Pictorial Essay
MDCT Evaluation of Congenital Coronary Anomalies: Pictorial Essay
Kumar K*, Viswanathan M, Dharan Venkatesh KA, Senthinathan V and Nasreen I
Department of Radio-diagnosis, Trichy SRM Medical college hospital & Research Centre, Irungalur, Trichy-621105, India
*Corresponding author: Krishna Kumar, Department of Radio-diagnosis, Trichy SRM Medical college hospital & Research
Centre, Irungalur, Trichy-621105, India; E-mail: drmkrishnakumar@gmail.com; Mobile: +91 9447345528
Copyright: © 2022 Kumar K, et al. This is an open access article distributed under the Creative Commons Attribution
License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is
properly cited.
Article Information: Submission: 09/09/2022; Accepted: 27/10/2022; Published: 31/10/2022
Abstract
Objective: The purpose of this pictorial essay is to review the multi detector computed tomography (MDCT) coronary angiography appearance of
congenital coronary anomalies [CCA]. CCA might also be classified as hemodynamically significant or insignificant. The clinical symptoms may include chest
pain, dyspnoea, palpitations, syncope, cardiomyopathy, arrhythmia, infarction and sudden cardiac death. Although CCA are relatively uncommon, they’re
the second most typical reason for sudden cardiac death among young athletes and so warrant detailed review. Familiarity with atypical anatomy and their
clinical presentation may facilitate appropriate diagnosis and management. This will be of immense help to the clinician planning interventional procedures
like stenting, balloon dilatation, or graft surgery particularly when there are secondary changes of calcification, plaque formation and stenosis.
Conclusion: Increasing use of MDCT for cardiac imaging has helped within the detection of the many benign congenital coronary anomalies (CCA), but
a little number is related to myocardial ischemia and sudden death. Increasing the employment of MDCT in cardiac imaging may yield diagnostic information
on CCA not obtained with invasive coronary angiography. Axial sections, multiplanar reconstructions, virtual angioscopy, and 3D volume-rendered images
should aid within the detection and improve the interpretation of such anomalies, which might be of immense help to the clinician planning interventional
procedures.
Keywords
Computed tomography; Coronary angiography; Congenital coronary anomalies; Malignant coronary artery
Introduction
Congenital coronary anomalies (CCA) are uncommon and most
of them are diagnosed incidentally during conventional coronary
angiography or MDCT angiography. In step with the literature,
CCAs affect around 1% of the general population, starting from 0.3%-
5.6% in studies on patients undergoing coronary angiography, and
in approximately 1% of routine autopsy [1]. Based on the functional
relevance of every abnormality, coronary artery anomalies may be
classified as anomalies with obligatory ischemia, without ischemia or
with exceptional ischemia. The clinical symptoms may include chest
pain, dyspnea, palpitations, syncope, cardiomyopathy, arrhythmia,
myocardial infarct and sudden cardiac death. Although congenital
coronary artery anomalies are relatively uncommon, they’re the
second most typical reason behind sudden cardiac death (SCD)
among young athletes. The chance of SCD in time of life or in elderly
individual with an incidentally discovered coronary anomaly is
unclear but is perhaps negligible. The anomaly is most often related to
SCD is the anomalous origin of a coronary artery, particularly
with a course between the aorta and the PA [1]. For several decades,
premorbid diagnosis of CCA has been made with conventional
angiography. Although catheter angiography is an efficient tool,
it’s invasive and related to procedural morbidity (1.5%) and
mortality (0.15%) [2]. Because of its two dimensional nature, catheter
angiography has projectional limitations and it cannot show the link
of aberrant vessels with the underlying cardiac structures [3]. Recent
development of ECG-gated MDCT coronary angiography allows
accurate and noninvasive depiction of coronary artery anomalies of origin, course, and termination. CT coronary angiography (CTCA)
is superior to standard catheter angiography in delineating the ostial
origin and proximal path of an anomalous coronary artery [4].
section3
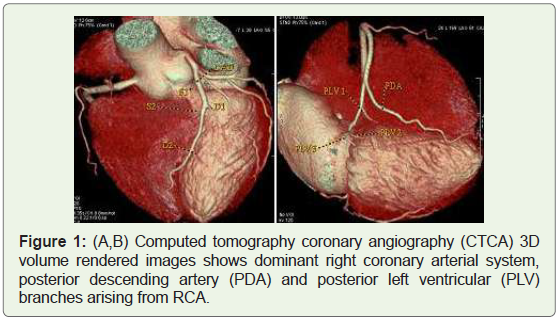
Case 1: 56 years old male with dominant Right coronary arterial
(RCA) system (Figure 1).
Figure 1: (A,B) Computed tomography coronary angiography (CTCA) 3D
volume rendered images shows dominant right coronary arterial system,
posterior descending artery (PDA) and posterior left ventricular (PLV)
branches arising from RCA.
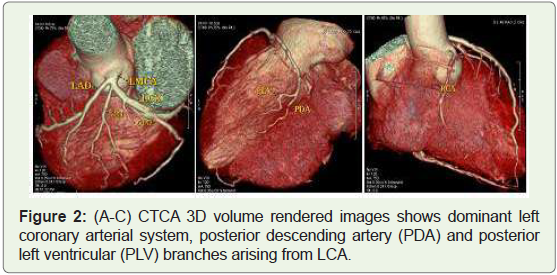
Case 2: 49 year old male with dominant Left coronary arterial
(LCA) system (Figure 2).
Figure 2: (A-C) CTCA 3D volume rendered images shows dominant left
coronary arterial system, posterior descending artery (PDA) and posterior
left ventricular (PLV) branches arising from LCA.
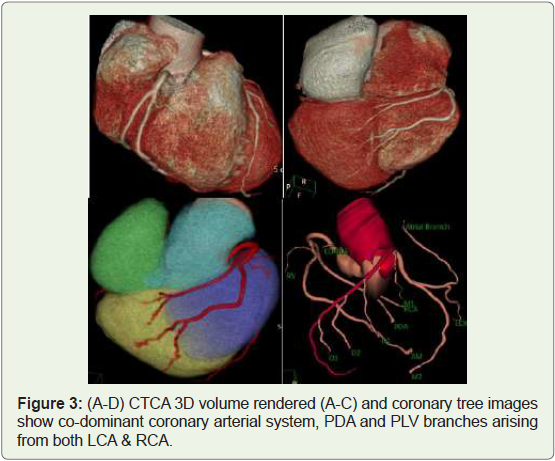
Case 3: 36 year old male with Co-dominant coronary arterial
system (Figure 3).
Figure 3: (A-D) CTCA 3D volume rendered (A-C) and coronary tree images
show co-dominant coronary arterial system, PDA and PLV branches arising
from both LCA & RCA.
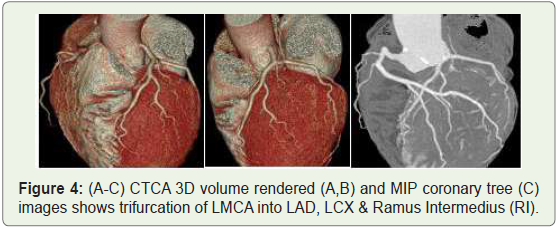
Case 4: 38 year old male with trifurcation of LMCA into LAD,
LCX & Ramus intermedius (Figure 4).
