated protein antibody (ACPA) levels decreased from 145 U/ mL to 95 U/mL.
Conclusion: This case demonstrates significant clinical and serological improvement in a patient with RA following a combined pharmacological and microbiome-targeted intervention. It provides supportive evidence for the role of the gut-joint axis in RA and suggests that probiotics and prebiotics may be a beneficial adjunct to conventional therapy, warranting further investigation in controlled trials.
Keywords:Gut Microbiota; Rheumatoid Arthritis; Dysbiosis; Probiotics; Prebiotics; Gut-Bone Axis; Prevotella copri; Lactobacillus reuteri
Introduction
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune
disorder characterized by erosive synovitis, progressive joint
destruction, and systemic bone loss [1]. While the etiology remains
incompletely understood, a complex interplay between genetic
predisposition and environmental triggers is widely accepted.
Emerging research has identified the gut microbiota as a critical environmental factor modulating immune homeostasis [2,3]. The concept of the “gut-bone axis” and “gut-joint axis” describes the biochemical communication between gut microbial communities and distant organ systems, including bones and joints [4,5]. This communication occurs via mechanisms including immune cell priming, regulation of systemic inflammation, production of microbial metabolites (e.g., short-chain fatty acids, SCFAs), and modulation of nutrient absorption [2,4,6].
Emerging research has identified the gut microbiota as a critical environmental factor modulating immune homeostasis [2,3]. The concept of the “gut-bone axis” and “gut-joint axis” describes the biochemical communication between gut microbial communities and distant organ systems, including bones and joints [4,5]. This communication occurs via mechanisms including immune cell priming, regulation of systemic inflammation, production of microbial metabolites (e.g., short-chain fatty acids, SCFAs), and modulation of nutrient absorption [2,4,6].
A state of microbial imbalance, or dysbiosis, is frequently observed
in RA patients [7,8]. This dysbiosis is often marked by a reduction
in microbial diversity, a decrease in beneficial SCFA-producing
bacteria, and an expansion of pathobionts such as Prevotella copri,
which has been specifically linked to RA pathogenesis and disease
activity [9,10,11]. This altered microbiota can compromise intestinal
barrier integrity, leading to increased translocation of microbial
products (e.g., lipopolysaccharide, LPS) into systemic circulation,
thereby potentiating chronic inflammation and accelerating bone
erosion through upregulated osteoclast activity [2,4,12].
Consequently, therapeutic strategies aimed at restoring eubiosis, including the use of probiotics and prebiotics, have garnered significant interest for their potential anti-inflammatory and bone-protective effects [4,13,14]. We present a case of a patient with RA and confirmed gut dysbiosis who exhibited a robust clinical and serological response to a combination of conventional disease-modifying antirheumatic drugs (DMARDs) and a targeted microbiome modulation regimen.
Consequently, therapeutic strategies aimed at restoring eubiosis, including the use of probiotics and prebiotics, have garnered significant interest for their potential anti-inflammatory and bone-protective effects [4,13,14]. We present a case of a patient with RA and confirmed gut dysbiosis who exhibited a robust clinical and serological response to a combination of conventional disease-modifying antirheumatic drugs (DMARDs) and a targeted microbiome modulation regimen.
Case Presentation
A 52-year-old female presented to the orthopaedic clinic with a
chief complaint of progressive pain, swelling, and morning stiffness
in her wrists and knees, persisting for over four months. She also
reported significant fatigue and long-standing bowel irregularities
consistent with a prior diagnosis of irritable bowel syndrome (IBS),
including bloating and diarrhoea. She was on no prior medications.
Physical examination revealed symmetric swelling and tenderness in both wrists and knees. Morning stiffness lasted more than 60 minutes. Initial diagnostic investigations were significant for:
• Serology: Elevated inflammatory markers (C-reactive protein [CRP] 12 mg/L; normal <3 mg/L) and positive serology for rheumatoid factor (RF; 58 IU/mL) and anti-citrullinated protein antibodies (ACPA; 145 U/mL). Interleukin-6 (IL-6) was elevated at 8.5 pg/mL.
• Stool Microbiome Analysis: A comprehensive stool analysis (via 16S rRNA sequencing) revealed significant gut dysbiosis[15]. Key findings included a notable overgrowth of the genus Prevotella, with Prevotella copri identified as the dominant species [9,10]. Furthermore, there was a substantial reduction in the abundance of beneficial genera, including Lactobacillus and Bifidobacterium [7,16].
