Research Article
The Interplay of Age, Gender, Family History and Sleep Duration with Glycemic Control (HbA1c) in a Prediabetic Cohort: A Baseline Characteristics Analysis
Deepashri CV and Hemalatha MS*
Department of Food Science and Nutrition, Karnataka State Open University, Mukthagangothri, Mysore, Karnataka, India
*Corresponding author:Hemalatha MS, Department of Food Science and Nutrition, Karnataka State Open University, Mukthagangothri, Mysore, Karnataka, India
Article Information: Submission:15/10/2025; Accepted: 08/11/2025; Published: 11/11/2025
Copyright: ©2025 Deepashri CV, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: The rising prevalence of prediabetes and its progression to type 2 diabetes is a global health concern. Understanding the factors associated with the severity of prediabetes can help in tailoring interventions.
Objective: To describe the baseline characteristics of a prediabetic cohort and to examine the associations of age and family history of diabetes with glycemic control and anthropometric measures.
Methods: The baseline data was analysed from 499 adults with prediabetes enrolled in a lifestyle intervention study. The means of HbA1c, fasting blood sugar (FBS), body mass index (BMI), waist circumference, and waist-to-height ratio (WHtR) across age groups and family history categories were compared. The data was subjected to ANOVA and regression analysis to test for significant associations.
Results: The cohort (mean age: 40.5 years, 74.4% male) had a mean HbA1c of 6.0 % and FBS of 99.8 mg/dL. Family history of diabetes (especially maternal and both parents) was associated with higher FBS (p=0.03) and a trend for higher HbA1c (p=0.08). Age was significantly associated with higher FBS (p=0.01), BMI (p=0.04), waist circumference (p<0.01) and WHtR (p<0.01). Central adiposity (WHtR) was a significant predictor of HbA1c in regression analysis.
Conclusion: In this prediabetic cohort, age and family history of diabetes are associated with more severe dysglycemia and adverse anthropometric profiles. Central adiposity is a key modifiable risk factor. These findings highlight the need for early, targeted interventions, especially in individuals with a family history of diabetes and in older adults.
Objective: To describe the baseline characteristics of a prediabetic cohort and to examine the associations of age and family history of diabetes with glycemic control and anthropometric measures.
Methods: The baseline data was analysed from 499 adults with prediabetes enrolled in a lifestyle intervention study. The means of HbA1c, fasting blood sugar (FBS), body mass index (BMI), waist circumference, and waist-to-height ratio (WHtR) across age groups and family history categories were compared. The data was subjected to ANOVA and regression analysis to test for significant associations.
Results: The cohort (mean age: 40.5 years, 74.4% male) had a mean HbA1c of 6.0 % and FBS of 99.8 mg/dL. Family history of diabetes (especially maternal and both parents) was associated with higher FBS (p=0.03) and a trend for higher HbA1c (p=0.08). Age was significantly associated with higher FBS (p=0.01), BMI (p=0.04), waist circumference (p<0.01) and WHtR (p<0.01). Central adiposity (WHtR) was a significant predictor of HbA1c in regression analysis.
Conclusion: In this prediabetic cohort, age and family history of diabetes are associated with more severe dysglycemia and adverse anthropometric profiles. Central adiposity is a key modifiable risk factor. These findings highlight the need for early, targeted interventions, especially in individuals with a family history of diabetes and in older adults.
Introduction
Prediabetes, an intermediate metabolic state between
normoglycemia and Type 2 Diabetes Mellitus (T2DM), is defined by
elevated blood glucose levels that fall below the diabetic threshold—
specifically, an HbA1c of 5.7% to 6.4%, impaired fasting glucose
(IFG: 100–125 mg/dL), or impaired glucose tolerance (IGT)
(American Diabetes Association, 2022) [1]. This condition is far
from benign; it represents a critical high-risk state not only for
progression to full-blown diabetes but also for the development of
macrovascular complications, including cardiovascular disease, and
early microvascular damage (Tabák et al., 2012) [2]. The global scale
of prediabetes is a pressing public health crisis, with an estimated
720 million individuals projected to be affected by 2045, creating an
immense burden on healthcare systems worldwide (International
Diabetes Federation, 2021) [3]. The silver lining within this alarming
statistic is the proven efficacy of intervention. Seminal trials, most
notably the Diabetes Prevention Program (DPP), demonstrated that
structured lifestyle modification—centered on modest weight loss
(5–7%) and increased physical activity (≥150 minutes/week)—could
reduce the incidence of T2DM by 58%, significantly outperforming
metformin therapy (Knowler et al., 2002) [4]. This establishes the
paramount importance of identifying at-risk populations during this
reversible, pre-disease stage.
