Case Report
Primary Mediastinal B Cell Lymphoma Presenting As Acute Abdomen
Fathima R1, Pratap T1, Jacob D1 and Jalal MJA2*
1Department of Radiology, VPS Lakeshore Hospital, Kochi, Kerala, India.
2Department of Internal Medicine and Rheumatology, Olive Healthcare, Thrissur, Kerala, India.
2Department of Internal Medicine and Rheumatology, Olive Healthcare, Thrissur, Kerala, India.
*Corresponding author:Muhammed Jasim Abdul Jalal, Department of Internal Medicine and Rheumatology Olive Healthcare, Thrissur, Kerala, India.E-mail Id:jasimabduljalal@yahoo.com
Copyright: © 2026 Fathima R, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information:Submission: 26/03/2026; Accepted: 05/05/2026; Published: 09/05/2026
Abstract
Primary mediastinal B-cell lymphoma (PMBCL) is a distinct subtype of non-Hodgkin lymphoma that typically presents as an anterior mediastinal mass, with limited extrathoracic involvement at diagnosis. Pancreatic involvement is rare and may mimic primary pancreatic pathology.
We describe a rare case of primary mediastinal B‑cell lymphoma (PMBCL) with synchronous mediastinal and pancreatic mass in a 22‑year‑old male who presented with features of acute pancreatitis. Imaging revealed a heterogeneous mass in the pancreatic head with double-duct obstruction, raising suspicion of pancreatic malignancy. A concurrent anterior mediastinal mass was incidentally detected on chest imaging. Endoscopic ultrasound-guided fine-needle aspiration of the pancreatic lesion and biopsy of the mediastinal mass demonstrated a malignant lymphoid neoplasm. Immunohistochemistry showed positivity for CD20, CD30, BCL6, and MUM1, with a high Ki-67 proliferation index, confirming PMBCL. Staging PET-CT demonstrated FDG-avid lesions limited to the mediastinum and pancreas. The patient was initiated on R-CHOP chemotherapy. This case highlights a rare presentation of PMBCL with synchronous pancreatic involvement presenting as acute pancreatitis. Recognition of such atypical presentations is essential to avoid misdiagnosis and unnecessary surgical interventions, emphasizing the importance of histopathology and immunohistochemistry in establishing the diagnosis.
We describe a rare case of primary mediastinal B‑cell lymphoma (PMBCL) with synchronous mediastinal and pancreatic mass in a 22‑year‑old male who presented with features of acute pancreatitis. Imaging revealed a heterogeneous mass in the pancreatic head with double-duct obstruction, raising suspicion of pancreatic malignancy. A concurrent anterior mediastinal mass was incidentally detected on chest imaging. Endoscopic ultrasound-guided fine-needle aspiration of the pancreatic lesion and biopsy of the mediastinal mass demonstrated a malignant lymphoid neoplasm. Immunohistochemistry showed positivity for CD20, CD30, BCL6, and MUM1, with a high Ki-67 proliferation index, confirming PMBCL. Staging PET-CT demonstrated FDG-avid lesions limited to the mediastinum and pancreas. The patient was initiated on R-CHOP chemotherapy. This case highlights a rare presentation of PMBCL with synchronous pancreatic involvement presenting as acute pancreatitis. Recognition of such atypical presentations is essential to avoid misdiagnosis and unnecessary surgical interventions, emphasizing the importance of histopathology and immunohistochemistry in establishing the diagnosis.
Introduction
Primary mediastinal B‑cell lymphoma (PMBCL) is a distinct
subtype of non‑Hodgkin’s lymphoma (NHL). Earlier, PMBCL was
considered a variant of diffuse large B‑cell lymphoma (DLBCL);
however, owing to its unique clinical, molecular, and histopathological
characteristics, it is now recognised as a separate entity in the World
Health Organization (WHO) classification of lymphoid neoplasms
[1]. PMBCL typically presents as a mediastinal mass with non‑specific
symptoms. Local spread to adjacent thoracic structures such as the
chest wall, pleura, lungs, pericardium, and heart is common. In
contrast, extrathoracic dissemination to extranodal sites including
kidneys, ovaries, central nervous system, gastrointestinal tract, liver,
and pancreas is uncommon [1].
Pancreatic involvement at presentation occurs in only 0.2– 2% of patients with NHL [2]. Synchronous involvement of the anterior mediastinum and pancreas by PMBCL, presenting as acute pancreatitis, is very rare. We report such an unusual presentation in a 22-year-old male who presented with acute pancreatitis.