Figure 4: (A-C) CTCA 3D volume rendered (A,B) and MIP coronary tree (C)
images shows trifurcation of LMCA into LAD, LCX & Ramus Intermedius (RI).
Anomalies of origin; A. Number of Ostia: single, Multiple (>2):
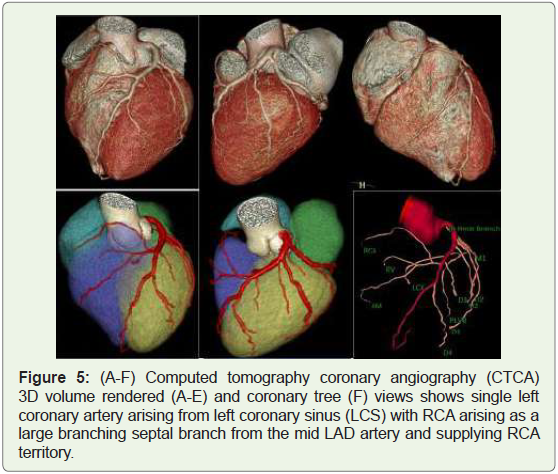
Case 5: Single Left coronary artery in a 39 year-old man (Figure 5).
Figure 5: (A-F) Computed tomography coronary angiography (CTCA)
3D volume rendered (A-E) and coronary tree (F) views shows single left
coronary artery arising from left coronary sinus (LCS) with RCA arising as a
large branching septal branch from the mid LAD artery and supplying RCA
territory.
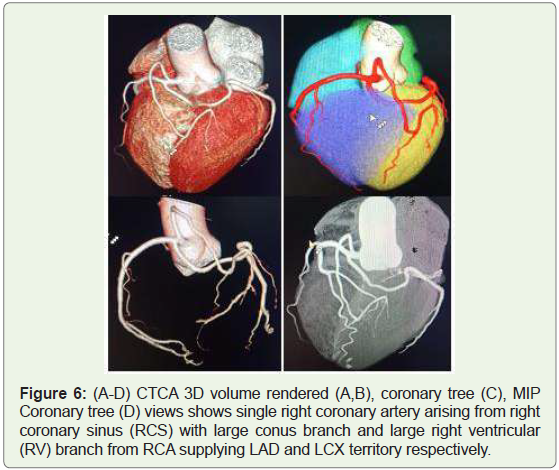
Case 6: Single Right coronary artery in a 46 year old man (Figure 6).
Figure 6: (A-D) CTCA 3D volume rendered (A,B), coronary tree (C), MIP
Coronary tree (D) views shows single right coronary artery arising from right
coronary sinus (RCS) with large conus branch and large right ventricular
(RV) branch from RCA supplying LAD and LCX territory respectively.
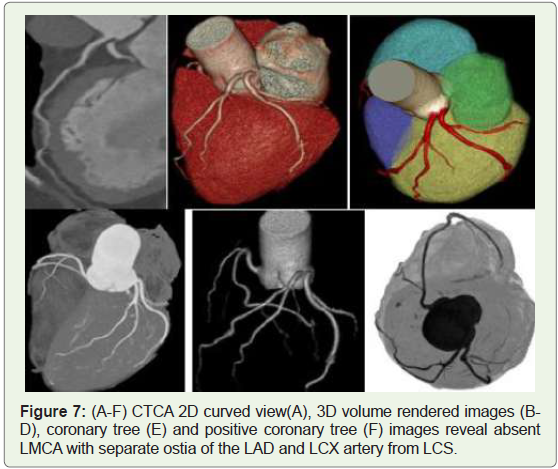
Case 7: Absent left main coronary artery (LMCA) in a 40year-old
woman with normal origin of RCA (Figure 7).
Figure 7: (A-F) CTCA 2D curved view(A), 3D volume rendered images (BD),
coronary tree (E) and positive coronary tree (F) images reveal absent
LMCA with separate ostia of the LAD and LCX artery from LCS.
B. Anomalous location of ostium in the appropriate coronary sinus:
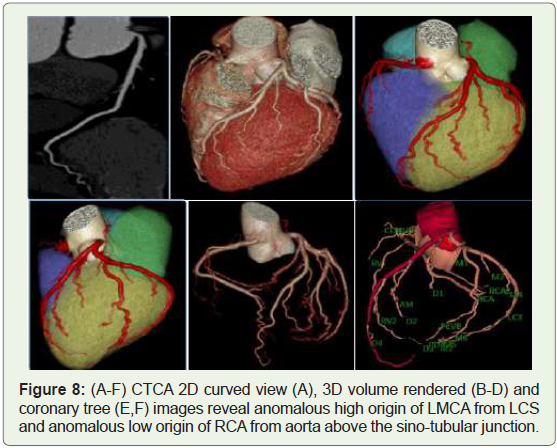
Case 8: 40 year old male with high origin of LMCA from Left
coronary sinus (Figure 8).
Figure 8: (A-F) CTCA 2D curved view (A), 3D volume rendered (B-D) and
coronary tree (E,F) images reveal anomalous high origin of LMCA from LCS
and anomalous low origin of RCA from aorta above the sino-tubular junction.
C. Origin from opposite coronary sinus:
LCX or LAD artery arising from the right coronary sinus (RCS)Case 9: 70 year-old women with absent LMCA and anomalous
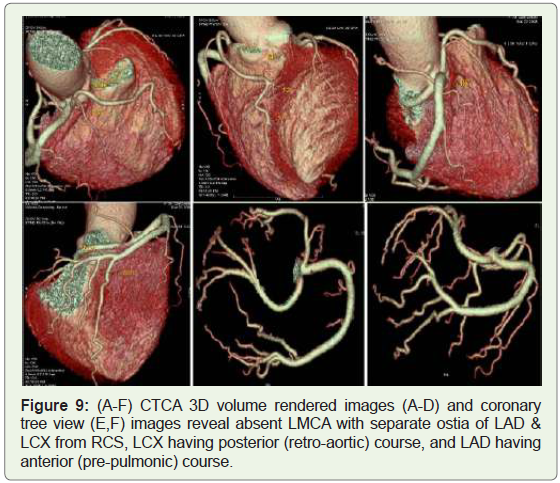
origin of LAD & LCX arteries from RCS (Figure 9).
Figure 9: (A-F) CTCA 3D volume rendered images (A-D) and coronary
tree view (E,F) images reveal absent LMCA with separate ostia of LAD &
LCX from RCS, LCX having posterior (retro-aortic) course, and LAD having
anterior (pre-pulmonic) course.