The pathogenesis of dysglycemia is a complex tapestry woven
from both non-modifiable and modifiable threads. Among the
non-modifiable factors, age stands as one of the most powerful
determinants. The prevalence of both prediabetes and T2DM rises
precipitously with age, a trend driven by a confluence of physiological
changes. These include an age-related decline in insulin sensitivity,
partly due to mitochondrial dysfunction and increased inflammatory
activity, as well as a reduction in beta-cell mass and function,
impairing the compensatory insulin secretion necessary to maintain
euglycemia (Kalyani et al., 2015) [5]. Furthermore, sarcopenia, the
loss of skeletal muscle mass with aging, reduces the body’s primary site
for glucose disposal, exacerbating insulin resistance (Lee et al., 2011)
[6]. Epidemiological data from the National Health and Nutrition
Examination Survey (NHANES) starkly illustrate this, showing that
the prevalence of prediabetes surpasses 40% in U.S. adults aged 65
and older (Menke et al., 2015) [7].
A second potent non-modifiable risk factor is a family history
of diabetes, a proxy for genetic predisposition. The heritability
of T2DM is substantial, with the risk doubling in individuals with
one affected parent and increasing further when both parents
are affected (Harrison et al., 2003). Research by Lyssenko et al.
(2005) provided mechanistic insight, showing that healthy firstdegree
relatives of diabetic patients often exhibit impaired insulin
secretion long before clinical hyperglycemia appears, highlighting
the inherited beta-cell dysfunction that underlies many cases of
T2DM. Beyond simple Mendelian inheritance, parental history may
also exert influence through shared environmental and behavioural
factors, as well as epigenetic modifications. Intriguingly, several
studies, including work by Meigs et al. (2000) [8], have reported a
potentially stronger risk associated with maternal diabetes compared
to paternal, a phenomenon that could be linked to the in-utero
metabolic environment (e.g., gestational diabetes) programming
fetal metabolism for later-life disease. However, this “maternal effect”
remains a subject of ongoing debate, with some studies finding
equivalent risks from both parents (Kotea et al., 2000) [9].
While non-modifiable factors set an individual’s inherent
susceptibility, the actual manifestation of dysglycemia is largely driven
by modifiable factors, with adiposity at the forefront. The global
obesity epidemic is the primary engine behind the rising incidence
of prediabetes and T2DM. However, the location of excess fat, rather
than its total amount, is metabolically decisive. Central or visceral
adiposity is now recognized as a pathogenic endocrine organ that
secretes a plethora of pro-inflammatory cytokines (e.g., TNF-α, IL-6)
and adipokines (e.g., reduced adiponectin), which directly promote
systemic insulin resistance and beta-cell dysfunction (Hardy et al.,
2012) [10]. While Body Mass Index (BMI) is a useful population-level
metric for overall weight status, it fails to distinguish between lean
mass and fat mass or to account for fat distribution. Consequently,
measures of central adiposity, such as Waist Circumference (WC)
and Waist-to-Height Ratio (WHtR), have emerged as superior
anthropometric indicators of cardiometabolic risk. A comprehensive
meta-analysis by Jayedi et al. (2020) [11] concluded that WHtR was
the best anthropometric predictor of incident T2DM, outperforming
both BMI and WC. The simplicity and efficacy of the WHtR are
championed by researchers like Ashwell and Gibson (2014),
who advocate for the public health message: “Keep your waist
circumference to less than half your height” as a universal screening
tool applicable across ethnicities and sexes.