Pancreatic involvement at presentation occurs in only 0.2– 2% of patients with NHL [2]. Synchronous involvement of the anterior mediastinum and pancreas by PMBCL, presenting as acute pancreatitis, is very rare. We report such an unusual presentation in a 22-year-old male who presented with acute pancreatitis.
Case Report
A 22‑year‑old male presented with acute‑onset abdominal pain,
jaundice, and generalized pruritus. He was referred from another
hospital with a provisional diagnosis of acute pancreatitis. Initial
laboratory investigations revealed markedly elevated serum amylase
(1224 U/L) and lipase (3911 U/L). Ultrasonography of the abdomen
demonstrated a hypoechoic lesion in the pancreatic head region,
without evidence of gallstones or biliary or pancreatic duct calculi.
The patient had no significant past medical history, no family
history of autoimmune disease or malignancy, and no history of
alcohol intake. On physical examination, icterus was present. There
was no lymphadenopathy, abdominal tenderness, or palpable mass
per abdomen.
Laboratory evaluation showed normal blood counts (total leukocyte count 9960/mm³; neutrophils 65%, lymphocytes 23%; haemoglobin 14.4 g/dL; platelet count 3.2 × 10⁵/mm³). Biochemical parameters revealed elevated liver enzymes (total bilirubin 15.4 mg/ dL; direct bilirubin 10.4 mg/dL, AST 123 U/L, ALT 276 U/L, alkaline phosphatase 391 U/L), and raised lactate dehydrogenase (LDH) 718 U/L.
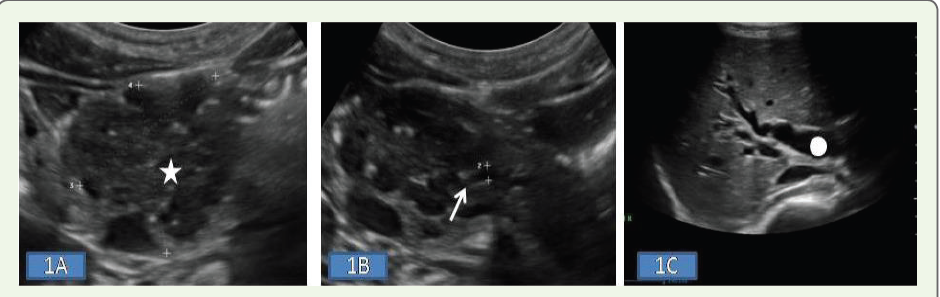
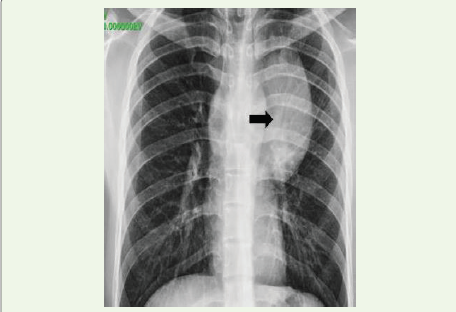
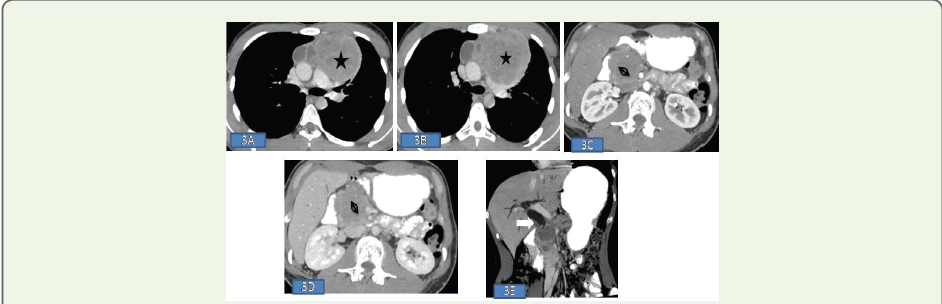
Abdominal ultrasonography [Figure 1A-C] showed a moderately sized heteroechoic mass in the pancreatic head with cystic areas and evidence of double‑duct obstruction. Serum CA 19‑9 was elevated (162 U/mL), raising suspicion of a pancreatic neoplasm. A routine chest radiograph [Figure 2A] revealed a large homogenous opacity with wide base towards the mediastinum with preserved aortic silhouette, suggestive of an anterior mediastinal mass. Contrast-enhanced CT of the thorax demonstrated a heterogeneous anterior mediastinal mass measuring approximately 6.7 × 10.5 × 9.1 cm, contiguous with the aortic arch, without significant mediastinal lymphadenopathy [Figure 3A-E]. CT of the abdomen showed a 4.4 × 4.6 cm heterogeneous mass in the pancreatic head with mild exophytic extension and double‑duct obstruction, without calcification or vascular invasion.
