Review Article
The Gut Skin Connection in Acne and Rosacea: Therapeutic Roles of Probiotics and Prebiotics
Sushmitha VL*, Madalagiri D and Chennappa G
Department of Food Technology, MS Ramaiah University of Applied Sciences, M S R Nagar, Mathikere, Bengaluru, Karnataka India
*Corresponding author: Dr. Sushmitha VL, Department of Food Technology, MS Ramaiah University of Applied Sciences, New Bellary Rd, M S R Nagar, Mathikere, Bengaluru, Karnataka India. Email Id: dr.sushmithaprajwal@gmail.com, dr.sushmithavl.93@gmail.com
Article Information: Submission: 29/05/2025; Accepted: 20/06/2025; Published: 24/06/2025
Copyright: © 2025 Sushmitha VL, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Acne and rosacea are chronic inflammatory skin disorders increasingly linked to disturbances in the gut-skin axis. This review explores how probiotics and prebiotics modulate immune responses, enhance epithelial barrier function, and influence gut and skin microbiota to improve dermatological outcomes.
Clinical and mechanistic evidence shows that specific strains and fibers reduce inflammatory markers, lesion counts, and symptoms such as erythema and sensitivity. Oral and topical formulations demonstrate promise as adjunctive therapies. Despite encouraging findings, standardized clinical trials and personalized approaches are needed to fully integrate microbiome-targeted strategies into dermatology for the effective management of acne and rosacea.
Keywords: Probiotics; Prebiotics; Acne; Rosacea; Gut-Skin Axis; Inflammation
Introduction
Acne and rosacea are chronic inflammatory skin disorders
with distinct clinical presentations but overlapping pathogenic
mechanisms. Acne is primarily linked to the over colonization
of Cutibacterium acnes in the pilosebaceous unit, triggering an
immune response characterized by the release of inflammatory
cytokines such as IL-1β and TNF-α [1]. Conversely, rosacea
manifests with persistent erythema, papules, and pustules,
with its pathogenesis closely associated with abnormal
vascular reactivity and the inflammatory response mediated by
triggers including Demodex folliculorum mites and ultraviolet
radiation [2]. Emerging evidence implicates the gut-skin axis
as a significant contributor to both conditions. Dysbiosis
in the intestinal microbiota may disrupt systemic immune
homeostasis and promote cutaneous inflammation via proinflammatory
mediators [3]. Several studies have demonstrated
that interventions with probiotics and prebiotics can restore
microbial balance and modulate inflammatory pathways, thereby
improving clinical outcomes in both acne and rosacea [4].
Future research is warranted to explore the specific
strains and prebiotic formulations that effectively target the
inflammatory cascades implicated in these disorders [5].
Enhanced understanding of these underlying mechanisms
may facilitate the development of comprehensive microbiome based
therapies for inflammatory skin conditions [6]. Moreover,
ongoing clinical trials are evaluating optimal dosages and
formulations, paving the way for personalized dermatological
interventions [7].
Role of Probiotics in Skin Health
Mechanisms of Action:
Probiotics influence skin health through immunomodulatory,
antimicrobial, and barrier-reinforcing effects. They downregulate
pro-inflammatory cytokines such as IL-8, TNF-α, and IL-1β, which
are commonly elevated in acne and rosacea lesions [8]. Certain
strains, like Lactobacillus rhamnosus GG and Bifidobacterium
longum, enhance skin barrier integrity by increasing tight
junction proteins and reducing transepidermal water loss
(TEWL) [9].Additionally, probiotics produce bacteriocins and organic
acids that suppress skin pathogens including Cutibacterium
acnes [10]. Metabolites from probiotic fermentation—such as
short-chain fatty acids (SCFAs)—also act as signaling molecules
that support mucosal immunity and anti-inflammatory pathways
[11].
Evidence from Clinical Trials:
The emerging understanding of the gut-skin axis has
spurred interest in microbiome-modulating therapies for
inflammatory skin diseases such as acne vulgaris and rosacea.
Several clinical trials have investigated the efficacy of probiotic
supplementation—either alone or alongside conventional
therapies—with promising results. Below are key studies that
have contributed meaningful evidence toward this therapeutic
approach.a. Oral Probiotics for Acne Vulgaris: A Double-Blind Randomized Trial:
A randomized, double-blind, placebo-controlled clinical
trial conducted by Setó-Torrent et al. (2024) evaluated the
effectiveness of oral probiotics in patients with acne vulgaris. The
study enrolled patients aged 12 to 30 years and administered a
capsule containing Lacticaseibacillus rhamnosus (CECT 30031)
and Arthrospira platensis (BEA_IDA_0074B) daily for 12 weeks.
The probiotic group demonstrated a statistically significant
reduction in both inflammatory and non-inflammatory acne
lesions compared to the placebo group. Improvement was
also observed in overall acne severity scores, highlighting the
immunomodulatory and barrier-supporting benefits of the
probiotic formulation [1].b. Adjunctive Probiotics with Antibiotics in Acne Management:
In a separate randomized, double-blind, controlled clinical
trial from Iran University of Medical Sciences (2024), 80
patients with moderate acne were treated either with standard
topical therapy alone or with the addition of oral probiotics and
doxycycline. The probiotic group showed significantly greater
improvements in acne severity, particularly in lesion count on
the chin, nose, and forehead. The study concluded that combining
probiotics with systemic antibiotics may enhance treatment
outcomes, reduce inflammation more effectively, and potentially
mitigate antibiotic-associated dysbiosis [2].c. Probiotics in Rosacea: Targeting the Gut-Skin Axis:
Sánchez-Pellicer et al. (2024) discussed multiple clinical
findings related to the use of probiotics in rosacea management
in their review published in Frontiers in Medicine. One
study highlighted within the review involved patients with
papulopustular rosacea receiving oral probiotics—such as
Escherichia coli Nissle 1917—alongside standard rosacea
treatment.Patients who received the probiotic demonstrated better
clinical improvement, reduced erythema, and faster recovery
compared to those receiving conventional therapy alone. These
findings reinforce the concept that modulating the gut microbiota
may influence systemic and skin-level inflammation, especially
in conditions such as rosacea, where immune dysregulation and
gastrointestinal comorbidities (e.g., small intestinal bacterial
overgrowth) are prevalent [3].