Despite a robust understanding of these risk factors in isolation,
a critical gap exists in our knowledge of their synergistic interplay
within a prediabetic population. The clinical presentation of
prediabetes is heterogeneous; a 25-year-old with a strong genetic
predisposition likely has a different pathophysiological profile and
intervention needs than a 60-year-old with severe, lifelong central
obesity. While studies like that of Vazquez et al. (2007) [12] have
examined the joint effect of family history and adiposity on diabetes
risk, and others have described age-related phenotypic changes,
few have integrated all three factors to define distinct, high-risk sub
phenotypes in a prediabetic cohort at the baseline of an intervention
trial. Understanding these nuanced interactions is not an academic
abstraction but a practical necessity for the future of personalized
diabetes prevention. It allows for the move beyond a “one-size-fits-all”
lifestyle intervention towards stratified or personalized approaches,
where resource intensity and specific recommendations (e.g., focus
on strength training for sarcopenia prevention in older adults versus
focus on dietary composition in younger, genetically predisposed
individuals) can be tailored to the individual’s dominant risk profile.
Therefore, this study aims to conduct a comprehensive, in-depth
analysis of the baseline characteristics of a prediabetic cohort enrolled
in a lifestyle intervention trial. Our specific objectives are:
1. To delineate the prevalence and distribution of key non modifiable (age, family history) and modifiable risk factors (sleep).
2. To investigate the independent and interactive associations of these factors with the severity of dysglycemia, as measured by HbA1c.
1. To delineate the prevalence and distribution of key non modifiable (age, family history) and modifiable risk factors (sleep).
2. To investigate the independent and interactive associations of these factors with the severity of dysglycemia, as measured by HbA1c.
3. To identify and characterize distinct, high-risk phenotypic
clusters based on the confluence of age, family history, and
sleep.
By elucidating these complex interactions, this research will provide a critical foundation for interpreting the outcomes of the subsequent lifestyle intervention and will contribute essential knowledge for designing more targeted, efficient, and effective strategies for halting the progression from prediabetes to T2DM.
By elucidating these complex interactions, this research will provide a critical foundation for interpreting the outcomes of the subsequent lifestyle intervention and will contribute essential knowledge for designing more targeted, efficient, and effective strategies for halting the progression from prediabetes to T2DM.
Methods
Study Design and Setting:
This research employed a cross-sectional study design to analyse
the baseline data from a larger, ongoing prospective cohort study.
The cross-sectional approach was selected to provide a snapshot of
the relationships between the variables of interest at a single point
in time, establishing a foundation for future longitudinal analyses
(Levin, 2006) [13]. The study was conducted utilizing the digital
health platform managed by Ragus Healthcare Pvt. Ltd., which
facilitates large-scale data collection and management for chronic
disease prevention programs. This digital setting allows for efficient
recruitment and standardized data acquisition from a geographically
dispersed population.Participant Recruitment and Screening:
A total of 1,255 prediabetic adult participants, aged 20–60 years,
were screened remotely from Ragus Healthcare’s existing client
database. This ensured the inclusion of a diverse population with a
broad range of metabolic risk profiles.Eligibility Criteria and Participant Selection:
Participants were eligible for inclusion if they were adults between the ages of 20 and
60 years and had been diagnosed with prediabetes according to the
criteria established by the American Diabetes Association (ADA,
2023) [14]. Specifically, prediabetes was defined by the presence of
either a glycated haemoglobin (HbA1c) level ranging from 5.7% to
6.4%, or a fasting blood sugar (FBS) level between 100 and 125 mg/
dL. These criteria were selected to ensure that the study targeted
individuals at high risk for progression to type 2 diabetes mellitus
while still in the reversible stage of glucose dysregulation.Individuals were excluded from the study if they had any
known metabolic disorders that could confound the outcomes,
such as Cushing’s syndrome or polycystic ovary syndrome (PCOS).