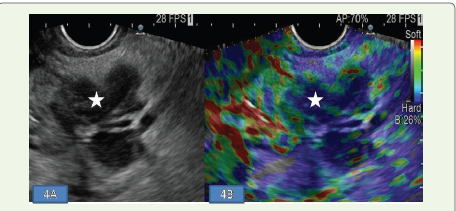
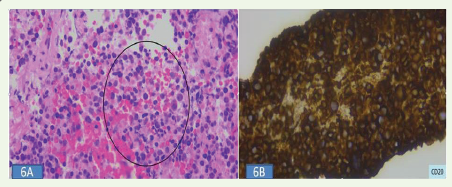
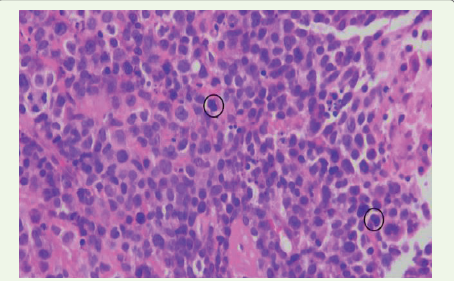
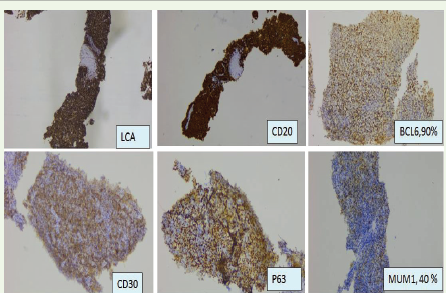
Given the unusual dual involvement, tissue diagnosis was pursued from both sites. Endoscopic ultrasound (EUS) with elastography demonstrated a hard pancreatic lesion, following which fine-needle aspiration (FNA) was performed [Figure 4A, B]. Ultrasound‑guided trucut biopsy was performed from the mediastinal mass. Cytology from the pancreatic lesion revealed a round‑cell neoplasm suggestive of lymphoma [Figure 6A]. Histopathological examination of the mediastinal mass showed small to medium sized cells arranged in sheets with round to oval nucleus and scanty cytoplasm, consistent with malignant round cell neoplasm [Figure 7]. Immunohistochemistry was positive for LCA, CD20, CD3, CD30, BCL6, MUM1, and P63, and negative for CD10, cyclin D1, TdT, BCL2, CD23, CD34, and PAX8. The Ki‑67 proliferation index was 85–90% [Figure 8]. FNA material from the pancreatic lesion was also positive for CD20 [Figure 6B]. These findings confirmed the diagnosis of PMBCL with pancreatic involvement.
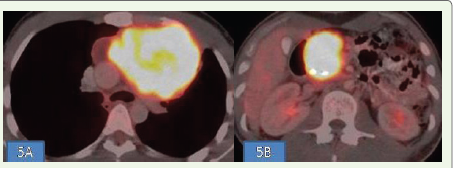
Staging PET‑CT demonstrated FDG‑avid lesions in the anterior mediastinum (SUV max 20) and pancreas (SUV max 17.5), with no other sites of disease [Figure 5A, 5B]. The patient was initiated on R‑CHOP chemotherapy.
Laboratory evaluation showed normal blood counts (total leukocyte count 9960/mm³; neutrophils 65%, lymphocytes 23%; haemoglobin 14.4 g/dL; platelet count 3.2 × 10⁵/mm³). Biochemical parameters revealed elevated liver enzymes (total bilirubin 15.4 mg/ dL; direct bilirubin 10.4 mg/dL, AST 123 U/L, ALT 276 U/L, alkaline phosphatase 391 U/L), and raised lactate dehydrogenase (LDH) 718 U/L.