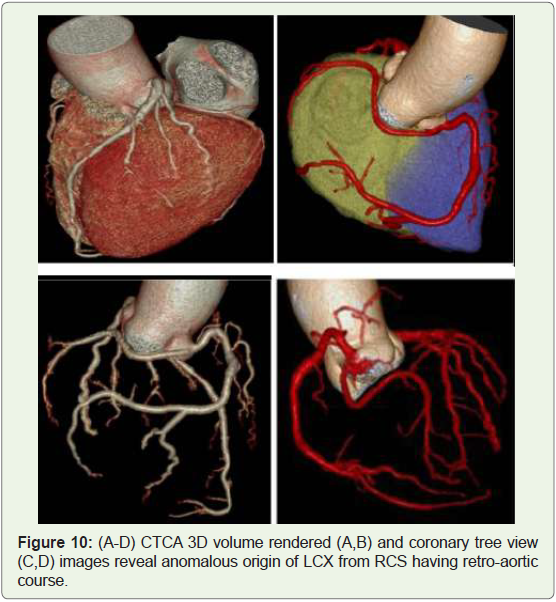
Case 10: 40 years old man with anomalous origin of LCX from
RCS (Figure 10).
Figure 10: (A-D) CTCA 3D volume rendered (A,B) and coronary tree view
(C,D) images reveal anomalous origin of LCX from RCS having retro-aortic
course.
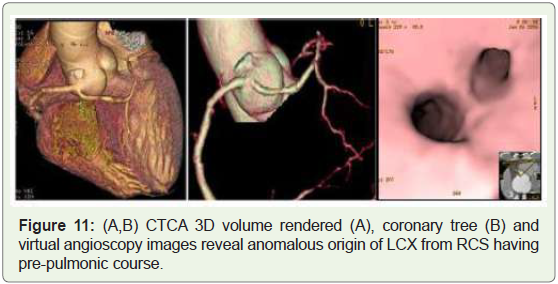
Case 11: 58 years old man with anomalous origin of LCX from
RCS (Figure 11).
Figure 11: (A,B) CTCA 3D volume rendered (A), coronary tree (B) and
virtual angioscopy images reveal anomalous origin of LCX from RCS having
pre-pulmonic course.
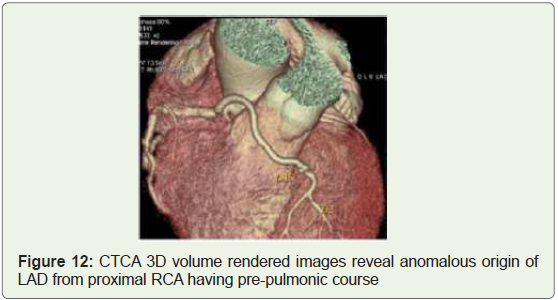
Case 12: 45 years old woman with anomalous origin of LAD
branching off from proximal RCA (Figure 12).
Figure 12: CTCA 3D volume rendered images reveal anomalous origin of
LAD from proximal RCA having pre-pulmonic course
RCA arising from the LCS:
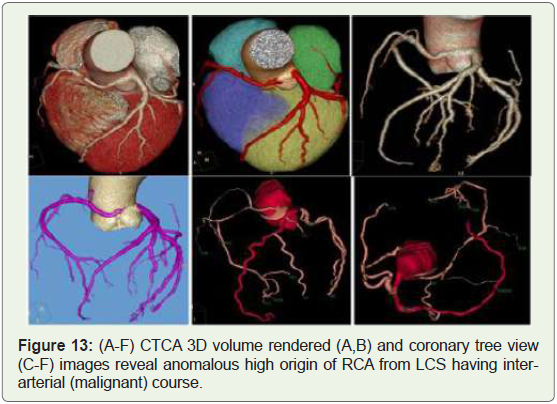
Case 13: 35 years old man with nonspecific chest pain on exertion
(Figure 13).
Figure 13: (A-F) CTCA 3D volume rendered (A,B) and coronary tree view
(C-F) images reveal anomalous high origin of RCA from LCS having interarterial
(malignant) course.
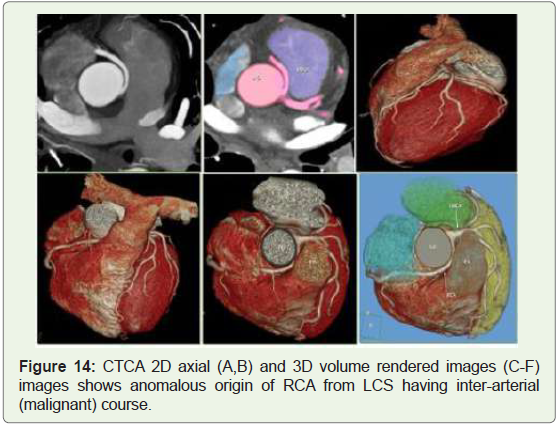
Case 14: 35 years old man with recurrent chest pain on exertion
(Figure 14).
Figure 14: CTCA 2D axial (A,B) and 3D volume rendered images (C-F)
images shows anomalous origin of RCA from LCS having inter-arterial
(malignant) course.
Origin from Non-coronary sinus (NCS)-LCA or RCA (or branch of either artery):
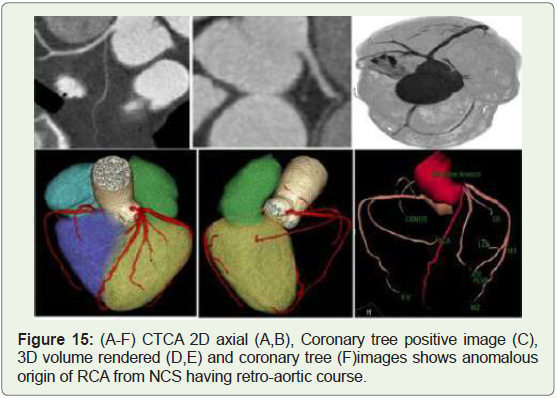
Case 15: 63 years old woman with anomalous origin of RCA from
NCS (Figure 15).
Figure 15: (A-F) CTCA 2D axial (A,B), Coronary tree positive image (C),
3D volume rendered (D,E) and coronary tree (F)images shows anomalous
origin of RCA from NCS having retro-aortic course.
Abnormalities of angle of origin:
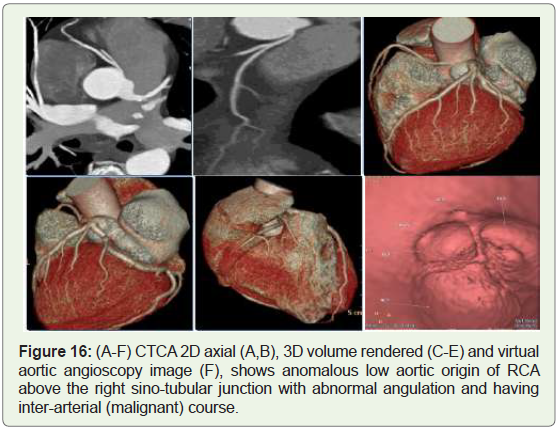
Case 16: 48-year-old male with recurrent chest pain on exertion
(Figure 16).
Figure 16: (A-F) CTCA 2D axial (A,B), 3D volume rendered (C-E) and virtual
aortic angioscopy image (F), shows anomalous low aortic origin of RCA
above the right sino-tubular junction with abnormal angulation and having
inter-arterial (malignant) course.