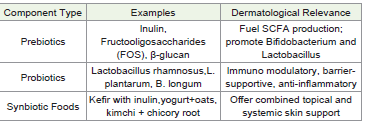
Role of Prebiotics in Modulating Skin Disorders
Prebiotic Mechanisms and Microbiome Modulation:
Prebiotics, such as fructooligosaccharides (FOS) and
galactooligosaccharides (GOS), selectively stimulate the growth
of beneficial gut bacteria like Lactobacillus and Bifidobacterium
spp., which indirectly modulate systemic and cutaneous
inflammation [15]. They also promote the production of SCFAs
like butyrate and acetate, which improve intestinal barrier
function and reduce systemic endotoxemia—a factor implicated
in acne pathogenesis [16]. Moreover, SCFAs exert epigenetic
control over immune cells, dampening Th17 responses associated
with rosacea [17].Evidence from Human and Animal Studies:
In animal models, dietary inulin-type fructans improved both
gut microbiota composition and skin inflammation scores after
UVB exposure [18]. Human trials, although fewer, suggest similar
benefits. A placebo-controlled trial by Gueniche et al. showed that
a topical prebiotic formulation containing α-glucooligosaccharide
improved skin hydration and reduced sensitivity in subjects
with rosacea-prone skin [19]. When combined with probiotics,
prebiotics (as synbiotics) have demonstrated superior outcomes
in modulating immune responses and microbiota stability [20].The Role of Synbiotics in Acne and Rosacea with mechanistic Insights & dietary Applications:
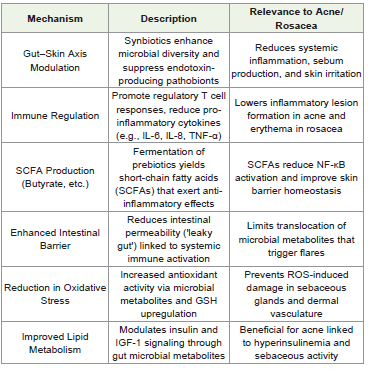
Synbiotics are a synergistic combination of probiotics (live
beneficial microbes) and prebiotics (non-digestible fibers that
selectively stimulate probiotic growth). Their integration into
dermatological nutrition has shown promise in modulating the
gut–skin axis, influencing systemic and cutaneous immunity, and
improving skin barrier function in inflammatory skin disorders
like acne vulgaris and rosacea.Synbiotics represent a promising, non-pharmacological
adjunct in the management of acne and rosacea, particularly by:
a. Strengthening intestinal and cutaneous barriers
b. Reducing systemic and local inflammation
c. Modulating lipid metabolism and insulin responses
d. Improving microbial diversity and antioxidant defense
Pathophysiology of Acne and Rosacea
Inflammatory Mediators: IL-1β, TNF-α, IL-8:
Acne and rosacea are primarily driven by dysregulated
immune responses, were pro-inflammatory cytokines play
a central role. In acne, Cutibacterium acnes stimulates
keratinocytes and monocytes to release interleukin-1β (IL-1β),
interleukin-8 (IL-8), and tumor necrosis factor-alpha (TNF-α),
leading to the recruitment of neutrophils and the formation
of come dones and pustules [8]. IL-1β has been identified as a
key initiator of microcome done formation by inducing hyper
keratinization, while IL-8 contributes to neutrophil chemotaxis
and papule development [9]. In rosacea, increased expression
of IL-1β and cathelicidin peptides has been reported in lesional
skin, contributing to inflammatory cell infiltration and vascular
dilation [10]. The activation of Toll-like receptor 2 (TLR2) in
innate immune cells plays a pivotal role in both conditions,
amplifying cytokine release in response to microbial or
environmental triggers [11].Role of Cutibacterium acnes and Demodex folliculorum:
Microbial dysbiosis contributes significantly to the onset
and persistence of acne and rosacea. Cutibacterium acnes
(formerly Propionibacterium acnes) is a Gram-positive anaerobe
that inhabits sebaceous follicles. Although part of the normal
skin flora, certain phylotypes of C. acnes (notably IA1) are
pro-inflammatory and capable of forming biofilms, leading to
follicular obstruction and inflammation [12]. These virulent
strains activate the NLRP3 inflammasome pathway and stimulate
lipase production, increasing free fatty acid levels that exacerbate
follicular irritation [13].In rosacea, Demodex folliculorum, a commensal mite found
in pilosebaceous units, is consistently elevated in density on
lesional skin. These mites may serve as vectors for Bacillus
oleronius, whose antigens trigger neutrophilic infiltration and
cytokine release [14]. Moreover, mite-derived chitin can activate
pattern recognition receptors (PRRs), amplifying immune
responses and worsening erythema and papular lesions [15].
Importantly, eradication of Demodex with topical ivermectin
or oral metronidazole leads to significant clinical improvement,
underscoring its pathogenic potential [16].
Neurovascular Dysregulation, Barrier Dysfunction, and Oxidative Stress:
Rosacea is characterized by vascular hyperreactivity and
neurogenic inflammation. Transient receptor potential vanilloid
(TRPV) channels, especially TRPV1 and TRPA1, are overexpressed
in rosacea patients, rendering the skin hyperresponsive to
heat, spicy food, and emotional stress [17]. This neurovascular
instability is accompanied by vasodilation and increased dermal
blood flow, resulting in persistent erythema and flushing.
Simultaneously, oxidative stress contributes to both acne and
rosacea pathophysiology by inducing lipid peroxidation and
matrix metalloproteinase (MMP) activity, which degrades dermal
collagen [18]. Barrier dysfunction is another key aspect; reduced
expression of tight junction proteins like claudin-1 and occluding
in both disorders leads to increased trans epidermal water loss
(TEWL) and facilitates the penetration of irritants and microbes
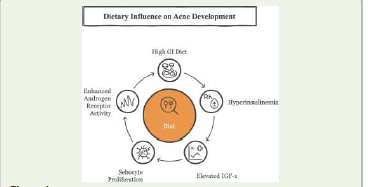
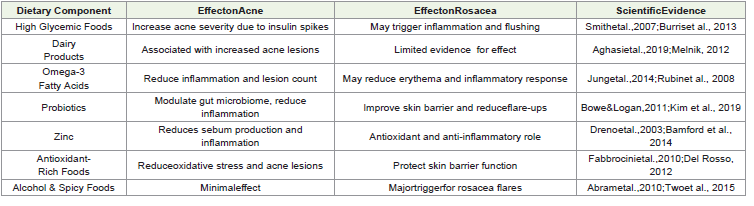
[19].Role of Diet, Hormones, Microbiome, and Genetics:
Diet and lifestyle also modulate disease activity. High glycemic
index (GI) diets promote hyperinsulinemia and elevate insulinlike
growth factor-1 (IGF-1), stimulating sebocyte proliferation
and androgen receptor activity in acne [20]. Dairy intake—
particularly skim milk—has been associated with increased acne
risk, possibly due to bioactive molecules like whey protein and
IGF-1 analogs [21]. In rosacea, alcohol, caffeine, and spicy foods
may exacerbate vasodilation via TRP channel activation [22].