Abdominal ultrasonography [Figure 1A-C] showed a moderately sized heteroechoic mass in the pancreatic head with cystic areas and evidence of double‑duct obstruction. Serum CA 19‑9 was elevated (162 U/mL), raising suspicion of a pancreatic neoplasm. A routine chest radiograph [Figure 2A] revealed a large homogenous opacity with wide base towards the mediastinum with preserved aortic silhouette, suggestive of an anterior mediastinal mass. Contrast-enhanced CT of the thorax demonstrated a heterogeneous anterior mediastinal mass measuring approximately 6.7 × 10.5 × 9.1 cm, contiguous with the aortic arch, without significant mediastinal lymphadenopathy [Figure 3A-E]. CT of the abdomen showed a 4.4 × 4.6 cm heterogeneous mass in the pancreatic head with mild exophytic extension and double‑duct obstruction, without calcification or vascular invasion.
Given the unusual dual involvement, tissue diagnosis was pursued from both sites. Endoscopic ultrasound (EUS) with elastography demonstrated a hard pancreatic lesion, following which fine-needle aspiration (FNA) was performed [Figure 4A, B]. Ultrasound‑guided trucut biopsy was performed from the mediastinal mass. Cytology from the pancreatic lesion revealed a round‑cell neoplasm suggestive of lymphoma [Figure 6A]. Histopathological examination of the mediastinal mass showed small to medium sized cells arranged in sheets with round to oval nucleus and scanty cytoplasm, consistent with malignant round cell neoplasm [Figure 7]. Immunohistochemistry was positive for LCA, CD20, CD3, CD30, BCL6, MUM1, and P63, and negative for CD10, cyclin D1, TdT, BCL2, CD23, CD34, and PAX8. The Ki‑67 proliferation index was 85–90% [Figure 8]. FNA material from the pancreatic lesion was also positive for CD20 [Figure 6B]. These findings confirmed the diagnosis of PMBCL with pancreatic involvement.
Staging PET‑CT demonstrated FDG‑avid lesions in the anterior mediastinum (SUV max 20) and pancreas (SUV max 17.5), with no other sites of disease [Figure 5A, 5B]. The patient was initiated on R‑CHOP chemotherapy.
Figure 1: (A–C):Ultrasound images demonstrating a hypoechoic mass (star) with internal cystic areas. The main pancreatic duct (MPD) is dilated distal to the mass (arrow). Dilated intrahepatic biliary radicals and common bile duct (CBD) are also seen (circle).
Figure 2:(A): Frontal chest radiograph showing a well-defined opacity broad based
to the mediastinum without silhouetting of the aorta. Hilar vessels are
visualized through the mass, suggestive of an anterior mediastinal mass.
Figure 3: (A-E):Contrast-enhanced CT images. (A, B) Axial sections of the chest showing a well-defined lobulated mass (star) with heterogeneous enhancement and cystic changes, with mass effect on the pulmonary vessels. (C, D) Axial abdominal sections revealing a circumscribed mass (arrowhead) with heterogeneous enhancement in the pancreatic head and dilated MPD. (E) Coronal reformatted image demonstrating biliary obstruction (arrow).
Figure 4: (A, B):Endoscopic ultrasound images with elastography depicting
a hypoechoic pancreatic mass (star) that appears hard on elastography.
Figure 5:(A, B): FDG PET-CT images through the chest and abdomen
showing FDG-avid lesions in the anterior mediastinum and pancreatic head.
Figure 6: (A, B):Endoscopic ultrasound-guided fine-needle aspiration
cytology (EUS-FNAC) and immunohistochemistry (IHC) from the pancreatic
head mass. Cytology (A) shows medium-sized cells admixed with small and
large cells. IHC (B) demonstrates diffuse CD20 positivity, suggestive of a
round cell neoplasm, possibly lymphoma.
Figure 7:Biopsy from the anterior mediastinal mass showing sheets of
small-to medium-sized atypical lymphoid cells with scant cytoplasm and
round to oval nuclei. Frequent mitotic figures are noted (marked), consistent
with a malignant round cell neoplasm.
Discussion
NHL is a heterogeneous group of lymphoproliferative disorders
primarily involving lymph nodes and lymphatic tissues; however,
approximately 50% of cases demonstrate extranodal involvement
[3]. Most common extranodal site of involvement in children is
gastrointestinal tract (stomach and small intestine being common)
followed by mediastinum. Involvement of pancreas is rarely
encountered [4]. Pancreatic involvement by NHL presenting as acute
pancreatitis is rare, with only a few cases reported [5].
