Case Report
Ruptured Slpenic Artery Aneurysm-Unusual and Interesting Imaging Findings
Sridevi Chinta1, Sujata Patnaik2*, Nava Kishore K3, Prasanth B4 and Shantiveer GU5
11Junior Resident, Dept of Radiology, NIMS, Hyderabad
21Professor of Radiology, NIMS, Hyderabad
31Assistant Professor of Surgical Gastroenterology, NIMS, Hyderabad
4Senior Resident, Dept of Surgical Gastroenterology, NIMS, Hyderabad
5Professor of Pathology, NIMS, Hyderabad
*Corresponding author: Dr. Sujata Patnaik, Professor of Radiology, NIMS, Hyderabad. 500082 Telangana, India,
Phone-9490793534 E-mail: sujata_patnaik222@yahoo.co.in
Copyright: © 2023 Chinta S, et al. This is an open access article distributed under the Creative Commons Attribution
License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is
properly cited.
Article Information: Submission: 17/12/2022; Accepted: 23/02/2023; Published: 28/02/2023
Abstract
Splenic artery aneurysms (SAA) are rare and can be fatal if they rupture. They present with abdominal pain, melena, distension and tenderness of
abdomen. We are reporting this case, since rupture of SAA is rare occurrence and there are a few unusual imaging findings observed. MDCT played a vital
role in diagnosing the rupture, facilitating the treatment. Presence of gas within the infarct is not always due to infection, it may be non-suppurative as in our
case.
Introduction
Splenic artery aneurysms (SAA) are rare and can be fatal if
they rupture. Precise etiology of true aneurysm of splenic artery is
unknown. It is associated with hypertension, portal hypertension,
cirrhosis, liver transplant, pregnancy, arteritis, collagen vascular
disease, Alfa anti-trypsin deficiency or inflammatory/infectious
diseases, pancreatitis. Splenic artery pseudo-aneurysm (SPA) is less
prevalent than true aneurysm and occurs in less than 1% in general
population [1]. In true aneurysm there is dilation of splenic artery
with all layers intact. Pseudo aneurysm occurs following disruption
of one or more layers of vessel wall. Underlying causes of splenic
artery aneurysm may be trauma, infection or weakness of vessel wall
from exposer to pancreatic enzymes. Due to widespread use of crosssectional
imaging, there is an increased detection rate of splenic artery
aneurysms. The true aneurysm often shows atheromatous plaque/
calcification. Pseudo-aneurysm is usually multilocular contour on
imaging. However, on many occasions, it is difficult to differentiate
true from pseudoaneurysm on clinical and imaging grounds.
Hemorrhage and abdominal pain are the most common presenting
symptoms [2]. Secondary hemorrhage may involve pancreatic duct,
peritoneum, retroperitoneum, adjacent organs or into pseudocyst if
present. The chance for rupture of SPA is higher than SAA due to lack
of all the three layers of the vessel wall. We are reporting this case,
since rupture of SAA is rare occurrence and there are a few unusual
imaging findings observed in our case.
Case Details
40 years male who was a chronic alcoholic presented with
abdominal pain and shortness of breath for 2 days. Patient had one
episode of melena 2 days back. No history of constipation, vomiting or
hematemesis. No other comorbidities were present. On examination there was abdominal distension and diffuse tenderness all over the
abdomen. There was no guarding or rigidity. Patient was dehydrated.
Pallor was present. SPO2 was 100% on room air. Respiratory rate:
28 breaths/min. Pulse rate: 126 bpm. BP: 100/60 mmHg. GRBS mg/
dl. GCS: E4V5M6. On blood investigations, hemoglobin was low
(8.3gm/dl), PCV-25%, TLC-22,700/ul, urea- 45mg/dl, creatinine-
1.2mg/dl, Na-132, K- 4.1 and Cl-92 mmol/l. The 2DEcho showed
structurally normal heart with good LV function.
Ultrasonography of abdomen revealed hepato-splenomegaly.