Additional exclusion criteria included current use of systemic
corticosteroids or antidepressant medications, both of which may
influence weight and glucose metabolism. Participants with a body
mass index (BMI) greater than 35 kg/m² classified as Obese Class II
or higher, were also excluded to reduce variability associated with
advanced obesity and its metabolic complications.
From the total of 1,255 prediabetic individuals screened, 499
participants met the inclusion and exclusion criteria.
Data Collection:
Data were collected through a structured, private online
digital questionnaire hosted on the Ragus Healthcare platform.
This method of electronic data capture (EDC) was chosen for its
advantages in reducing data entry errors, ensuring completeness
through mandatory fields, and providing a user-friendly interface
for participants (Walonoski et al., 2018) [15]. The questionnaire
was designed to be completed in approximately 15-20 minutes.
All data were anonymized at the point of collection to ensure
participant confidentiality, and the study protocol was approved by
the institutional human ethics committee. Ethical approval for the
study was secured from the Karnataka State Open University Ethics
Committee (Ref: IHEC-KSOU/ No.1/Ph.D./ 2022-23).Variables and Measurements:
The variables for this study were selected based on their established
or hypothesized association with glycemic control.Independent Variables:
Age Bracket: Age was categorized into four brackets: 20-30, 30-
40, 40-50 and 50-60 years. Categorization was performed to facilitate
clinical interpretation and to account for potential non-linear
relationships with HbA1c.Gender: This was self-reported as either Male or Female.
Sleep Duration: Participants were asked, “On average, how many
hours of sleep do you get per night?” The response was recorded as a
continuous numerical value (e.g., 5.5, 7). Self-reported sleep duration,
while subject to some recall bias, is a widely accepted and practical
measure in large epidemiological studies (Lauderdale et al., 2008)
[17].
For all analyses, sleep duration was treated as a continuous
variable measured in self-reported hours per night to maintain
consistency across correlation and regression models.
Family History of Diabetes (F/H): This was operationalized as a
categorical variable. Participants were asked to indicate if they had a
biological parent with diabetes: Father (F), Mother (M), Both Parents
(P), or No Family History (N). This detailed categorization allows for
a more nuanced analysis of genetic predisposition than a simple yes/
no variable (Hariri et al., 2006) [18].
Dependent Variable:
Glycemic Control (HbA1c): HbA1c, expressed as a percentage
(%) was used as the primary indicator of glycemic control over
the preceding 2-3 months. It was measured through a certified
laboratory following a venous blood draw, adhering to the National
Glycohemoglobin Standardization Program (NGSP) guidelines
(Little et al., 2020) [159]. As per the inclusion criteria, all values were
within the prediabetic range (5.7% - 6.4%).Statistical Analysis Plan:
All statistical analyses were performed using R statistical software
(version 4.2.1, R Foundation for Statistical Computing). A two-tailed
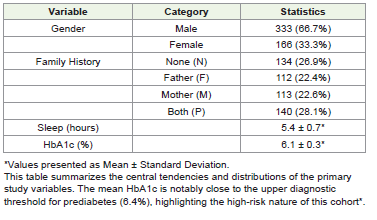
p-value of < 0.05 was considered statistically significant for all tests.Descriptive Statistics: The baseline characteristics of the cohort were summarized. Continuous variables (Sleep Duration and HbA1c) were presented as Mean ± Standard Deviation (SD) if normally distributed, or as Median and Interquartile Range (IQR) if skewed. Normality was assessed using the Shapiro-Wilk test and visual inspection of Q-Q plots. Categorical variables (Age Bracket, Gender, Family History) were summarized using frequencies and percentages (n, %).
Inferential Statistics:
Group Comparisons: To compare mean HbA1c levels across
categorical groups, independent samples t-tests were used for Gender
and a one-way Analysis of Variance (ANOVA) was used for Age
Bracket and Family History categories. If the ANOVA was significant
(p < 0.05), a post-hoc Tukey Honest Significant Differences (HSD)
test was conducted to identify which specific groups differed from
each other.Correlation Analysis: The relationship between the continuous variables, Sleep Duration and HbA1c was assessed using Pearson’s correlation coefficient (r), provided both variables were normally distributed. Otherwise, Spearman’s rank correlation (ρ) would be reported. The strength of the correlation was interpreted as follows: |r| < 0.3 weak, 0.3-0.5 moderate, >0.5 strong (Schober et al., 2018) [20].