PMBCL typically affects young adults, with a female predominance
in the third or fourth decade of life [1]. Although the incidence of
NHL has historically been lower in Asian populations, recent data
indicate a rising trend [6]. However, recent study conducted in India
showed that about 60% of the cases had no comorbidities or risk
factors at the time of presentation [7]
PMBCL originates from thymic B cells and is characterized
by activation of the JAK–STAT and NF‑κB pathways, along with
immune‑evasion mechanisms involving altered MHC expression
and programmed death ligand upregulation [8]. The malignant cells
express B-cell markers such as CD19, CD20, CD22 and CD79a but
not surface immunoglobulin. CD30 expression is often weak, and
CD15 is negative in contrast to classical Hodgkin lymphoma. B-cell
transcription factors are often positive, including PAX5, BCL6, and
OCT2 [8].
Often, PMBCL involves the mediastinum alone. Involvement of other systems like gastrointestinal tract, kidneys, ovaries, central nervous system and liver is rare [9]. Findings such as pleural or pericardial effusion is indicative of poor prognosis. Extra mediastinal involvement is uncommon at presentation. PMBCL in our case shows both mediastinal and pancreatic masses in which mediastinal mass was asymptomatic. All clinical symptoms were pertaining to pancreatic mass. The close pathologic differentials DLBCL, classical Hodgkin lymphoma presents as cervical, mediastinal or abdominal lymphadenopathy with or without mediastinal mass. Lymphoblastic leukemia is another haematological malignancy which may present with a mediastinal mass. Lymph node involvement is infrequent with PMBCL [5].
Pancreatic lymphoma is a rare entity. The clinical presentation of pancreatic lymphoma is usually non-specific which include abdominal pain (83%), abdominal mass (58%), weight loss (50%), and jaundice (37%). Lymphoma presenting as acute pancreatitis is seen in 12% of the cases [2]. Laboratory findings of pancreatic lymphoma are often non-diagnostic. Elevated LDH is seen in more than half of the patients. Elevated CA19.9 and CEA may also be found [10,11].
1. Imaging of pancreatic lymphoma has two different morphologic pattern Localised well-circumscribed tumoral form mimicking adenocarcinoma of pancreas.
2. Diffusely enlarged infiltrating mass which mimics pancreatitis.
Findings such as the presence of a large, relatively homogenous, hypovascular mass in the pancreatic head, in the absence of pancreatic ductal dilatation or vascular invasion are features more suggestive of lymphoma rather than adenocarcinoma [12].
The absence of peri-pancreatic collections, pseudocysts, and pancreatic necrosis may help to differentiate lymphoma from pancreatitis. Autoimmune pancreatitis, which may present with diffuse pancreatic enlargement or focal mass can also mimic pancreatic lymphoma. The presence of elevated serum IgG4 levels and imaging findings of pancreatic mass with dotted enhancement during the pancreatic phase and delayed enhancement secondary to fibrosis is in favour of autoimmune pancreatitis [13,14].
CT is the usual first line imaging for characterisation of lymphoma, to know the extent of adenopathy/mass and for staging. Usual CT findings include low attenuating mass with necrosis or cystic changes [5].
EUS is an excellent tool to characterise the pancreatic mass, to look for dilatation of duct and peripancreatic lymphadenopathy. EUS guided FNA has a sensitivity of 86.6% and specificity of 95.8% [15].
In T1 weighted MR, pancreatic lymphoma appears as homogeneous, hypointense mass (focal or diffuse) and on T2 weighted MR, fat spin echo sequence has variable signal intensity and mild homogenous enhancement on post contrast study. MR cholangiopancreatography (MRCP) images help to evaluate the pancreatic duct and biliary tree [16].
PET-CT is another useful tool. Initial PET-CT is helpful to look for extramediastinal involvement as well as staging. Later after initiating treatment PET-CT is used to assess the metabolic response to treatment or in case of mediastinal relapse [1]. Lymphomas are usually managed without surgery [1].
They respond well to anthracycline based chemotherapeutic agents. The discovery of rituximab revolutionised the treatment of NHL. Most cases show excellent outcome without requiring radiotherapy. 6-8 cycles of RCHOP regimen are the widely used regimen [2]. Many centres in the United States have now started using DA-EPOCH-R regimen (dose-adjusted etoposide, prednisolone, vincristine, cyclophosphamide, doxorubicin, rituximab) as standard of care for PMBCL due to promising results obtained in various recent studies [8].