There was peri-splenic heterogeneously hypoechoic collection with
evidence of internal echoes. Splenic artery showed out-pouching
ofsize-1.2x1.1 cms close to the hilum. Ying-yang sign was observed
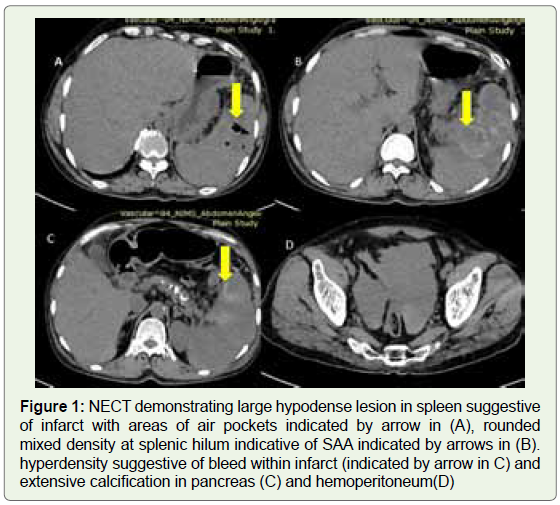
on color Doppler examination. On CT scan of abdomen there was a
large hypodense lesion in spleen with areas of hyperdensity suggestive
of bleed and large infarct (Figure 1). A few air pockets were observed
in the infarcted parenchyma. A rounded area of central hypo and
peripheral hyperdensity was noted within splenic parenchyma close
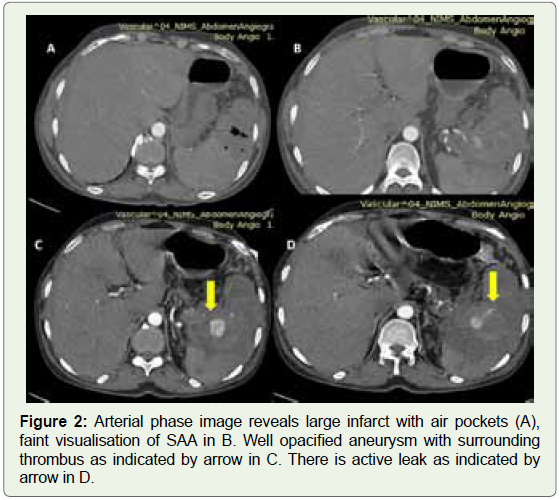
to the hilum. In arterial phase there is contrast pooling in the central
part of hypodense lesion close to splenic hilum which was arising
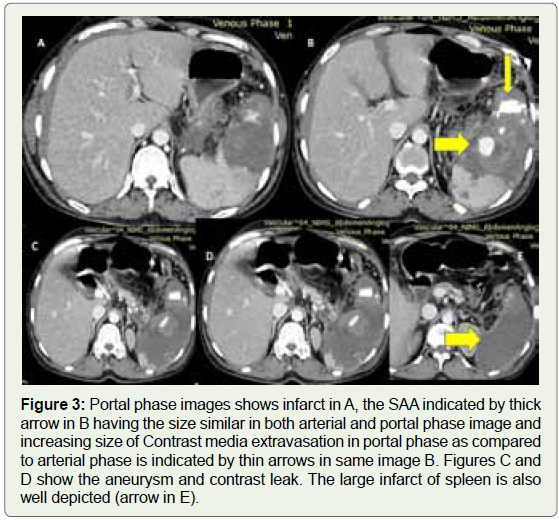
from splenic artery (Figure 2). On portal phase there was increased
opacification of central enhancing area with active contrast leak into
surrounding splenic parenchyma within the areas of bleed suggestive
of active bleed from ruptured pseudo-aneurysm (Figure 3). There
was moderate hemoperitoneum. Pancreas was atrophic and showed
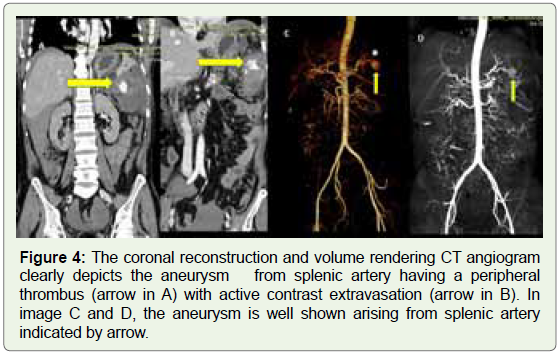
parenchymal calcification suggestive of chronic pancreatitis. (Figure 4) clearly demonstrated the SAA and contrast media extravasation
(CME) was noted in volume rendering image.
Figure 1: NECT demonstrating large hypodense lesion in spleen suggestive
of infarct with areas of air pockets indicated by arrow in (A), rounded
mixed density at splenic hilum indicative of SAA indicated by arrows in (B).
hyperdensity suggestive of bleed within infarct (indicated by arrow in C) and
extensive calcification in pancreas (C) and hemoperitoneum(D)
Figure 2: Arterial phase image reveals large infarct with air pockets (A),
faint visualisation of SAA in B. Well opacified aneurysm with surrounding
thrombus as indicated by arrow in C. There is active leak as indicated by
arrow in D.