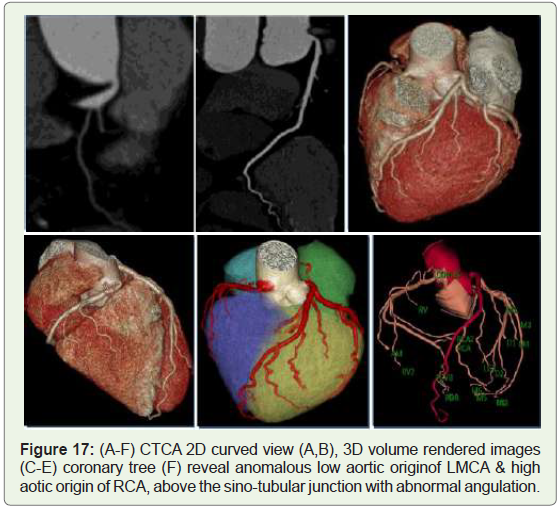
Case 17: 45 year old woman with lowaortic origin of LMCA and
high aortic of RCA (Figure 17).
Figure 17: (A-F) CTCA 2D curved view (A,B), 3D volume rendered images
(C-E) coronary tree (F) reveal anomalous low aortic originof LMCA & high
aotic origin of RCA, above the sino-tubular junction with abnormal angulation.
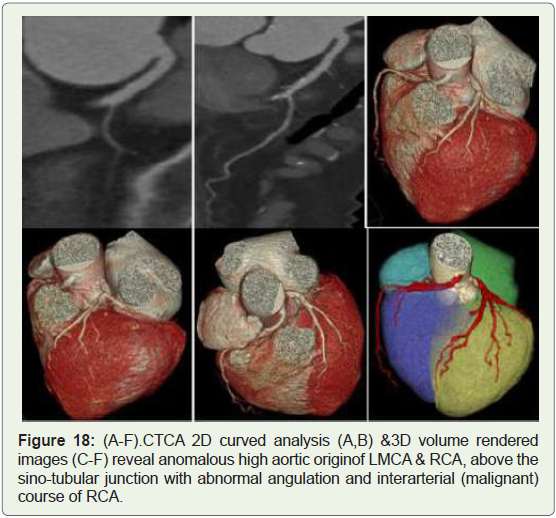
Case 18: 65 year old female with recurrent chest pain on exertion
(Figure 18).
Figure 18: (A-F).CTCA 2D curved analysis (A,B) &3D volume rendered
images (C-F) reveal anomalous high aortic originof LMCA & RCA, above the
sino-tubular junction with abnormal angulation and interarterial (malignant)
course of RCA.
Anomalies of course (normal origin); Myocardial bridging, Duplication:
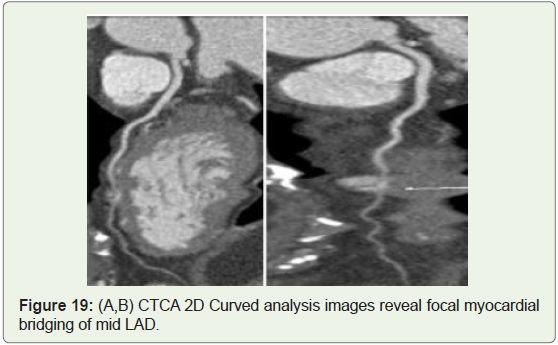
Case 19: 38 yerars old man with atypical chest pain on exertion
(Figure 19).
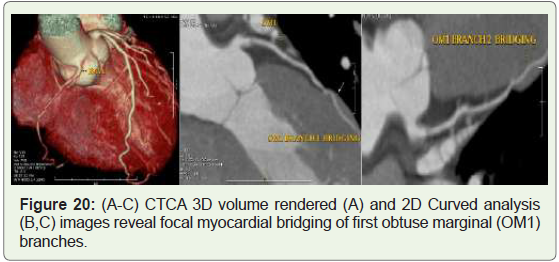
Case 20: 49 years old man with atypical chest pain (Figure 20).
Figure 20: (A-C) CTCA 3D volume rendered (A) and 2D Curved analysis
(B,C) images reveal focal myocardial bridging of first obtuse marginal (OM1)
branches.
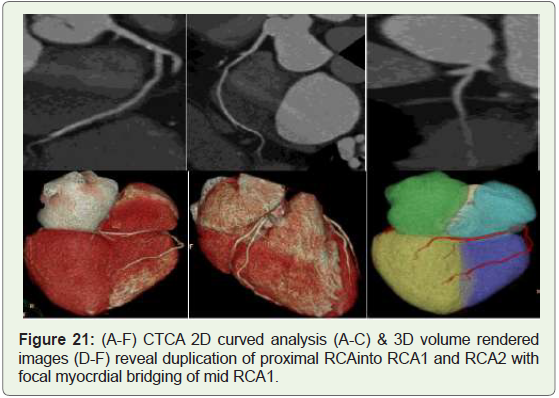
Case 21: 64 year old male with atypical chest pain (Figure 21).
Figure 21: (A-F) CTCA 2D curved analysis (A-C) & 3D volume rendered
images (D-F) reveal duplication of proximal RCAinto RCA1 and RCA2 with
focal myocrdial bridging of mid RCA1.
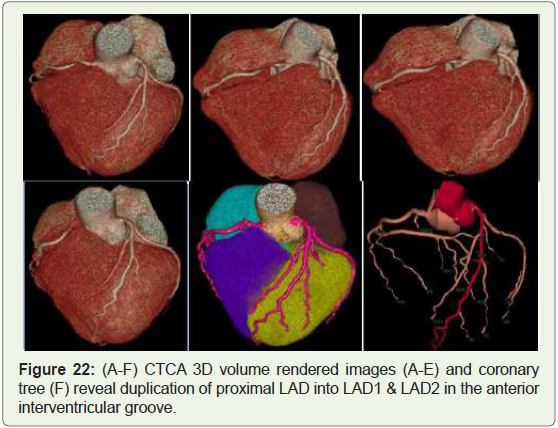
Case 22: 45 year old male with duplication of LAD (Figure 22).
Figure 22: (A-F) CTCA 3D volume rendered images (A-E) and coronary
tree (F) reveal duplication of proximal LAD into LAD1 & LAD2 in the anterior
interventricular groove.
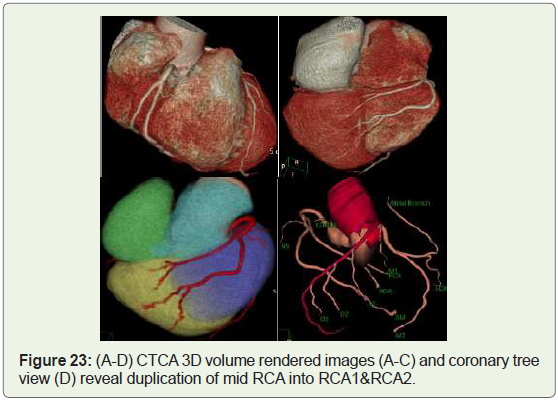
Case 23: 48 year old female with RCA duplication (Figure 23).