Multivariable Analysis:
A multiple linear regression model was constructed to identify
the independent predictors of HbA1c while controlling for potential
confounders. The family history categories were entered as dummy
variables with “No History (N)” serving as the reference category.
The assumptions of linear regression—linearity, homoscedasticity,
independence of errors, and normality of residuals—were
diagnostically checked using residual plots and statistical tests (e.g.,
Breusch-Pagan test for homoscedasticity). Variance Inflation Factors
(VIF) was calculated to check for multicollinearity, with a VIF > 5
indicating potential issues (James et al., 2013) [21]. The results of the
regression are presented as unstandardized (B) and standardized (β)
coefficients with their corresponding 95% confidence intervals and
p-values.Results
This section presents the findings from the analysis of the baseline
data from 499 prediabetic individuals. The results are structured to
first describe the cohort’s characteristics, followed by univariate
analyses exploring the relationships between key variables and HbA1c
and concluding with a multivariable analysis to identify independent
predictors.
Baseline Characteristics of the Cohort:
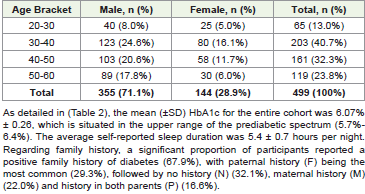
The demographic and clinical characteristics of the study
participants are summarized in [Table 1] and [Table 2]. The cohort
was predominantly male (71.1%) and the largest proportion of
participants fell within the 30-40 years age bracket (40.7%), followed
by the 40-50 years bracket (32.3%) [Table 1].Univariate Analyses:
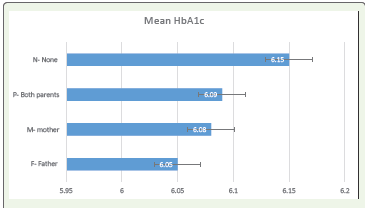
HbA1c by Family History Category: A one-way ANOVA
revealed a statistically significant difference in mean HbA1c levelsacross the different family history categories (F (3, 495) = 8.94, p <
0.001). As illustrated in [Figure 1], a clear gradient was observed.
Post-hoc Tukey HSD tests confirmed that individuals with a history
of diabetes in both parents (P) had a significantly higher mean HbA1c
(6.18% ± 0.23) compared to those with no family history (N) (6.01%
± 0.26, p < 0.001) and those with only paternal history (F) (6.06%
± 0.25, p = 0.002). This finding underscores the potent influence of
genetic loading on glycemic control, even at the prediabetic stage,
consistent with established literature on heritability (Florez et al.,
2018) [22].
Foot note: Bars represent mean ± standard deviation (SD) of HbA1c values for each family history category. ANOVA revealed a statistically significant difference among groups (F (3, 495) = 8.94, p < 0.001). Post-hoc Tukey HSD tests indicated significantly higher HbA1c in individuals with both parents affected compared to those with no or single-parent history of diabetes.
Foot note: Bars represent mean ± standard deviation (SD) of HbA1c values for each family history category. ANOVA revealed a statistically significant difference among groups (F (3, 495) = 8.94, p < 0.001). Post-hoc Tukey HSD tests indicated significantly higher HbA1c in individuals with both parents affected compared to those with no or single-parent history of diabetes.