Often, PMBCL involves the mediastinum alone. Involvement of other systems like gastrointestinal tract, kidneys, ovaries, central nervous system and liver is rare [9]. Findings such as pleural or pericardial effusion is indicative of poor prognosis. Extra mediastinal involvement is uncommon at presentation. PMBCL in our case shows both mediastinal and pancreatic masses in which mediastinal mass was asymptomatic. All clinical symptoms were pertaining to pancreatic mass. The close pathologic differentials DLBCL, classical Hodgkin lymphoma presents as cervical, mediastinal or abdominal lymphadenopathy with or without mediastinal mass. Lymphoblastic leukemia is another haematological malignancy which may present with a mediastinal mass. Lymph node involvement is infrequent with PMBCL [5].
Pancreatic lymphoma is a rare entity. The clinical presentation of pancreatic lymphoma is usually non-specific which include abdominal pain (83%), abdominal mass (58%), weight loss (50%), and jaundice (37%). Lymphoma presenting as acute pancreatitis is seen in 12% of the cases [2]. Laboratory findings of pancreatic lymphoma are often non-diagnostic. Elevated LDH is seen in more than half of the patients. Elevated CA19.9 and CEA may also be found [10,11].
1. Imaging of pancreatic lymphoma has two different morphologic pattern Localised well-circumscribed tumoral form mimicking adenocarcinoma of pancreas.
2. Diffusely enlarged infiltrating mass which mimics pancreatitis.
Findings such as the presence of a large, relatively homogenous, hypovascular mass in the pancreatic head, in the absence of pancreatic ductal dilatation or vascular invasion are features more suggestive of lymphoma rather than adenocarcinoma [12].
The absence of peri-pancreatic collections, pseudocysts, and pancreatic necrosis may help to differentiate lymphoma from pancreatitis. Autoimmune pancreatitis, which may present with diffuse pancreatic enlargement or focal mass can also mimic pancreatic lymphoma. The presence of elevated serum IgG4 levels and imaging findings of pancreatic mass with dotted enhancement during the pancreatic phase and delayed enhancement secondary to fibrosis is in favour of autoimmune pancreatitis [13,14].
CT is the usual first line imaging for characterisation of lymphoma, to know the extent of adenopathy/mass and for staging. Usual CT findings include low attenuating mass with necrosis or cystic changes [5].
EUS is an excellent tool to characterise the pancreatic mass, to look for dilatation of duct and peripancreatic lymphadenopathy. EUS guided FNA has a sensitivity of 86.6% and specificity of 95.8% [15].
In T1 weighted MR, pancreatic lymphoma appears as homogeneous, hypointense mass (focal or diffuse) and on T2 weighted MR, fat spin echo sequence has variable signal intensity and mild homogenous enhancement on post contrast study. MR cholangiopancreatography (MRCP) images help to evaluate the pancreatic duct and biliary tree [16].
PET-CT is another useful tool. Initial PET-CT is helpful to look for extramediastinal involvement as well as staging. Later after initiating treatment PET-CT is used to assess the metabolic response to treatment or in case of mediastinal relapse [1]. Lymphomas are usually managed without surgery [1].
They respond well to anthracycline based chemotherapeutic agents. The discovery of rituximab revolutionised the treatment of NHL. Most cases show excellent outcome without requiring radiotherapy. 6-8 cycles of RCHOP regimen are the widely used regimen [2]. Many centres in the United States have now started using DA-EPOCH-R regimen (dose-adjusted etoposide, prednisolone, vincristine, cyclophosphamide, doxorubicin, rituximab) as standard of care for PMBCL due to promising results obtained in various recent studies [8].
Conclusion
PMBCL is a type of NHL which usually presents as a mediastinal
mass without involvement of other systems. PMBCL with involvement
of mediastinum and pancreas presenting with pancreatitis in a young
male is a rare entity. Histopathology with immunohistochemistry is
essential for confirming the diagnosis.
Statement and Declarations:
Ethical Statement: The authors are accountable for all aspects of
the work (if applied, including full data access, integrity of the data and
the accuracy of the data analysis) in ensuring that questions related to
the accuracy or integrity of any part of the work are appropriately
investigated and resolved.References
Citation
Fathima R, Pratap T, Jacob D, Jalal MJA. Primary Mediastinal B Cell Lymphoma Presenting As Acute Abdomen. Indian J Appl Radiol. 2026;12(1): 233.