Figure 23: (A-D) CTCA 3D volume rendered images (A-C) and coronary tree
view (D) reveal duplication of mid RCA into RCA1&RCA2.
Intrinsic coronary arterial abnormality; Coronary stenosis, Atresia, Ectasia / Aneurysm:
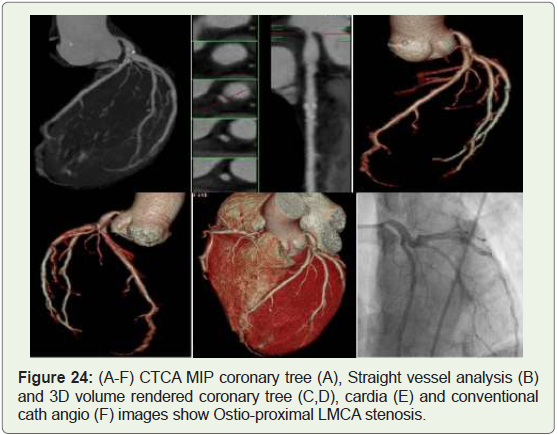
Case 24: 59 year old man with unsuccesful attempts at
catheterisation of LMCA,who developed arrythmias while trying to
enter the ostia (Figure 24).
Figure 24: (A-F) CTCA MIP coronary tree (A), Straight vessel analysis (B)
and 3D volume rendered coronary tree (C,D), cardia (E) and conventional
cath angio (F) images show Ostio-proximal LMCA stenosis.
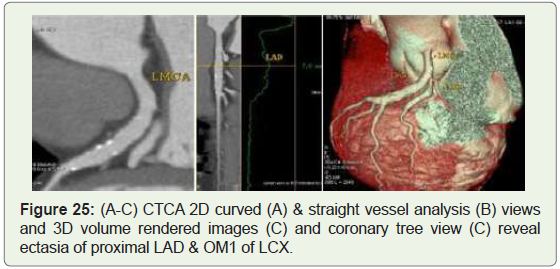
Case 25: 71 year old male with LAD & first obtuse marginal
(OM1) ectasia (Figure 25).
Figure 25: (A-C) CTCA 2D curved (A) & straight vessel analysis (B) views
and 3D volume rendered images (C) and coronary tree view (C) reveal
ectasia of proximal LAD & OM1 of LCX.
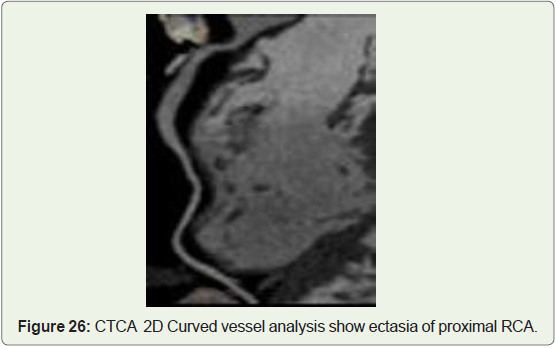
Case 26: 45 old female with RCA ectasia (Figure 26).
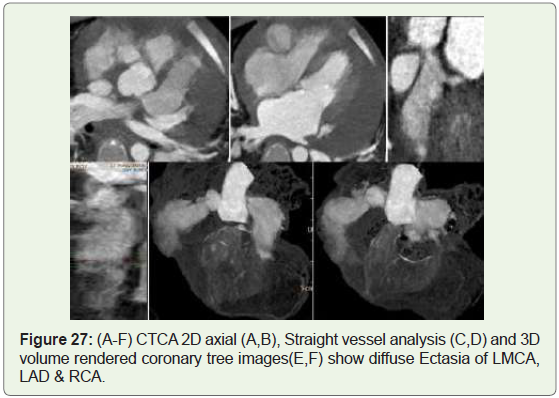
Case 27: 3 year old female child with Kawasaki disease (Figure 27).
Figure 27: (A-F) CTCA 2D axial (A,B), Straight vessel analysis (C,D) and 3D
volume rendered coronary tree images(E,F) show diffuse Ectasia of LMCA,
LAD & RCA.
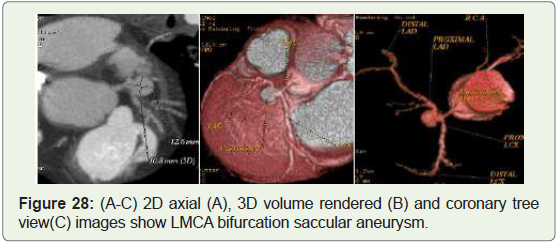
Case 28: 35 year old male with LMCA bifurcation aneurysm
(Figure 28).
Figure 28: (A-C) 2D axial (A), 3D volume rendered (B) and coronary tree
view(C) images show LMCA bifurcation saccular aneurysm.
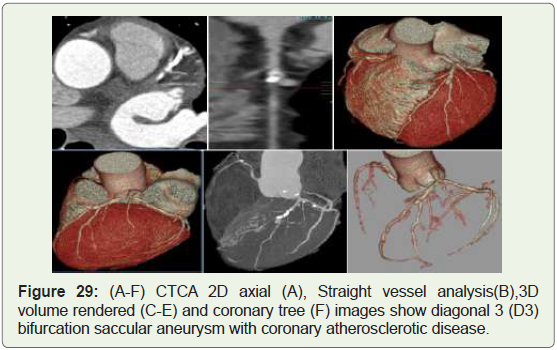
Case 29: 65 year old male with LAD aneurysm (Figure 29).
Figure 29: (A-F) CTCA 2D axial (A), Straight vessel analysis(B),3D
volume rendered (C-E) and coronary tree (F) images show diagonal 3 (D3)
bifurcation saccular aneurysm with coronary atherosclerotic disease.
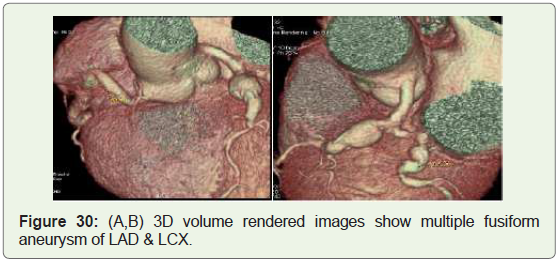
Case 30: 65 year man with LAD & LCX aneurysm (Figure 30).
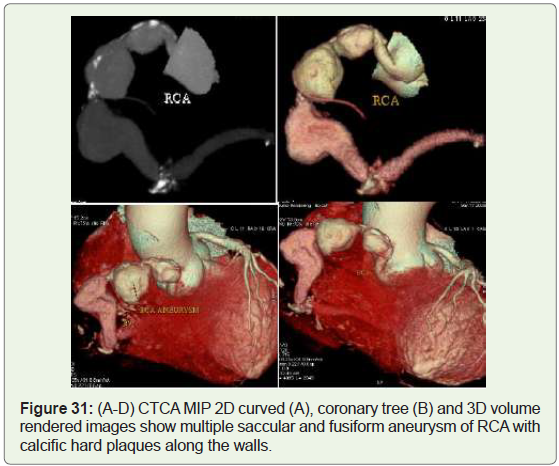
Case 31: 71year old man with multiple atherosclerotic RCA
aneurysm (Figure 31).