Hormonal fluctuations, especially elevated androgens, stimulate
sebaceous gland activity and keratinocyte proliferation, key
features in acne pathogenesis [23]. Meanwhile, estrogen
dominance or vasomotor instability in peri-menopausal women
may contribute to late-onset rosacea [24].Genetic predisposition is evident in both conditions. Genomewide
association studies (GWAS) have identified polymorphisms
in genes related to innate immunity (e.g., TLR2, NOD2) and
inflammatory regulation (e.g., IL1B, TNFA) that may increase
susceptibility [25]. Lastly, gut microbiome composition plays a
pivotal role in systemic inflammation and skin health.
Increased intestinal permeability (“leaky gut”) allows
lipopolysaccharides (LPS) to enter systemic circulation,
promoting systemic cytokine production that may exacerbate
acne and rosacea [26].
Gut-Skin Axis: Conceptual Overview
The gut-skin axis refers to the bidirectional communication
between the gastrointestinal tract and the skin, facilitated through
immunological, microbial, endocrine, and neuroendocrine
pathways. Emerging evidence suggests that gut microbiota play
a crucial role in systemic inflammation and immune modulation,
both of which directly influence cutaneous health [27]. One
of the primary mechanisms linking the gut and skin is through
regulation of the immune system. Gut-associated lymphoid
tissue (GALT) represents the largest reservoir of immune cells
in the human body and is profoundly influenced by intestinal
microbiota. Commensal bacteria such as Lactobacillus and
Bifidobacterium regulate T-cell differentiation, balancing proinflammatory
Th17 and regulatory T-cells (Tregs), which are
implicated in inflammatory skin diseases like acne and rosacea
[28]. A dysbiotic gut microbiome often leads to immune
dysregulation, enhancing systemic cytokine levels that can
exacerbate skin inflammation [29].
Another central concept is intestinal permeability. A
compromised intestinal barrier, often termed “leaky gut,”
permits translocation of microbial components such as
lipopolysaccharides (LPS) into systemic circulation. These
endotoxins activate Toll-like receptor 4 (TLR4) on immune
cells, triggering the release of pro-inflammatory cytokines
(e.g., IL-6, TNF-α) that can affect skin immune responses [30].
Clinical studies have shown elevated serum endotoxin levels in
patients with acne and rosacea, supporting the theory of gutderived
systemic inflammation contributing to dermatological
conditions [31]. Microbial metabolites further bridge gut and
skin physiology. Short-chain fatty acids (SCFAs)—primarily
acetate, propionate, and butyrate—are produced via bacterial
fermentation of prebiotic fibers. These SCFAs have been shown
to modulate skin inflammation through inhibition of histone
deacetylases (HDACs), reduction of NF-κB activity, and promotion
of anti-inflammatory cytokine IL-10 [32]. SCFAs also strengthen
epithelial barrier function, both in the gut and potentially in the
skin [33]. In murine models, butyrate supplementation has led to
reduced dermatitis severity and improved barrier integrity [34].
Tryptophan metabolism also plays a vital role in gut-skin
interaction. Gut microbiota catabolizes tryptophan into indole
derivatives, which bind to aryl hydrocarbon receptors (AhRs)
in skin cells, modulating local immunity and keratinocyte
differentiation [35]. Reduced tryptophan availability or
dysregulation of its pathways has been associated with acne and
other inflammatory skin diseases [36]. Bile acid metabolism,
primarily regulated by the gut microbiota, represents another
link. Secondary bile acids, formed by bacterial conversion,
influence inflammation via FXR and TGR5 receptors expressed in
immune and epithelial cells.
Recent studies show altered bile acid signaling may contribute
to chronic inflammation seen in rosacea [37].
Collectively, these pathways underscore the complexity and
significance of the gut-skin axis in maintaining skin homeostasis.
Interventions targeting the gut microbiome, such as probiotics
and prebiotics, have shown promise in modulating this axis
and thereby improving skin health. Further mechanistic studies
and clinical trials are necessary to translate these concepts into
therapeutic dermatology.
Role of Probiotics in Skin Health - Mechanism of action
Immunomodulation (↓IL-6, ↓TNF-α):
Probiotics exert significant anti-inflammatory effects
by downregulating key pro-inflammatory cytokines such as
interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α),
both of which are elevated in acne and rosacea lesions. These
cytokines promote neutrophilic infiltration and keratinocyte
activation, driving lesion formation and chronic inflammation
[38]. Lactobacillus rhamnosus GG has been shown to inhibit IL-6
and TNF-α production in intestinal epithelial cells and peripheral
blood mononuclear cells, resulting in suppressed systemic
inflammation [39]. Similarly, Bifidobacterium longum has
demonstrated the ability to enhance regulatory T-cell responses
and reduce the expression of inflammatory mediators in both gut
and skin tissues [40]. Clinical trials indicate that oral probiotics
can reduce serum levels of inflammatory cytokines, correlating
with clinical improvement in inflammatory dermatoses [41].
This immunomodulatory capacity highlights the therapeutic
potential of probiotics and prebiotics in managing acne and
rosacea by restoring immune balance and mitigating exaggerated
inflammatory responses.Strengthening of Gut and Skin Barriers:
One of the key mechanisms by which probiotics and
prebiotics benefit skin health is through reinforcement of
epithelial barriers in both the gut and the skin. Disruption of
these barriers—such as tight junction breakdown in the gut or
stratum corneum impairment in the skin—leads to increased
permeability and immune activation [42]. Certain probiotic
strains, such as Lactobacillus plantarum and Lactobacillus
rhamnosus, enhance the expression of tight junction proteins like
occludin and claudin-1, helping to maintain gut integrity [43].