Figure 3: Portal phase images shows infarct in A, the SAA indicated by thick
arrow in B having the size similar in both arterial and portal phase image and
increasing size of Contrast media extravasation in portal phase as compared
to arterial phase is indicated by thin arrows in same image B. Figures C and
D show the aneurysm and contrast leak. The large infarct of spleen is also
well depicted (arrow in E).
Figure 4: The coronal reconstruction and volume rendering CT angiogram
clearly depicts the aneurysm from splenic artery having a peripheral
thrombus (arrow in A) with active contrast extravasation (arrow in B). In
image C and D, the aneurysm is well shown arising from splenic artery
indicated by arrow.
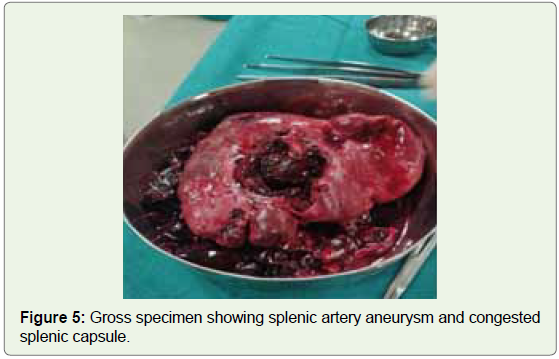
On gross examination, resected the spleen was 14x12x5 cm and
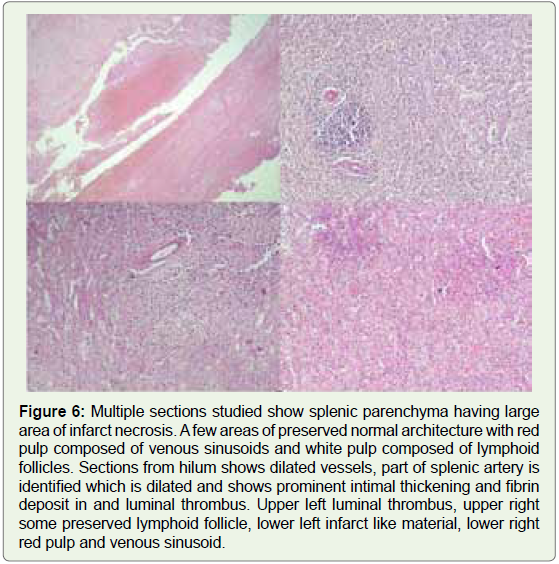
weight was 330gm. The capsule was congested. Microscopic section
revealed a large area of infarct necrosis. A few areas of normal
architecture were seen. At hilum there were dilated vessels. A localized
part of splenic artery was dilated with prominent intimal thickening,
fibrin deposition and luminal thrombus. The final diagnosis was
splenic artery true aneurysm with thrombosis (Figure 5,6). In the
peritoneum, a large blood clot was noted close to the splenic hilum.
There was adhesion of omentum with the pancreatic tail.
Figure 6: Multiple sections studied show splenic parenchyma having large
area of infarct necrosis. A few areas of preserved normal architecture with red
pulp composed of venous sinusoids and white pulp composed of lymphoid
follicles. Sections from hilum shows dilated vessels, part of splenic artery is
identified which is dilated and shows prominent intimal thickening and fibrin
deposit in and luminal thrombus. Upper left luminal thrombus, upper right
some preserved lymphoid follicle, lower left infarct like material, lower right
red pulp and venous sinusoid.
Discussion
Splenic artery aneurysms are infrequently encountered and critical
to recognize. Pathologically, the aneurysms are subdivided into true
aneurysms (77%) and pseudo-aneurysms (13%) [3]. However, the
term splenic artery aneurysm (SAA) is used interchangeably. The true
aneurysm (SAA) is nothing but localized widening of splenic artery.
Splenic pseudo-aneurysm (SPA) is different from true aneurysm
histologically in that a collection of blood forms between the tunica
media and tunica adventitia rather than circumferential dilatation
of the vessel. Wall weakening in SAA occurs through traumatic,
inflammatory, infective or iatrogenic causes [1]. Aneurysm is
common in splenic artery due to proximity of artery to pancreas.
Other arteries that can be involved are gastroduodenal, pancreaticoduodenal,
hepatic and left gastric [4]. In case of pancreatitis,
pancreatic enzymes cause a necrotizing arteritis with destruction of
vessel wall structures and fragmentation of elastic tissue leading to
aneurysm/pseudoaneurysm [1]. Though repeated acute exacebations of inflammation in the background of chronic pancreatitis is a
possible mechanism for SAA formation and rupture, it is difficult
to say for sure that our patient had such repeated acute episodes of
pancreatitis in absence of suggestive history.