Figure 31: (A-D) CTCA MIP 2D curved (A), coronary tree (B) and 3D volume
rendered images show multiple saccular and fusiform aneurysm of RCA with
calcific hard plaques along the walls.
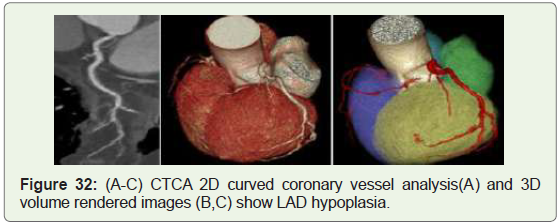
Case 32: 62 year old man with LAD hypoplasia (Figure 32).
Discussion
The coronary arteries arise from the aortic sinuses, converging
towards the apex of the heart. Normally, there are three main coronary
arteries, the right coronary artery (RCA) which typically arises
from the right sinus of Valsalva (RSV) of the ascending aorta and
supplies the right side of the heart, left circumflex artery (LCX) and
left anterior descending (LAD), artery arising from a common stem,
the left main coronary artery (LMCA) which arises from left sinus of
Valsalva (LSV). Among these, the origin of the posterior descending
coronary artery (PDA) from either the right (70%) (Figure 1) or the
left (10%) (Figure 2) coronary artery defines the coronary dominance,
co-dominance (Figure 3) in 20% of cases, with the dominant artery
usually providing blood supply to the sino-atrial (SA) and atrioventricular
(AV) nodes, albeit with some exceptions. Other common
possible findings include trifurcation of the LMCA, with a Ramus
intermedius (in ≈20% of the cases) (Figure 4), distributing across a
variable portion of the lateral wall of the left ventricle [7].
Congenital coronary anomalies (CCA) may be defined as a
coronary pattern or feature that’s encountered in less than 1% of the
general population.
In summary, we are able to divide the coronary feature in two
groups:
(1) Normal coronary anatomy, defined as any morphological
characteristics seen in > 1% of unselected sample. This group also
includes normal anatomical variants, defined as alternative and
comparatively unusual morphological feature observed in > 1% of
the population; and
(2) Anomalous coronary anatomy, defined as morphological
features found in < 1% of the population [5-7].
Most of the coronary anomalies remain asymptomatic and are
incidental to investigations by coronary angiography. Coronary
artery anomalies are classified as benign (80.6%) but potentially
serious anomalies (19.4%) [2].
Anatomic characteristics that make a CCA malignant include-
(1) Single coronary artery (Figures 5 & 6),
(2) Origin from the pulmonary artery,
(3) Origin from the opposite aortic sinus (Figure 9-11,Figure 13,14),
(4) Passing between the aorta and pulmonary artery (Figure 13,14,Figure 16,Figure 18),
(5) Acute-angle take off resulting in a slitlike orifice (Figure 16 & 17),
(6) Passing intramurally (Figure 19-21), and
(7) Small artery due to ostial stenosis or atresia [2].
For several decades, these anomalous coronary arteries were
identified by conventional catheter coronary angiography. MDCT
coronary angiography has been accepted as the ideal method for
evaluation of patients with atypical chest pain due to its excellent
temporal and spatial resolution [2,8].
Among patients with CCA identified with MDCT, conventional
angiography alone allowed correct identification of the anomalies in
precisely 53% of cases [2].
Magnetic resonance coronary angiography could be a noninvasive
method that doesn’t require the utilization of contrast
agents or ionizing radiation, and thus is superior compared to
cardiac CT angiography and conventional coronary angiography.
Its disadvantages are lengthy acquisition time and lower spatial
resolution [9].
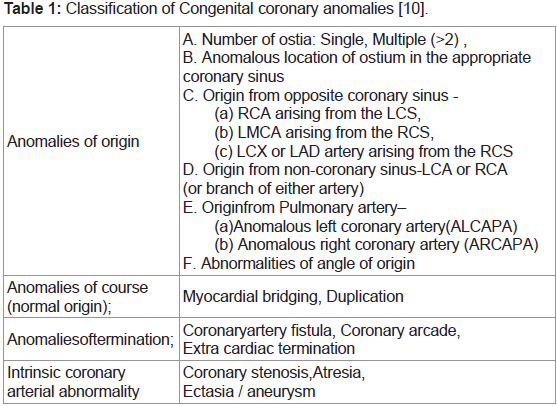
Congenital coronary anomalies are classified according to origin,
course, termination and intrinsic arterial abnormality [10] (Table 1).
Table 1: Classification of Congenital coronary anomalies [10].
Anomalies of origin:
A. Number of Ostia: Single, Multiple (>2):
Single coronary artery is an extremely rare congenital abnormality
seen in 0.024%-0.044% of the population (Figure 5 & 6) [11]. In most
of the cases, aberrant RCA originates from the left main coronary
artery and traverses anterior to the right ventricle or between the
pulmonary trunk and ascending aorta [12]. The presence of a single
coronary artery with an inter-arterial course may increase the danger
of major adverse cardiac events [13]. A proximal obstruction within
the main trunk can be devastating, due to the unfeasibility of collateral
circulation development.The RCA originating as a branch from the midportion of the LAD
may be a very rare anomaly (Figure 5). Six cases have been reported
within the literature up to now, and no patient had underlying
congenital heart disease [14].
The absence of the LMCA with a separate origin of the LAD and
LCX arteries is found in up to 0.67% of subjects (Figure 7) [7]. They
may cause difficulties in catheterization during angiography, but they
allow for the development of collateral circulation in the event of
proximal obstruction in one of those vessels [8].
B. Anomalous location of ostium in the appropriate coronary sinus (Figure 8):
Ectopic RCA from right sinus of valsalva has a frequency of
1.13% and therefore by some classifications can be considered a
variant.C. Origin from opposite coronary sinus:
According to historical reports, both the right and left coronary
arteries originating from the opposite sinus could also be related to an
increased risk of SCD [15].(a) LCX or LAD artery arising from the RCS:
The LCX artery most ordinarily arises from a separate ostium
within the right sinus or as a proximal branch of the RCA in
approximately 0.32%–0.67% of the population (Figure 9-11)
[16]. Retro-aortic pathway is its commonest course, and there’s no
association with sudden death [8] (Figure 10).An aberrant origin of the LMCA or LAD from the right sinus of
Valsalva could be a rare anomaly that has been related to myocardial
ischemia and sudden cardiac death [17] (Figure 9). Depending on the anatomic relationship of the anomalous vessel to the aorta
and the pulmonary trunk, the anomaly is classified into 4 common
courses: posterior (retro-aortic), interarterial (preaortic), anterior
(prepulmonic), and septal (subpulmonic) course.