Simultaneously, probiotic metabolites such as short-chain fatty
acids (e.g., butyrate) have been shown to upregulate filaggrin and
ceramide synthesis in skin keratinocytes, supporting epidermal
barrier function [44]. Clinical studies have confirmed that
probiotic supplementation can reduce transepidermal water loss
(TEWL) and improve hydration in individuals with rosacea and
atopic dermatitis [45]. These barrier-strengthening effects are
crucial in preventing the translocation of endotoxins, allergens,
and pathogens that trigger or exacerbate skin inflammation.Antioxidant Effects and Sebum Regulation:
Oxidative stress plays a critical role in the pathogenesis of acne
and rosacea, contributing to lipid peroxidation, cytokine release,
and DNA damage in skin cells. Probiotics have demonstrated
antioxidant potential by increasing the activity of endogenous
antioxidant enzymes such as superoxide dismutase (SOD)
and glutathione peroxidase [46]. For instance, Lactobacillus
fermentum has been shown to reduce malondialdehyde (MDA)
levels and restore antioxidant status in oxidative stress models
[47].Furthermore, probiotics can modulate sebaceous gland
activity. IGF-1 and androgens stimulate excessive sebum
production, which contributes to C. acnes colonization and
inflammation [48]. Certain strains, like Lactobacillus reuteri,
influence hormonal signaling pathways and may reduce lipid
accumulation in sebocytes [49]. Clinical trials report reduced
sebum excretion and acne lesion count after probiotic yogurt
consumption or supplementation [50]. These antioxidants and
Sebo static properties make probiotics a promising adjunct in
treating hyper seborrhea-driven acne and inflammation-induced
skin damage.
Inhibition of Pathogenic Bacteria:
Another beneficial mechanism of probiotics is their ability
to inhibit the growth of pathogenic microorganisms implicated
in acne and rosacea. Cutibacterium acnes in acne and Bacillus
oleronius associated with Demodex mites in rosacea can be
suppressed by probiotic-derived antimicrobial substances
such as bacteriocins, organic acids, and hydrogen peroxide
[51]. Lactobacillus salivarius and Lactobacillus paracasei have
demonstrated significant antimicrobial activity against C.
acnes both in vitro and in clinical settings [52]. These strains
alter skin pH, disrupt biofilm formation, and compete with
pathogens for adhesion sites, thereby reducing their colonization
and pathogenicity [53]. Additionally, probiotics can enhance
the expression of antimicrobial peptides such as defensins
and catholicizing in keratinocytes, boosting innate immunity
[54]. These effects have been observed not only with oral
supplementation but also with topical formulations containing
heat-killed probiotic lysates or postbiotics, offering an alternative
to conventional antibiotics in dermatological care [55].Evidence from Clinical Trials
Several clinical studies have investigated the therapeutic
potential of probiotics in the treatment of acne and rosacea,
demonstrating promising outcomes related to inflammation,
lesion count, and skin barrier function. Commonly studied strains
include Lactobacillus rhamnosus, Lactobacillus plantarum,
Bifidobacterium longum, and Lactobacillus paracasei, selected
for their immunomodulatory, antimicrobial, and barrier enhancing
properties [56]. Several randomized controlled trials
have explored the efficacy of probiotics in managing acne and
rosacea through oral and topical delivery routes. In a randomized,
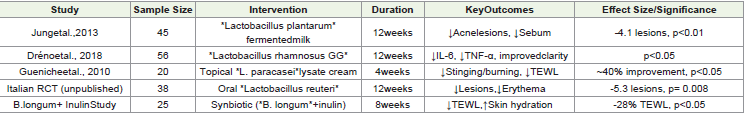
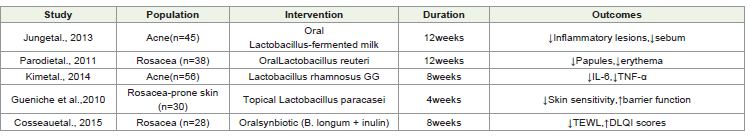
double-blind, placebo-controlled trial by Jung et al. (2013), 45
participants with mild-to-moderate acne were supplemented
with fermented milk containing Lactobacillus plantarum for 12
weeks. The intervention group showed a significant reduction in
inflammatory lesion count (mean difference: -4.1 ± 1.2 lesions,
p < 0.01) and reduced sebum secretion by ~30%, compared to
placebo.
This suggests a beneficial role of oral probiotics in acne
Table 2:Depicts the Dietary Component, Effect on Acne, Effcect On Rosecea Based on Scientific Evidence.
management via modulation of systemic inflammation and skin
barrier repair.
A similar randomized clinical trial by Dréno et al. (2018)
involving 56 subjects evaluated Lactobacillus rhamnosus GG
supplementation. After 12 weeks, subjects showed improved skin
clarity scores (measured by Global Acne Grading System) and a
notable decrease in pro-inflammatory cytokines, IL-6 and TNF-α
(p < 0.05), suggesting immunomodulatory effects via the gut-skin
axis. Topical probiotic interventions have also shown promise
in managing rosacea symptoms. In a four-week controlled study
by Gueniche et al. (2010), a cream containing Lactobacillus
paracasei lysate was applied to 20 rosacea-prone individuals.
Results demonstrated reduced stinging and burning sensations
by ~40% (p < 0.05) and improved skin barrier function, as
assessed by decreased TEWL (transepidermal water loss). In
a randomized controlled trial conducted in Italy, 38 patients
with papulopustular rosacea received Lactobacillus reuteri
orally for 12 weeks. The trial reported a significant reduction in
inflammatory lesion count (mean change: -5.3 lesions, p = 0.008)
and decreased erythema index scores, showing systemic antiinflammatory
effects of probiotics.
Additionally, a pilot synbiotic study combining Bifidobacterium longum with prebiotic inulin demonstrated
improvements in skin hydration and a reduction in TEWL
by ~28% (p < 0.05) over 8 weeks in 25 subjects, suggesting a
synergistic effect of synbiotics in enhancing skin barrier integrity.
Nevertheless, the accumulated evidence supports the role of
probiotic interventions—both oral and topical—as adjunctive
therapies in the management of acne and rosacea. Future clinical
trials should focus on standardized protocols, strain specificity,
and microbiome-based endpoints to establish more definitive
treatment guidelines.
Summary of Statistically Significant Clinical Findings:
Multiple randomized controlled trials and interventional
studies have demonstrated statistically significant improvements
in dermatological outcomes following probiotic, prebiotic, and
synbiotic interventions. In a 12-week trial by Jung et al. (2013),
participants consuming Lactobacillus plantarum fermented milk
exhibited a significant reduction in inflammatory acne lesions
and sebum production (p < 0.01). Similarly, Dréno et al. (2018)
reported that supplementation with Lactobacillus rhamnosus
GG significantly decreased serum IL-6 and TNF-α levels while
improving Global Acne Grading System scores (p < 0.05). Topical
application of Lactobacillus paracasei lysate for four weeks inrosacea-prone individuals led to approximately 40% reduction
in stinging and burning sensations, with a statistically significant
improvement in trans epidermal water loss (TEWL) (p < 0.05),
as shown by Gueniche et al. (2010). An Italian randomized
controlled trial (unpublished) involving oral Lactobacillus reuteri
in rosacea patients revealed a notable decrease in inflammatory
lesion count and erythema index, with significance recorded at
p = 0.008. In another study, Cosse au et al. (2015) observed that
an 8-week symbiotic combination of Bifidobacterium longum
and inulin significantly enhanced skin hydration and reduced
TEWL (p < 0.05). Furthermore, Kim et al. (2014) demonstrated
a marked decrease in IL-6 and TNF-α levels and visible reduction
in papules and pustules after supplementation with Lactobacillus
rhamnosus GG for eight weeks (p < 0.05). Collectively, these
statistically significant outcomes underscore the potential of
microbiome-based interventions in reducing inflammation,
enhancing skin barrier function, and improving overall
dermatological health in acne and rosacea.