Physical examination revealed symmetric swelling and tenderness in both wrists and knees. Morning stiffness lasted more than 60 minutes. Initial diagnostic investigations were significant for:
• Serology: Elevated inflammatory markers (C-reactive protein [CRP] 12 mg/L; normal <3 mg/L) and positive serology for rheumatoid factor (RF; 58 IU/mL) and anti-citrullinated protein antibodies (ACPA; 145 U/mL). Interleukin-6 (IL-6) was elevated at 8.5 pg/mL.
• Stool Microbiome Analysis: A comprehensive stool analysis (via 16S rRNA sequencing) revealed significant gut dysbiosis[15]. Key findings included a notable overgrowth of the genus Prevotella, with Prevotella copri identified as the dominant species [9,10]. Furthermore, there was a substantial reduction in the abundance of beneficial genera, including Lactobacillus and Bifidobacterium [7,16].
Diagnosis:
Based on the 2010 ACR/EULAR classification criteria, a diagnosis
of seropositive rheumatoid arthritis (RA) was confirmed, supported
by the positive RF and ACPA (anti-CCP) results. The concomitant
gut dysbiosis was diagnosed in the context of her IBS symptoms and
microbiological findings.Therapeutic Intervention:
A multi-faceted treatment plan was initiated by orthopaedic
surgeon, Lifestyle medicine expert and a clinical nutritionist:
1. Pharmacological Therapy: Standard RA treatment was
commenced with oral methotrexate (15 mg weekly) and a
subcutaneous TNF-α inhibitor (adalimumab, 40 mg every
two weeks).2. Microbiome-Targeted Therapy: To address the underlying
dysbiosis, an adjunctive regimen was introduced:
a. Probiotic: A commercially available formulation
of Lactobacillus reuteri, providing 1 × 10⁹ colony-forming
units (CFU) per capsule. One capsule was administered
orally once daily in the morning on an empty stomach
[17,18].
b. Prebiotic: A purified chicory root-derived inulin
powder. The patient was instructed to dissolve 10
g (approximately one level tablespoon) in water or a cold
beverage and consume it daily before breakfast [19,20].
3. Lifestyle Modifications: The patient was counselled on
adopting an anti-inflammatory diet rich in fiber (from
diverse fruits, vegetables, and legumes), and low in processed
foods and added sugars. She was also encouraged to maintain
adequate hydration and engage in regular, low-impact
physical activity (e.g., walking, swimming) as tolerated.
Follow-up and Outcomes:
At the three-month follow-up, the patient reported a
substantial improvement in her clinical symptoms. Joint
pain and swelling were significantly reduced, and morning
stiffness decreased to less than 30 minutes. Her gastrointestinal
symptoms, including bloating and diarrhea, had resolved.
Repeat laboratory studies demonstrated remarkable improvement:• CRP decreased to 3.2 mg/L (normal: <3 mg/L)
• IL-6 decreased to 2.1 pg/mL (normal: <1.5 pg/mL)
• RF decreased to 32 IU/mL (normal: <14 IU/mL)
• ACPA decreased to 95 U/mL (normal: <20 U/mL)
This represented a significant reduction in systemic inflammation and autoantibody levels.
Discussion
This case illustrates the potential synergistic benefit of combining
conventional RA therapy with a gut microbiome modulation strategy.
The patient’s significant clinical and serological improvement,
alongside the resolution of GI symptoms, suggests a positive systemic
effect beyond standard care alone. The existing body of literature, as
summarized in Table 1, provides a strong scientific rationale for this
combined interventional approach and the mechanisms behind the
observed outcomes [7,9,17,19].
The observed dysbiosis, featuring Prevotella copri overgrowth and depleted Lactobacillus and Bifidobacterium, aligns with the described microbial signature in RA patients [7,8,9]. P. copri has been shown to promote Th17-mediated immune responses, leading to increased production of pro-inflammatory cytokines like IL-6 and IL-17, which are central to RA pathogenesis and osteoclast activation [9,11,12].
The intervention with Lactobacillus reuteri and inulin likely acted through several
The observed dysbiosis, featuring Prevotella copri overgrowth and depleted Lactobacillus and Bifidobacterium, aligns with the described microbial signature in RA patients [7,8,9]. P. copri has been shown to promote Th17-mediated immune responses, leading to increased production of pro-inflammatory cytokines like IL-6 and IL-17, which are central to RA pathogenesis and osteoclast activation [9,11,12].