LAD artery branching off the RCA called the Type IV “Dual LAD”
artery whose incidence ranges from 0.01 to 0.03% [18] (Figure 12).
(b) RCA arising from the LCS:
RCA originating from the left coronary sinus or as a branch of a
single coronary artery is found in 0.03% to 0.17% of the individuals
submitted to angiography (Figures 13-15) [19]. The most common
course of anomalous RCA from LCSis inter-arterial (malignant)
(Figures 13,14). This variant will be related to sudden cardiac
death in up to 30% of patients [20]. According to the finding on
CT angiography, the interarterial course of the RCA is classified as
either “high” or “low”. A high course/outflow tract predisposes the
patient to more adverse effects, like angina and sudden death, and
requires more attention on the part of the radiologist. This is due to
the fact that, during systole, both vessels adjacent to the coronary
artery (the aorta and pulmonary artery) dilate, narrowing the channel
through which the anomalous coronary artery passes, a phenomenon
that’s aggravated during physical work out. Conversely, when
the interarterial course is low, the right ventricular outflow tract
contracts during systole, counterbalancing the systolic expansion of
the aorta and creating less narrowing within the coronary arterial
course between the right ventricular outflow tract and the aorta [21].(c) LMCA arising from the RCS:
LMCA originating from the right coronary sinus (RCS) or as a
branch of a single coronary artery occurs in 0.09% to 0.11% of the
individuals submitted to angiography [19]. Proximal inter-arterial
course occurs in 75% of such patients [19]. The origin of the LMCA
from the RCS will be classified into 4 types- the LMCA passes between
the aorta and pulmonary trunk (interarterial), anteriorly over the
right ventricular outflow tract (pre-pulmonic), right of the RCA and
pass posteriorly to the aortic root (retroaortic) and along the crista
supraventricularis intramyocardially or subendocardially, surfacing
within the proximal interventricular sulcus.Sudden death can result from transient compression of the
anomalous left coronary artery course, caused by dilation of the
aorta and pulmonary artery, which is successively caused by the
rise in blood flow during intense exercise, thus creating torsion or
compression of the coronary artery between the aorta and also the
right ventricular outflow tract [22].
D. Origin from Non-coronary sinus-LCA or RCA (or branch of either artery):
RCA from posterior sinus or Non-coronary sinus is a very rare
anomaly (0.003%) in hearts without other congenital anomalies
(Figure 15). The anomaly is sometimes not related to symptoms or
complications so should be considered benign [23].The origin of the LMCA from posterior sinus of Valsalva is
sometimes seen in patients with other anomalies of heart and
great vessels. This situation is extremely rare (0.0008%) and even more rarely associated with sudden death. This anomaly should be
considered benign [23].
E. Origin from Pulmonary artery (PA):
(a) Anomalous left coronary artery from PA (ALCAPA)It is also referred as Bland-white-Garland syndrome from the
eponym of the authors who described it for the primary time in
1956. ALCAPA is one in all the foremost serious congenital coronary
artery anomalies with prevalence of 1 in 300,000 live births [24]. Most
affected patients show symptoms in infancy and early childhood.
Approximately 90% of untreated infants die within the 1st year of
life, and only some patients survive to adulthood [25]. In a study by
Alexander. et al, the patients with ALCAPA had the extra finding of
aortic coarctation with a patent ductus arteriosus [26].
(b) Anomalous right coronary artery from PA (ARCAPA):
ARCAPA has an incidence of 0.002% within the general
population. Only 25-30% cases are related to structural heart defects
[27]. Patients with associated cardiac anomalies are diagnosed
early in life compared to patients with isolated ARCAPA. Those
with associated cardiac defects may present with cardiac murmur,
congestive symptoms, and sudden cardiac death or may remain
asymptomatic and detected incidentally during evaluation of other
problem [28]. The associated cardiac defects found in these patients
were aorto-pulmonary window, tetralogy of Fallot, VSD, PDAAQ3,
and aortic stenosis [28].Left anterior descending artery from PA- Pulmonary origin of the
LAD could be a very rare with a frequency of 0.0008%. This anomaly
leads to myocardial ischaemia and sudden cardiac death [2].
All coronary arteries from PA - In this anomaly all the coronary
arteries arise from PA, therefore the entire coronary circulation is
supplied by the pulmonary artery. The prognosis of this CAA is poor
and these patients usually die during the primary month of life. This
CAA is additionally related to patent ductus arteriosus and with other
major anomalies of the heart or great arteries [26].
F. Abnormalities of angle of origin:
Acute take-off of LCX: Angelini et al defined this coronary
anomaly in patients with an angle ≤ 45º between LMCA and LCX,
within the left anterior oblique/caudal and/or right anterior oblique/
caudal angiography Xray
projections [29]. This anatomic variant has
an incidence around 2% and should be relevant in reference to the
technical difficulties that can complicate angiographic procedures on
the LCX [29].
Anomalous location of the coronary ostium in the aortic root (Figure 16-18):
High origin of a coronary artery or left coronary trunk -Is defined
as origin over 1 cm above the sino-tubular junction [30]. Preoperative
identification of this anomaly is very important just in case of
ascending aorta surgery and should cause difficulties in catheterization
during angiography. Cross clamping of the aorta below a high-origin
coronary artery may lead to unsuccessful induction of cardioplegia
(Figure 18). Moreover, the higher the position of a coronary ostium, the higher the risk of coronary hypoperfusion, because the sinuses of
Valsalva facilitate maximal coronary diastolic perfusion [30].Anomalies of course (normal origin); Myocardial bridging, Duplication.:
Myocardial bridging: A muscular or myocardial bridge
is defined as an atypical course of artery during which it dips
intramyocardially with resulting compression of the vessel during
systole (Figure 19-21). The prevalence of this anomaly incorporates
a wide selection from 0.15%-25% angiographically, to between 5%
and 86% at autopsy. However, many reports from angiography
may underestimate the prevalence of this anomaly, as recent studies
with computed axial tomography (CT) have shown that myocardial
bridging may be found in up to 25% of patients [31]. In some cases,
myocardial bridging is responsible for angina pectoris, myocardial
infarction, life-threatening arrhythmias, or perhaps death [32].
Duplication: Duplication of RCA (60%) &duplication of the
LAD artery (40%) and their incidence in normal hearts is about
0.13%-1% of the overall population [33].
A split RCA is defined as an RCA that features a split PDA with
the anterior subdivision of the RCA resulting in the distal portion
of the PDA in the anterior free wall of the RV (Figures 21,23). The
posterior bifurcation of the RCA maintains a course within the
atrio-ventricular groove. Split RCA is typically called “double RCA”,
whether or not truly there don’t seems to be two RCAs, but split
portions of the posterior descending branch of the RCA with separate
proximal courses and forms the uppermost portion of the posterior
descending branch [1].
Duplication of LAD isn’t intrinsically haemodynamically
significant, but its presence may complicate surgical intervention
when aorto-coronary bypass or other coronary artery surgery is
performed [30] (Figure 22).