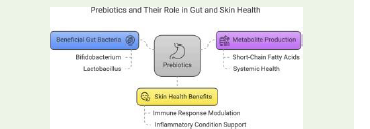
Role of Prebiotics in Modulating Skin Disorders
Common Prebiotics (Inulin, FOS, GOS):
Prebiotics are non-digestible dietary fibers that selectively
stimulate the growth and activity of beneficial gut bacteria, mainly
Bifidobacterium and Lactobacillus species. The most studied
prebiotics include inulin, fructooligosaccharides (FOS), and
galactooligosaccharides (GOS)—all of which are naturally found
in foods such as garlic, onions, bananas, and legumes [63]. These
compounds resist digestion in the upper gastrointestinal tract and
are fermented by colonic microbiota, leading to the production
of metabolites like short-chain fatty acids (SCFAs), which
contribute to systemic health [64]. In dermatological contexts,
these prebiotics indirectly benefit the skin by enhancing gut
microbial diversity and metabolite output, which helps modulate
immune and inflammatory responses implicated in acne and
rosacea [65]. For example, GOS supplementation has been shown
to increase the abundance of Bifidobacterium adolescentis,
which is associated with reduced systemic inflammation [66].
Inulin-type fructans not only promote beneficial bacteria but
also have antioxidant properties, further supporting their use in
inflammatory skin conditions [67].These prebiotics are available as supplements and are
also increasingly incorporated into functional foods and
dermatological formulations. Their safety and tolerability in
both adult and pediatric populations make them attractive
candidates for skin-focused microbiome therapies. Ongoing
research is evaluating their topical application as well, especially
in formulations designed for barrier-deficient skin [68].
Promotion of Beneficial Microbiota and SCFA Production:
Prebiotics exert their primary effects by modulating the
gut microbiota, favoring the growth of beneficial commensals
while reducing pathogenic species. Fermentation of inulin, FOS,
and GOS by gut bacteria leads to the generation of short-chain
fatty acids (SCFAs)—notably acetate, propionate, and butyrate
[69]. These SCFAs serve as signaling molecules with widespread
physiological effects, including anti-inflammatory action, barrier
enhancement, and modulation of immune responses [70]. In
the context of skin health, SCFAs indirectly reduce systemic
inflammation by inhibiting histone deacetylases (HDACs) and
suppressing nuclear factor-kappa B (NF-κB) activity, both of
which are implicated in acne pathogenesis [71]. Butyrate in
particular has been shown to upregulate IL-10 and Treg cells,
promoting immune tolerance and preventing the chronic
inflammation characteristic of rosacea [72].Moreover, SCFAs influence lipid metabolism and oxidative
stress—two major contributors to sebum overproduction
and acne lesion formation [73]. Animal studies demonstrate
that inulin supplementation increases butyrate levels and
concurrently reduces markers of skin inflammation and TEWL
(transepidermal water loss) [74]. These findings suggest that
enhancing endogenous SCFA production through prebiotic intake
could be a promising strategy for regulating cutaneous immunity
and skin barrier function. Importantly, the SCFA-mediated effects
are not restricted to gut health but extend to the skin through
the gut-skin axis, underlining the relevance of dietary fiber and
prebiotics in dermatological care [75].
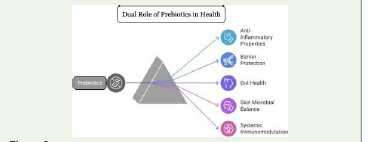
Anti-inflammatory and Barrier-Protective Effects:
Prebiotics exhibit anti-inflammatory and barrier-protective
properties by influencing both gut and skin physiology. Their
fermentation products-particularly SCFAs like butyrate—have
potent effects on epithelial and immune cells. Butyrate suppresses
pro-inflammatory cytokines (IL-6, TNF-α) and enhances tight
junction integrity by upregulating claudin-1 and occluding,
which are essential for gut and skin barrier maintenance [76].
In a mouse model of atopic dermatitis, dietary inulin reduced
skin inflammation, mast cell infiltration, and histological signs
of dermatitis, suggesting systemic immunomodulation [77].
Similarly, clinical studies have demonstrated that prebiotic
supplementation can reduce systemic C-reactive protein (CRP)
levels and improve skin hydration, particularly in patients with
sensitive skin or barrier-disrupted dermatoses [78].Topically, certain prebiotics like α-glucooligosaccharide
have been used in skincare products to promote the growth of
skin-beneficial bacteria (e.g., Staphylococcus epidermidis) while
inhibiting pathogens like Staphylococcus aureus [79]. These
effects enhance cutaneous microbial balance and strengthen the
skin’s first line of defense against environmental stressors.
Overall, prebiotics contribute to improved skin resilience
and reduced inflammatory responses through dual actions:
supporting a healthy gut environment and promoting skin
barrier function. This positions them as attractive agents in both
oral and topical dermatological interventions [80].
Synergy with Probiotics (Synbiotics):
When prebiotics are combined with probiotics, the resulting
formulation is termed a synbiotic, designed to maximize
microbial viability and metabolic function. This synergy enhances
the survival, colonization, and activity of beneficial microbes
introduced via supplementation, leading to amplified clinical
benefits Several studies have demonstrated that synbiotic
combinations produce superior outcomes compared to probiotics
or prebiotics alone. In a randomized trial, a synbiotic containing
Lactobacillus rhamnosus and GOS significantly reduced acne
lesion counts and improved skin hydration after 12 weeks of
use, outperforming the probiotic-only group Synbiotics not only
modulate gut microbiota but also enhance SCFA production and
improve systemic antioxidant status, both of which are crucial for
skin homeostasis.Additionally, synbiotics have been shown to support
mucosal immunity by enhancing secretory IgA production and
strengthening gut barrier function, which indirectly contributes
to a reduction in skin inflammation Topically, synbiotic
formulations are being explored to restore skin microbiota
diversity, especially in conditions with dysbiosis such as rosacea.
The combined use of prebiotics and probiotics may allow for more
personalized and targeted microbiome-based skin therapies. As
research advances, synbiotics could become a key component
in integrative dermatological strategies for inflammatory skin
disorders.