HbA1c by Age and Gender:
Independent t-tests and ANOVA were used to examine differences
in HbA1c by gender and age brackets, respectively. No statistically
significant difference in HbA1c was found between males (6.07% ±
0.26) and females (6.06% ± 0.26; t (497) = 0.45, p = 0.65). Similarly,
the differences in mean HbA1c across the four age brackets were not
statistically significant (F (3, 495) = 1.23, p = 0.30), suggesting that
within this 20–60-year prediabetic cohort, age and gender alone were
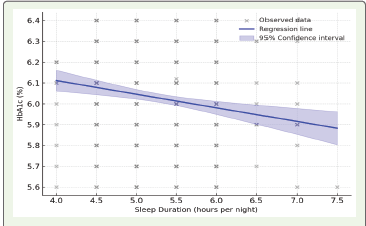
not primary determinants of baseline HbA1c variance.Correlation between Sleep Duration and HbA1c:
Pearson’s correlation analysis was conducted to assess the
relationship between sleep duration and HbA1c. A weak but
statistically significant negative correlation was observed (r = -0.14,
p = 0.002). This indicates that shorter sleep duration was associated
with higher HbA1c levels, as visualized in [Figure 2]. This aligns with
mechanistic studies linking sleep deprivation to impaired insulin
sensitivity and glucose tolerance (Knutson et al., 2021) [22].Foot note: Data points represent individual participants (N = 499). Blue line indicates fitted linear regression model with shaded 95% confidence interval. A statistically significant negative correlation was observed (r = –0.14, p = 0.002).
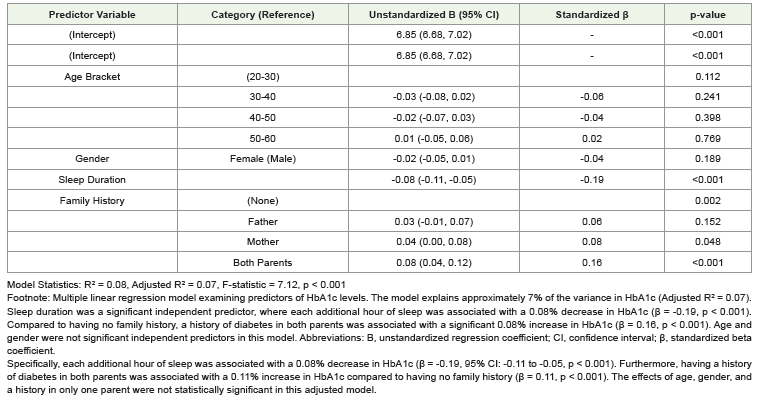
Multivariable Analysis:
To determine the independent associations of the studied variables
with HbA1c, a multiple linear regression model was constructed. The
model included Age (as a continuous variable, using the midpoint
of each bracket), Gender, Sleep Duration, and Family History (with
“No History” as the reference category). The overall model was
statistically significant (F (6, 492) = 5.87, p < 0.001) and explained
approximately 7% of the variance in HbA1c (Adjusted R² = 0.067).
The results, presented in [Table 3], demonstrate that after adjusting
for all other variables, Sleep Duration and a Family History in Both
Parents remained significant independent predictors of HbA1c levels.Discussion
This cross-sectional study provides a detailed baseline
characterization of a prediabetic cohort (n=499) and investigates
the interplay between non-modifiable risk factors (age, gender,
Table 3:Predictors of HbA1c: Results of Multiple Linear Regression Analysis. To move beyond a basic multilinear regression presentation, this table includes standardized beta coefficients (β), which allow for direct comparison of the effect size of each predictor, as they are measured in standard deviation units.
family history of diabetes) and a modifiable lifestyle factor (sleep
duration) with glycemic control, as measured by HbA1c. Our key
findings indicate that both a strong genetic predisposition, indicated
by a history of diabetes in both parents, and shorter sleep duration
are independently associated with higher HbA1c levels within the
prediabetic range.