Fistulous:
Communication to adjacent organs in background of pancreatitis
is also a dreaded complication. GIT bleed and hemoperitoneum
should prompt the suspicion of rupture as in our patient. SAA
has potential risk of rupture in 10% with mortality rate of 10-25%
in non- pregnant patients and up to 70% during pregnancy [5].
When aneurysm is > 2 cm, the chance of rupture is higher [6].
Portal hypertension, liver transplantation and pregnancy increase
the chances of rupture in SAA [7]. Mortality is high (90% to 100%)
with bleeding into peritoneal cavity or retroperitoneal cavity. With
aggressive treatment the mortality is 18-29% [8].Prompt reorganization of hemorrhagic shock, aggressive
hemodynamic stabilization, an focused diagnostic approach and
specific treatment are the key to the management of ruptured
SAA. Our patient was a chronic alcoholic with chronic pancreatitis
presented with sudden rupture of SAA. MDCT played a vital role
in early and accurate diagnosis and immediate surgery saved our
patient. The images of our patient were analysed and following
issues were discussed. CT is useful for demonstration of CME. Since
it is not a dynamic scan it cannot demonstrate the degree, rate and
quantity of active bleed. Arterial CME appear as high attenuation
similar to adjacent artery. CT can differentiate contained aneurysm
from ruptured aneurysm. Intra-parenchymal pseudo-aneurysms
have similar appearance to active hemorrhage on initial scan and
do not increase in size on delayed phases and follow the blood
pools on all phases. In contrast, active hemorrhage appears as high
density due to contrast leak which increases in size. This point is
well demonstrated in figures 2 and 3, and CME is arterial. Venous
CME demonstrates extravasation only in late equilibrium phase and
is less defined than arterial contrast extravasation. CT cannot detect
intermittent extravasation and is not useful in clinically stable patient.
Radionuclide scans have advantage of detecting slower bleeds and in
stable patients.
Infarcts are wedge shaped, hypodense situated peripherally
Splenic infarct may show liquefactive necrosis and intra-parenchymal
gas formation. Air-pocket may suggest either abscess formation
or they may be due to liberation of oxygen from Oxyhemoglobin
in splenic infarct [9]. Rankin believes that amount of gas in infarct
depends on amount of infarct and vascularity of tissue [10]. Splenic
infarct with gas formation is also described by Levy JM et al in their
case report [11]. Multiple bubbles of gas present in infarct throughout
the parenchyma with central distribution. These air pockets are
usually nonsuppurative. Splenic abscess is fatal outcome of splenic
infarct. Differentiation between the infarct with pockets of gas from
splenic abscess is important as splenic abscess needs intervention
and infarcts are managed conservatively. Abscess appears as round
/oval hypodense lesion containing gas and has enhancing capsule.
Nonsuppurative air pockets are noted in our case and microscopic
examination of operated specimen revealed infarct necrosis in spleen
and there was no evidence of abscess.
Another explanation of air pockets in our case may be due to SAA
eroding adjacent colon Causing melena and air pockets in splenic
parenchyma. On laparotomy there was adhesion of pancreatic tail
and omentum. However, there was no communication of colon with
splenic parenchyma. Hence the possibility of fistulous communication
is less likely. When the infarct matures it undergo three processescomplete
resolution or contraction/scarring or liquefaction. Portal
phase is best for detecting infarct. SAA with low risk of rupture may
be followed up 6monthly. When there is suspicion of increasing size
more than 2cms in diameter or patients having symptoms or if patient
is pregnant, SAA should be treated aggressively. All false aneurysms
of splenic artery should be treated as soon as diagnosed, irrespective
of size, symptoms or rupture [12]. Endovascular treatment with either
embolization or stent graft applications are treatment of choice for
SAA. However surgical intervention is considered in case of rupture
as done in our patient.
Conclusion
SAA /SPA though rare, can rupture and present with abdominal
pain, melena, distension and tenderness of abdomen. Prompt
reorganization of hemorrhagic shock, aggressive hemodynamic
stabilization, focused diagnostic approach and specific treatment are
the key to save the patient. MDCT played a vital role in diagnosing
the rupture, facilitating the treatment in our patient. Though we
thought the aneurysm to be pseudo-aneurysm on imaging findings,
pathological examination confirmed it as a true aneurysm. Presence
of gas within the infarct is not always due to infection, it may be nonsuppurative
as in our case.
References
Citation
Chinta S, Patnaik S, Nava Kishore K, Prasanth B, Shantiveer GU. Ruptured Slpenic Artery Aneurysm-Unusual and Interesting Imaging Findings. Indian J Appl Radiol. 2023;9(1): 177.