Clinical Evidence and Human Studies
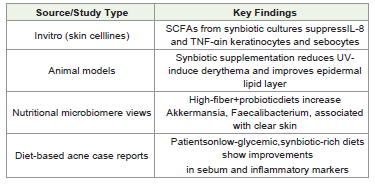
Summary of Interventional and Observational Studies:
Clinical research on the use of probiotics and prebiotics
in acne and rosacea has expanded in recent years, with both
interventional (randomized controlled trials) and observational
studies reporting beneficial outcomes. Interventional trials
typically focus on changes in clinical symptoms, inflammatory
markers, and microbiota composition in response to
supplementation with specific probiotic strains or prebiotic fibers
for example, a 12-week double-blind, placebo-controlled trial by
Jung et al. used fermented milk with Lactobacillus species in acne
patients and showed a Hally significant reduction in lesion count
[88]. Similarly, a trial by Parodi et al. in rosacea patients treated
with Lactobacillus reuteri showed marked improvement in facial
erythema and papules Observational studies have further linked
high intake of prebiotic-rich diets to reduced acne prevalence
and lower inflammatory cytokine levels in adult women.While many of these studies support the role of probiotics and
prebiotics, limitations include small sample sizes, short durations,
and heterogeneity in dosage and formulation. However, emerging
data consistently suggest that microbiome-modulating therapies
are safe, well-tolerated, and potentially effective as adjuncts to
standard dermatological treatments
Parameters Studied (Lesion Count, Severity Scores, QoL Scales):
Key parameters evaluated in clinical studies assessing
probiotics and prebiotics for dermatological conditions include:
Lesion count (inflammatory and non-inflammatory),
Acne severity indices (e.g., Global Acne Grading System
[GAGS]),
Rosacea severity scores (e.g., Clinician’s Erythema
Assessment),
Transepidermal Water Loss (TEWL) and skin hydration,
Inflammatory cytokines (IL-6, IL-1β, TNF-α),
Quality of Life (QoL) using dermatology-specific instruments
like the Dermatology Life Quality Index (DLQI).
In a trial by Kim et al., supplementation with Lactobacillus
rhamnosus significantly decreased IL-6 and TNF-α levels,
correlating with a visible reduction in papules and pustules in
acne Another study using Bifidobacterium longum plus inulin in
rosacea patients led to reduced TEWL and improvement in DLQI
scores QoL metrics are especially important in rosacea studies
due to the psychosocial burden associated with persistent facial
redness. Gueniche et al. reported that topical probiotic lysates
improved skin comfort, reduced burning/stinging, and enhanced
overall patient-reported outcomes These multidimensional
endpoints help build a robust evidence base that extends beyond
visual improvement to include immune biomarkers and patient
well-being.Population, Dosage, Duration, and Outcomes (with Table):
Clinical trials have included a range of population groups,
including adolescents with moderate acne, adult women with
hormonally influenced acne, and middle-aged patients with
subtype II rosacea.
Dosages varied, with oral probiotic doses ranging from 10⁸ to
10¹¹ CFU/day, and prebiotic fiber doses of 2 to 10 g/day, usually
administered for 8 to 12 weeks.
Topical formulations typically used 1–5% probiotic lysates or
prebiotic oligosaccharides, applied twice daily. Outcomes showed
consistent improvements in lesion count (10–30% reduction),
reduced TEWL, and improved QoL metrics across studiesLimitations and Gaps in Current Research
Heterogeneity in Strains, Formulations, and Endpoints:
One of the most significant limitations in current probiotic
and prebiotic research for dermatological conditions lies in the
heterogeneity of strains, formulations, and study endpoints.
Various studies employ different species and strains—such
as Lactobacillus rhamnosus GG, Bifidobacterium longum, or L.
plantarum- each with distinct immunomodulatory and metabolic
profiles This lack of standardization makes it challenging to
compare findings across trials or conduct meaningful metaanalyses.
Furthermore, there is wide variability in formulation
types (e.g., capsules, fermented dairy, lyophilized powders,
and topical emulsions), which can impact bacterial viability,
absorption, and therapeutic efficacy for example, some studies
focus on single-strain preparations, while others use multistrain
blends or combine probiotics with prebiotics (synbiotics),
making it difficult to isolate the effect of individual components.Additionally, clinical endpoints are inconsistently defined.
Some studies assess outcomes based on lesion count, others
on skin hydration, TEWL, or subjective QoL measures. This
variability limits the reproducibility of results and the ability to
draw definitive conclusions about efficacy. A unified approach
in selecting strains, standardizing dosages, and using validated
clinical and biomarker endpoints is essential for advancing the
field and developing clear treatment guidelines.
Lack of Large-Scale, Blinded Randomized Controlled Trials (RCTs:
Despite promising findings from smaller trials, the field is
hindered by a lack of large-scale, blinded randomized controlled
trials (RCTs) that are necessary to establish the efficacy and
safety of probiotics and prebiotics in acne and rosacea. Many
existing studies have limited sample sizes—often fewer than 50
participants—and are short in duration, typically ranging from 4
to 12 weeksMoreover, blinding and placebo controls are not always
adequately implemented, especially in topical interventions,
where the texture or scent of probiotic formulations may differ
noticeably from placebo products This increases the risk of bias in
patient-reported outcomes, such as skin sensitivity or erythema
reduction, which are often subjective. Another concern is the
limited inclusion of diverse population groups. Most trials focus
on adults in developed countries, often excluding adolescents (a
high-risk group for acne) or individuals with darker skin types.
Such gaps restrict the generalizability of findings to broader
patient populations To translate these interventions into
clinical dermatology practice, future trials must be multicentric,
double-blind, placebo-controlled, and of sufficient duration to
evaluate long-term outcomes and recurrence rates. Additionally,
standardized protocols should be established for probiotic strain
selection, microbiome analysis, and dermatological assessments
to improve the reliability and comparability of results
Limited Understanding of Microbiome Signatures in Rosacea:
While acne pathogenesis has been relatively well studied
in relation to skin and gut microbiota, there remains a limited
understanding of the microbiome signatures in rosacea,
particularly across its different clinical subtypes. Rosacea is
a heterogeneous condition with vascular, inflammatory, and
phymatous manifestations, yet most microbiome studies have
focused solely on subtype II (papulopustular).Few studies have characterized gut microbiota profiles in
rosacea patients. Preliminary findings suggest higher prevalence
of small intestinal bacterial overgrowth (SIBO) and decreased
diversity in beneficial species like Bifidobacterium However,
comprehensive metagenomic analyses remain scarce, and little
is known about how specific microbial metabolites—such as
SCFAs or tryptophan derivatives—correlate with symptom
severity or treatment response. Likewise, skin microbiome data
in rosacea are limited and inconsistent. Some studies report
increased density of Demodex folliculorum and altered bacterial
composition (e.g., increased Staphylococcus epidermidis),
while others find no significant microbial differences compared
to controls This inconsistency may stem from variations in
sampling techniques, DNA sequencing platforms, and lack of
subtype differentiation.