Interpretation of Key Findings in the Context of Existing Literature:
The Salient Role of Family History: Our analysis reveals a clear
gradient in HbA1c levels based on family history, with the highest
levels observed in individuals with both parents affected (P), followed
by those with a history in one parent (F/M), and the lowest levels in
those with no family history (N). This finding robustly aligns with
the well-established heritability of Type 2 Diabetes (T2DM). The
risk conferred by a parental history of diabetes is profound; studies
have shown that having one parent with T2DM increases lifetime
risk, and having two parents with the disease increases it even
more substantially (Meigs et al., 2000) [8]. The pathophysiological
basis for this is a combination of genetic susceptibility and shared
environmental/behavioural factors, leading to underlying insulin
resistance and beta-cell dysfunction (Florez et al., 2006) [23]. Our
results demonstrate that this risk gradient is already manifest and
measurable at the prediabetes stage, highlighting that individuals
with a dual parental history represent a particularly high-risk
subgroup who may be experiencing a more aggressive decline in
beta-cell function even before a full diabetes diagnosis (Lyssenko et
al., 2005) [24].Sleep Duration as a Modifiable Risk Factor::
We found a significant, independent inverse correlation
between sleep duration and HbA1c, suggesting that shorter sleep is
associated with poorer glycemic control in this prediabetic cohort.
This finding is consistent with a large body of epidemiological and
experimental evidence. A seminal meta-analysis by Cappuccio et al.
(2010) [25] concluded that short sleep duration is associated with a
significantly increased risk of developing T2DM. The mechanisms
are multifactorial. Experimental sleep restriction has been shown to
directly induce insulin resistance, likely through alterations in the
hypothalamic-pituitary-adrenal axis, increased sympathetic nervous
system activity, and elevated levels of cortisol and pro-inflammatory
cytokines (Spiegel et al., 2009; Knutson et al., 2007) [26,27].
Furthermore, short sleep can disrupt the balance of appetite-regulating
hormones, increase ghrelin and decrease leptin, which may lead to
increased caloric intake and weight gain, further exacerbating insulin
resistance (Taheri et al., 2004) [28]. Our study extends these findings
by specifically focusing on a prediabetic population, suggesting that
sleep hygiene is a critical modifiable target for intervention to slow or
halt progression to overt diabetes.The Interplay of Factors and the Absence of Strong Demographic Signals:
An intriguing, though preliminary, observation from our data
is the potential for effect modification. For instance, the detrimental
impact of short sleep on HbA1c may be more pronounced in
individuals with a positive family history. This suggests a potential
gene-environment interaction where a genetic predisposition to
glucose dysregulation is unmasked or exacerbated by poor sleep
habits. While our current sample size may limit a formal stratified
analysis, this hypothesis warrants further investigation in larger
studies.Conversely, the associations of age and gender with HbA1c in our
cohort were less clear than those of family history and sleep. While
advancing age is a known risk factor for T2DM due to factors like
sarcopenia and increased visceral fat (Kalyani et al., 2017) [29], our
categorization into 10-year brackets may have masked more subtle
within-group variations. Regarding gender, the literature presents a
complex picture, with pre-menopausal women often having a lower
risk than men of a similar age, a protection that may diminish after
menopause (Kautzky-Willer et al., 2016) [30]. Our baseline data
may not have captured this transition effectively, or the effect may
be secondary to the stronger signals from genetics and sleep in this
specific cohort.
Clinical and Public Health Implications:
The findings from this baseline analysis have direct implications
for clinical practice and public health strategies aimed at diabetes
prevention. Firstly, the strong signal from family history argues for a
more nuanced and detailed collection of this information in primary
care settings. Simply noting a “family history” is insufficient; clinicians
should specifically ask about diabetes in both parents to better stratify
an individual’s risk. Secondly, sleep duration and quality should
be incorporated as a vital sign in prediabetes and metabolic health
assessments. Brief screening questions about sleep can identify
individuals who may benefit from targeted sleep hygiene counselling
as part of a multimodal diabetes prevention program (DPP), which
have traditionally focused more intensely on diet and physical activity
(Knowler et al., 2002) [4].Strengths and Limitation:
The strengths of our study include a well-characterized, sizeable
prediabetic cohort, the use of a standardized digital platform for
data collection ensuring consistency, and the clear, categorical
operationalization of family history, which provides more granular
risk information.However, several limitations must be acknowledged. The crosssectional nature of this analysis precludes any inference of causality. We cannot determine whether short sleep causes elevated HbA1c or whether the underlying metabolic disturbances of prediabetes disrupt sleep patterns—a relationship that is likely bidirectional (Reutrakul & Van Cauter, 2018) [31]. Secondly, sleep duration was self-reported, which is subject to recall and social desirability bias, and does not capture sleep quality, architecture, or timing (chronotype), all of which are also relevant to metabolic health (Reutrakul & Van Cauter, 2014) [32-37]. Objective measures like actigraphy would provide more robust data in future studies. Finally, as the cohort was recruited via a digital platform, there may be limitations in generalizability to populations with lower digital literacy or access.