The intervention with Lactobacillus reuteri and inulin likely acted through several
Complementary mechanisms:
1. Competitive Exclusion and Barrier Fortification: L.
reuteri can inhibit the growth of pathobionts, produce
antimicrobial compounds (e.g., reuterin), and enhance mucin
production, thereby strengthening the intestinal epithelial
barrier and reducing bacterial translocation [17,22].
2. Immunomodulation: Certain probiotic strains, including L. reuteri, can modulate the host immune system by promoting regulatory T-cell (Treg) differentiation and downregulating pro-inflammatory Th17 responses, leading to decreased systemic levels of TNF-α, IL-6, and IL-17 [18,23,24].
3. Metabolic Effects: The prebiotic inulin is fermented by colonic bacteria into SCFAs (e.g., butyrate, propionate). Butyrate is a crucial energy source for colonocytes, further enhancing barrier function [25]. More importantly, SCFAs have demonstrated direct immunomodulatory and boneprotective effects by suppressing osteoclastogenesis and promoting osteoblast activity [4,6,19,20].
The parallel improvement in both musculoskeletal and gastrointestinal symptoms underscores the interconnectedness of the gut-joint axis. The reduction in ACPA levels is particularly intriguing, as it suggests a potential modulation of the autoantibody response, possibly via effects on citrullinating enzymes or the presentation of autoantigens, a mechanism proposed in recent studies [26,27].
2. Immunomodulation: Certain probiotic strains, including L. reuteri, can modulate the host immune system by promoting regulatory T-cell (Treg) differentiation and downregulating pro-inflammatory Th17 responses, leading to decreased systemic levels of TNF-α, IL-6, and IL-17 [18,23,24].
3. Metabolic Effects: The prebiotic inulin is fermented by colonic bacteria into SCFAs (e.g., butyrate, propionate). Butyrate is a crucial energy source for colonocytes, further enhancing barrier function [25]. More importantly, SCFAs have demonstrated direct immunomodulatory and boneprotective effects by suppressing osteoclastogenesis and promoting osteoblast activity [4,6,19,20].
The parallel improvement in both musculoskeletal and gastrointestinal symptoms underscores the interconnectedness of the gut-joint axis. The reduction in ACPA levels is particularly intriguing, as it suggests a potential modulation of the autoantibody response, possibly via effects on citrullinating enzymes or the presentation of autoantigens, a mechanism proposed in recent studies [26,27].
Limitations:
As a single case report, our findings cannot establish causality
or be generalized to the broader RA population. The observed
improvements cannot be definitively disentangled from the effects
of the potent DMARD therapy (methotrexate and adalimumab).
The placebo effect, particularly on subjective symptoms, must also
be considered. Furthermore, while stool analysis showed a microbial
shift, sequential microbiome sequencing would be required to confirm
a direct correlation between the intervention and microbial changes.
Finally, the use of specific commercial products, while detailed for
reproducibility, may limit the generalizability of the results to other
probiotic strains or prebiotic sources.Conclusion and Future Directions
This case adds to the growing body of evidence supporting
the role of the gut microbiome in RA and highlights the potential
of microbiome-targeted therapies as a safe and promising adjunct
to standard treatment. It provides a strong rationale for designing
robust, randomized, placebo-controlled clinical trials to rigorously
evaluate the efficacy of specific probiotic and prebiotic strains, both
in improving RA disease activity and in mitigating the systemic bone
loss associated with chronic inflammation.
Declarations:
• Written informed consent was obtained from the patient for
the publication of this case report and any accompanying
data.• Competing Interests: The authors declare no competing interests.
• Funding: No funding was received for this work.
• Availability of Data and Materials: The data supporting this case report are available within the article. Further details are available from the corresponding author upon reasonable request.
• Disclaimer: The views expressed in this case report are solely those of the authors. The specific interventions described (including the use of Lactobacillus reuteri and inulin) are for informational and reproducibility purposes only and do not imply endorsement of any specific product or brand. The information contained herein is presented for academic and educational purposes and is not intended to replace professional medical advice, diagnosis, or treatment.
References
1. McInnes IB, Schett G (2011) The pathogenesis of rheumatoid arthritis. N Engl J Med 365: 2205-2219.
Citation
Kour H, Kulkarni SL. The Impact of Gut Microbiota Modulation on Bone Health and Rheumatoid Arthritis: A Case Report and Literature Review. Indian J Nutri. 2025;12(2): 330.