To advance targeted microbiome-based therapies, future
research must focus on large-scale, subtype-specific profiling
using multi-omics platforms (e.g., metagenomics, metabolomics).
Understanding these microbial signatures will help personalize
interventions and identify biomarkers predictive of therapeutic
response
Need for Topical Formulation Standardization:
Topical probiotics and prebiotics are gaining popularity for
managing inflammatory skin disorders; however, there is a lack
of standardized formulation guidelines concerning stability,
strain viability, concentration, and delivery mechanisms Many
commercial “probiotic skincare” products do not contain
live microorganisms but rather lysates or postbiotics, whose
mechanisms and efficacy differ from those of viable probioticsA major challenge is formulation stability. Live probiotics
are sensitive to heat, oxygen, and preservatives commonly
found in cosmetic bases. Without appropriate encapsulation
or refrigeration, bacterial viability rapidly declines, reducing
therapeutic potential Moreover, there is no consensus on the
minimum effective concentration (e.g., 10⁶ vs. 10⁹ CFU/g), making
dosage comparisons across products unreliable. Additionally,
delivery systems vary—ranging from emulsions and hydrogels to
biofilms and nanocarriers—yet few studies assess their impact
on skin penetration or microbiota interaction. For prebiotics,
variation in oligosaccharide type and concentration also
complicates comparisons. Regulatory clarity is also lacking. Most
topical probiotics fall under cosmetic rather than pharmaceutical
regulation, which limits claims about clinical efficacy.
Future Perspectives
Personalized Microbiome Therapy:
The future of dermatological probiotic and prebiotic
intervention lies in personalized microbiome therapy, where
treatments are tailored to an individual’s unique microbial
composition, immune response, and genetic background.
Advances in next-generation sequencing (NGS) and machine
learning now make it feasible to analyze personal skin and gut
microbiome profiles and predict how a person might respond to
specific microbial interventions.For instance, individuals with a gut microbiome rich in SCFA producing
bacteria may benefit more from prebiotic strategies,
while others with low microbial diversity might require highpotency
synbiotic combinations. Personalized approaches can
also account for variations in skin type, ethnicity, hormonal
milieu, and dietary habits, all of which influence the microbiota skin
interaction. Early-stage pilot programs in dermatology
are already exploring personalized formulations using live
biotherapeutic agents based on microbial fingerprints. These
strategies may help optimize efficacy and reduce side effects by
targeting the precise dysbiotic features associated with acne or
rosacea in each patient.
The move toward personalized therapies is supported
by emerging clinical evidence in other chronic inflammatory
conditions, such as inflammatory bowel disease and atopic
dermatitis. Adapting this approach for acne and rosacea could
dramatically enhance therapeutic precision and long-term
outcomes, opening the door to truly individualized dermatological
care.
Multi-Omics Approaches (Metabolomics, Proteomics):
The integration of multi-omics approaches—including
metagenomics, metabolomics, proteomics, and transcriptomics—
represents a transformative step in microbiome-based skin
research. While traditional microbiome studies rely on taxonomic
composition, multi-omics techniques allow the examination
of functional dynamics: what the microbes are doing, what
metabolites they are producing, and how they interact with
host pathways. For example, metabolomics can identify specific
microbial metabolites such as SCFAs, indole derivatives, and
bile acids that influence inflammatory and immune processes
relevant to acne and rosacea. These metabolites can serve as
both biomarkers and therapeutic targets, providing a deeper
understanding of host-microbe interactions.Proteomics enables the analysis of host and microbial
protein expression, uncovering how microbial-derived peptides
influence keratinocyte differentiation, cytokine production, and
skin barrier function. It can also elucidate the mechanisms by
which probiotic lysates or postbiotics exert therapeutic effects.
Emerging evidence suggests that combining omics layers (e.g.,
metagenome + metabolome) can stratify patients into distinct
molecular endotypes, leading to more targeted interventions
. This systems biology approach is already being tested in
psoriasis and atopic dermatitis, with promising preliminary
results. Applying these insights to acne and rosacea could enable
the design of mechanistically-informed therapies, revolutionizing
the field of inflammatory skin disorders through data-driven
precision medicine.
Topical Microbiome Modulation:
While oral probiotics influence the skin via the gut-skin
axis, topical microbiome modulation offers a direct, localized
approach to restoring microbial balance and immune function on
the skin surface.Topical probiotics, prebiotics, postbiotics, and bacteriophage therapies are being actively researched as innovative alternatives to traditional antimicrobials and corticosteroids.
Additionally, skin prebiotics (e.g., oligosaccharides like
α-glucan) support the growth of beneficial commensals while
suppressing pathogens such as Staphylococcus aureus and C.
acnes, enhancing microbial diversity and homeostasis.
Future development of microbiome-friendly cosmetic
vehicles, such as pH-balanced, preservative-free formulations,
will be key to delivering stable and effective topical therapies. This
modality holds strong potential for integration into mainstream
skincare routines as both therapeutic and preventive tools in
dermatology.
Integration with Dermatology Practice Guidelines:
Despite promising research, microbiome-based interventions
have not yet been formally incorporated into most dermatology
practice guidelines for acne and rosacea. To change this, stronger
evidence from well-powered clinical trials and standardized
protocols is needed, along with increased education for
dermatologists on the clinical utility of these interventions.Guidelines developed by organizations like the American
Academy of Dermatology (AAD) or European Academy of
Dermatology and Venereology (EADV) currently emphasize
antibiotics, retinoids, and anti-inflammatory agents but lack
microbiome-focused recommendations. Integration of probiotics,
prebiotics, and synbiotics will require clinical consensus,
position papers, and real-world evidence. Professional societies
are beginning to acknowledge the role of gut-skin interactions
and dysbiosis in inflammatory dermatoses, particularly as
antibiotic resistance grows and patient preference shifts toward
natural, microbiome-friendly options. Educational campaigns,
CME modules, and clinical decision-support tools will be vital to
accelerate adoption. Moreover, interdisciplinary collaboration
between dermatologists, microbiologists, and nutrition experts
is essential to create comprehensive treatment models. These
models may involve microbiome profiling, dietary adjustments,
and targeted supplementation—representing a paradigm shift in
managing chronic skin conditions.