Anomalies of termination; Coronary artery fistula, Coronary arcade, Extracardiac termination:
Coronary artery fistula(CAF): Could be a condition within
which a communication exists between one or two coronary arteries
and either a cardiac chamber, the coronary sinus, the superior vena
cava, or the pulmonary artery. In CAF, the involved coronary artery
is dilated because of increased blood flow and is usually tortuous to
an extent determined by the shunt volume [34]. The drainage site of
the fistula encompasses a greater clinical and physiologic importance
than does the artery of origin. The foremost common site of drainage
is that the RV (45%), RA (25%),& PA (15%) [35]. The fistula drains
into the LA or LV in less than 10% of cases [36].
When the shunt leads into a right-sided cardiac chamber, the
hemodynamics resembles those of an extra cardiac left-to-right
shunt; when the connection is to a left-sided cardiac chamber, the
hemodynamics mimic those of aortic insufficiency. Myocardial
perfusion is also diminished for that portion of the myocardium
supplied by the abnormally connecting coronary artery. This situation
represents a hemodynamic steal phenomenon and will result in
myocardial ischemia [37].
Coronary arcade: Could be a rare instance of communication
that’s large enough to be identified angiographically between the
RCA and therefore the LCA within the absence of coronary artery
stenosis [38].
Extracardiac termination: Connections may exist between the
coronary arteries and extracardiac vessels (ie, the bronchial, internal
mammary, pericardial, anterior mediastinal, superior and inferior
phrenic, and intercostal arteries and also the esophageal branch of
the aorta [9].
Intrinsic coronary arterial abnormality; Coronary stenosis, Atresia, Ectasia / aneurysm:
Atresia: Coronary ostial stenosis or atresia may be a spectrum
of rare developmental conditions with different patho-physiologic
mechanisms and clinical implications. Coronary ostial stenosis or
atresia affects the LMCA (Figure 24), more frequently than it does
the RCA. In adult patients with coronary ostial or proximal coronary
stenosis it can be difficult to rule out acquired causes, like arteritis,
which might produce conditions morphologically same as COSA; for
instance, atherosclerotic, syphilitic, Kawasaki, and Takayasu aortitis
have all been cited as causes of acquired ostial stenosis or occlusion
[39]. LMCA atresia ia a rare congenital malformation, which is
charecterised by absence of left coronary ostium and left main trunk
within the left coronary arterial system. Patients is also asymptomatic
or present with syncope,angina pectoris, myocardial infarction or
sudden death [40].
Coronary artery aneurysms and ectasia are characterized by an
abnormal dilatation of a coronary artery.
Ectasia: Ectasia is diffuse dilatation (>1.5 times the normal
diameter) of the coronary arteries that involves 50% or more of the
length of the artery [46]. Coronary artery ectasia is more common
than coronary artery aneurysm (Figure 25-27) [42].
In Western countries, atherosclerotic aneurysms are most
typical (50%), followed by congenital (17%) and infectious causes
(10%); and in Japan, Kawasaki disease represents the predominant
cause of coronary artery aneurysm [41,44]. Most of the patients are
asymptomatic, and dilatation is an incidental finding. There’s the
chance of thrombosis, embolization and rupture.
Aneurysm: Is a focal dilatation of the vessel [43]. In true
aneurysm the vessel wall consists of three layers: adventitia, media,
and intima and in False aneurysm, the Vessel wall consists of one or
two layers.Definition; coronary artery segments that have a diameter
that exceeds the diameter of normal adjacent coronary segments or
the diameter of the patient’s largest coronary vessel by 1.5 times and
involve less than 50% of the overall length of the vessel [41].
The reported frequency of coronary artery aneurysms varies
widely from 0.3% to 5% [43]. The incidence is higher in men than
in women (2.2% vs 0.5%)[38]. Atherosclerotic aneurysms are usually
multiple & involve more than one coronary artery (Figures 29-31), as
compared with congenital, traumatic, or dissecting aneurysms, which
are mainly solitary [43,44]. The RCA is that the most often involved
vessel(40%-61%), followed by LAD (15%-32%) & the LCX (15%–
23%) [45] (Figure 29-31), left main trunk involvement is rare (0.1%- 3.5%), & its presence is sometimes related to significant underlying
two- or three-vessel artery disease [45] (Figure 28).
According to the American Heart Association statement,
aneurysms are also classified in keeping with internal diameter
as, small (<5-mm), medium (5–8-mm), or giant (>8-mm) [46].
In children, according to Japanese Ministry of Health in 1984, a
coronary artery aneurysm is present when the diameter of the lumen
is > 3 mm in children younger than 5 years old or > 4 mm in those 5
years old or older [46].
Kawasaki disease(mucocutaneous lymph node syndrome), It is an acute self-limited multisystemic panarteritis which will
occur worldwide; nevertheless, it’s markedly more prevalent in
Japan and in children of Japanese ancestry [47]. The etiology of
mucocutaneous lymph node syndrome remains unclear, although
several epidemiologic and clinical features are strongly suggestive
of an infectious cause because the initiating factor in a genetically
susceptible host, but autoimmunity can also play a role in the
pathogenesis [48].
Cardiac sequelae are the foremost important manifestations
of Kawasaki disease and include coronary artery dilatation (Figure
27), Premature atherosclerosis and stenosis (4.7%), thrombosis, or
occlusion with myocardial infarct (1.9%) [45].
Coronary artery aneurysm or coronary artery ectasia (Figure 27),
develops in 15%-25% of untreated children with Kawasaki disease,
in most cases within 3-6 months of the acute illness [49], but since
the introduction of γ-globulin therapy, coronary artery aneurysm or
ectasia occurs in exactly 5% of the cases [50]. Moreover, by 2 years
after the onset of mucocutaneous lymph node syndrome, 49% of
the patients have spontaneous regression of the aneurysms [51].
The LMCAis involved in 12% of the cases, the RCA in 3%,and both
arteries in 8% (Figure 27) [52].
Hypoplasia of coronary arteries: Congenital hypoplasia of
coronary arteries presents as a narrowed luminal diameter (less than
1.5 mm) in one or two of the three main coronary arteries with no
compensatory branches. Limitations to blood flow caused by the
narrow lumen result in symptoms of myocardial ischemia and sudden
cardiac death. Most frequent variants in reported cases are hypoplasia
of both LCX and RCA and hypoplasia of the LAD (Figure 32) [53].
Conclusion
Increasing use of MDCT for cardiac imaging has helped in the
detection of many benign congenital coronary anomalies, but a
little number is related to myocardial ischemia and sudden death.
Increasing the employment of MDCT in cardiac imaging may
yield diagnostic information on congenital coronary anomalies not
obtained with invasive coronary angiography. Axial sections, multi
planar reconstructions, virtual angioscopy, and 3D volume-rendered
images should aid in the detection and improve the interpretation
of such anomalies, which can be of immense help to the clinician
planning interventional procedures.
References
Citation
Kumar K, Viswanathan M, Dharan Venkatesh KA, Senthinathan V, Nasreen I. MDCT Evaluation of Congenital Coronary Anomalies:
Pictorial Essay. Indian J Appl Radiol. 2022;8(1): 175.