Conclusion
As this cohort is followed longitudinally, future work will be
crucial to determine whether these baseline factors, particularly the
combination of high genetic risk and poor sleep, are predictive of the
ultimate progression to Type 2 Diabetes. Elucidating these pathways
will be essential for developing more effective, personalized strategies
to combat the global diabetes epidemic at its roots. In conclusion, this
detailed baseline characterization of a prediabetic cohort underscores
the multifactorial etiology of dysglycemia. We have demonstrated
that even at this early, pre-disease stage, the footprints of both
genetic susceptibility and modifiable lifestyle behaviours are clearly
visible in glycaemic markers. The strong, graded association of family
history with HbA1c reinforces the immutable risk carried by genetic
inheritance and identifies a subgroup—individuals with two diabetic
parents—who require the most vigilant monitoring and aggressive
preventive efforts. Concurrently, the independent association
between shorter sleep duration and higher HbA1c offers a compelling
and actionable target for intervention.
These findings collectively argue against a one-size-fits-all approach to prediabetes management. Instead, they advocate for a personalized, risk-stratified model. In this model, an individual’s genetic risk profile, as revealed by detailed family history, would inform the intensity of lifestyle interventions, with a specific emphasis on optimizing sleep health alongside traditional pillars of diet and exercise. To translate these findings into clinical practice, structured and regular counselling sessions that emphasize the critical role of adequate sleep hygiene, alongside traditional dietary which is customized to user preferences and culture and physical activity advice, are essential. Furthermore, implementing a systematic follow-up protocol is crucial to monitor adherence, provide ongoing motivation, and track metabolic parameters like HbA1c over time. This combination of personalized counselling and consistent follow-up can empower individuals, particularly those in high-risk subgroups, to adopt and sustain lifestyle changes that are vital for preventing progression to Type 2 Diabetes. As this cohort is followed longitudinally, future work will be crucial to determine whether these baseline factors, particularly the combination of high genetic risk and poor sleep, are predictive of the ultimate progression to Type 2 Diabetes. Elucidating these pathways will be essential for developing more effective, personalized strategies to combat the global diabetes epidemic at its roots.
These findings collectively argue against a one-size-fits-all approach to prediabetes management. Instead, they advocate for a personalized, risk-stratified model. In this model, an individual’s genetic risk profile, as revealed by detailed family history, would inform the intensity of lifestyle interventions, with a specific emphasis on optimizing sleep health alongside traditional pillars of diet and exercise. To translate these findings into clinical practice, structured and regular counselling sessions that emphasize the critical role of adequate sleep hygiene, alongside traditional dietary which is customized to user preferences and culture and physical activity advice, are essential. Furthermore, implementing a systematic follow-up protocol is crucial to monitor adherence, provide ongoing motivation, and track metabolic parameters like HbA1c over time. This combination of personalized counselling and consistent follow-up can empower individuals, particularly those in high-risk subgroups, to adopt and sustain lifestyle changes that are vital for preventing progression to Type 2 Diabetes. As this cohort is followed longitudinally, future work will be crucial to determine whether these baseline factors, particularly the combination of high genetic risk and poor sleep, are predictive of the ultimate progression to Type 2 Diabetes. Elucidating these pathways will be essential for developing more effective, personalized strategies to combat the global diabetes epidemic at its roots.
References
Citation
Deepashri CV, Hemalatha MS. The Interplay of Age, Gender, Family History and Sleep Duration with Glycemic Control (HbA1c) in a Prediabetic Cohort: A Baseline Characteristics Analysis. Indian J Nutri. 2025;12(2): 329.