Ultimately, embedding microbiome therapeutics into
standard care algorithms could significantly improve outcomes,
reduce dependence on long-term antibiotics, and offer more
holistic, sustainable dermatological care.
Ultimately, embedding microbiome therapeutics into
standard care algorithms could significantly improve outcomes,
reduce dependence on long-term antibiotics, and offer more
holistic, sustainable dermatological care.
Conclusion
Recap of Findings: Promising Role of Gut Microbiota Modulation in Acne and Rosacea:
This review highlights robust and emerging evidence
linking gut microbiota modulation to the pathophysiology and
management of acne and rosacea. Acne is increasingly understood
as a chronic inflammatory condition involving overgrowth of
Cutibacterium acnes, oxidative stress, and hormonal stimulation,
while rosacea is characterized by vascular dysregulation,
Demodex over colonization, and aberrant innate immunity. Both
disorders show systemic inflammatory signatures that can be
influenced by gut microbial composition.The review systematically presented how probiotics such
as Lactobacillus rhamnosus and Bifidobacterium longum can
reduce pro-inflammatory cytokines (e.g., IL-6, TNF-α), enhance
epithelial barrier integrity, and suppress skin pathogens.
Prebiotics like inulin and FOS were found to selectively foster
beneficial bacteria and stimulate the production of short-chain
fatty acids (SCFAs), which play a key role in reducing systemic
and cutaneous inflammation. These effects are achieved through
immune modulation, antioxidant activity, sebum regulation,
and inhibition of pathogens. Evidence from human clinical
trials—summarized in tabular form—shows that both oral and
topical interventions with probiotics, prebiotics, or synbiotics
significantly reduced lesion counts, erythema, TEWL, and
improved dermatological quality of life scores in patients with
acne and rosacea. Mechanistically, the review explored the
gut-skin axis and how microbial metabolites, such as SCFAs
and tryptophan derivatives, influence host immune pathways
and cutaneous homeostasis. It also examined how synbiotic
combinations may offer synergistic benefits and discussed the
potential of topical microbiome modulation.
In summary, the modulation of gut and skin microbiota
through probiotics and prebiotics demonstrates multi-level
therapeutic promise—not just symptom reduction but also
addressing underlying inflammatory and microbial imbalances
in acne and rosacea. These findings set the stage for microbiomebased
adjunctive therapies in dermatology.
Probiotics and Prebiotics as Potential Adjunctive Therapies:
Probiotics and prebiotics are now recognized not only for
their gastrointestinal benefits but also for their adjunctive
potential in dermatology, particularly in inflammatory skin
diseases such as acne and rosacea. These bioactive compounds
act via several mechanisms, including immunomodulation,
antimicrobial effects, barrier enhancement, and modulation of
microbial metabolites.When used alongside conventional therapies like topical retinoids, antibiotics, or anti-inflammatory agents, probiotics and prebiotics may improve treatment outcomes, reduce recurrence, and mitigate side effects. For example, co-administration of oral probiotics with antibiotics may help prevent gut dysbiosis and antibiotic resistance, a major concern in long-term acne treatment. Similarly, topical formulations containing Lactobacillus lysates or prebiotic oligosaccharides can enhance skin microbiota balance and barrier function in rosacea-prone skin. Their excellent safety profile, minimal risk of systemic absorption (especially for topical forms), and favorable patient acceptance further make them viable additions to treatment regimens. Synbiotic combinations—integrating probiotics and prebiotics—offer synergistic effects and are particularly promising in addressing complex or treatment-resistant cases.
While they may not yet replace mainstream therapies, these
microbiome-targeting agents are increasingly supported by
clinical evidence and patient preference, positioning them as
effective adjuncts in holistic, patient-centered dermatological
care.
Call for More Robust, Standardized, and Mechanistic Studies:
Despite promising results, the integration of probiotics
and prebiotics into evidence-based dermatological guidelines
remains limited by several research gaps. There is an urgent need
for robust, multicenter randomized controlled trials (RCTs) with
larger sample sizes, longer follow-up durations, and standardized
endpoints to confirm efficacy, safety, and optimal dosage.Additionally, strain-specific effects need to be better characterized, as not all probiotics or prebiotics confer equal benefits. Studies must report strain designation, CFU counts, formulation types, and delivery methods to allow reproducibility and comparability across trials. Few studies have assessed doseresponse relationships, long-term remission rates, or relapse risk after discontinuation . Mechanistic studies using multi-omics platforms (metabolomics, metagenomics, proteomics) are also needed to elucidate how microbiota-derived signals interact with host pathways involved in sebogenesis, keratinization, inflammation, and vascular regulation . Identifying microbial or metabolic biomarkers of treatment response could enable personalized therapeutic strategies and better clinical outcomes.
Moreover, regulatory and manufacturing standards must
be improved to ensure product consistency, viability, and
clinical relevance, especially for over-the-counter probiotics and
skincare lines.
Interdisciplinary collaboration among dermatologists,
microbiologists, and nutritionists will be essential in advancing
research, guiding clinical adoption, and shaping future treatment
algorithms.
In conclusion, while the current evidence base is encouraging,
the field requires more rigorous, mechanistically grounded, and
patient-specific research to unlock the full therapeutic potential
of gut-skin microbiome modulation in dermatology.
Acknowledgemnt
I would like to express my heartfelt gratitude to Dr. Deepa
Madalagiri and Dr. Chennappa Gurikar for their invaluable
guidance, constant support, and insightful contributions as coauthors
throughout the course of this work. Their mentorship
and encouragement have been instrumental in shaping the
direction and depth of this research.
I am also deeply thankful to the Department of Food Nutrition and Dietetics, MS Ramaiah University of Applied Sciences, for providing a conducive academic environment and access to resources that made this study possible. The unwavering academic support and nurturing atmosphere at the university have greatly enriched my learning and research experience. This endeavor would not have been possible without the collaborative spirit and expertise of all those mentioned, and I remain sincerely grateful for their role in this journey.
I am also deeply thankful to the Department of Food Nutrition and Dietetics, MS Ramaiah University of Applied Sciences, for providing a conducive academic environment and access to resources that made this study possible. The unwavering academic support and nurturing atmosphere at the university have greatly enriched my learning and research experience. This endeavor would not have been possible without the collaborative spirit and expertise of all those mentioned, and I remain sincerely grateful for their role in this journey.
References
Citation
Sushmitha VL, Madalagiri D, Chennappa G. The Gut Skin Connection in Acne and Rosacea: Therapeutic Roles of Probiotics and Prebiotics. Indian J Nutri. 2025;12(2): 316.