Case Report
Computed Tomography Cerebral Angiography Evaluation of Cerebral Arterial Aneurysms Case Series
Kumar K*, Raveendran C, Murugaiah V and Senthilnathan V
Department of Radio-diagnosis. Trichy SRM Medical College Hospital and Research Centre. Irungalur, Trichy, Tamil Nadu,
India
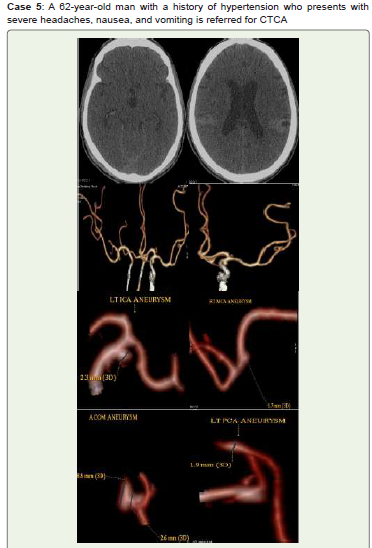
*Corresponding author:Krishna Kumar M, Department of Radio-diagnosis. Trichy SRM Medical College Hospital and Research Centre. Irungalur, Trichy, Tamil Nadu, India. E-mail Id: drmkrishnakumar@gmail.com
Copyright: © 2026 Kumar K, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information:Submission: 07/01/2026; Accepted: 15/04/2026; Published: 18/04/2026
Abstract
Objective: Saccular (berry-shaped) aneurysms that develop at arterial bifurcations—branch points where an artery splits into two branches—are the most common type of intracranial aneurysms, which are aberrant focal dilatations (localized bulges) of cerebral arteries. When they burst, cerebral artery aneurysms (CAA) pose a serious risk of morbidity and death. This case series aims to analyze computed tomography cerebral angiography (CTCA), a
medical imaging method for the screening, diagnosis, and follow-up of patients with cerebral artery aneurysms that uses CT scans to show cerebral arteries.
Conclusion: Recent years have seen a significant change in the diagnosis and treatment of cerebral aneurysms. With CTCA, radiologists can accurately describe pertinent results, such as the risk of aneurysm rupture, and reliably and noninvasively identify the majority of intracranial aneurysms. Additionally, it helps identify high-risk groups and recommend screening. Treatment is becoming safer and more efficient thanks to nonoperative endovascular treatments, which are minimally invasive operations carried out inside blood vessels.
Conclusion: Recent years have seen a significant change in the diagnosis and treatment of cerebral aneurysms. With CTCA, radiologists can accurately describe pertinent results, such as the risk of aneurysm rupture, and reliably and noninvasively identify the majority of intracranial aneurysms. Additionally, it helps identify high-risk groups and recommend screening. Treatment is becoming safer and more efficient thanks to nonoperative endovascular treatments, which are minimally invasive operations carried out inside blood vessels.
Keywords:Subarachnoid Hemorrhage; Fusiform Aneurysm; Dolichoectasia; Bilobed Aneurysm; Mycotic Aneurysm; Giant Aneurysm; Computed Tomography Cerebral Angiography
Introduction
Although the majority of cerebral artery aneurysms [CAA]
may not cause any symptoms over a patient’s lifetime, ruptures
can have disastrous consequences. About 80–90% of nontraumatic
subarachnoid hemorrhages (SAH) are caused by
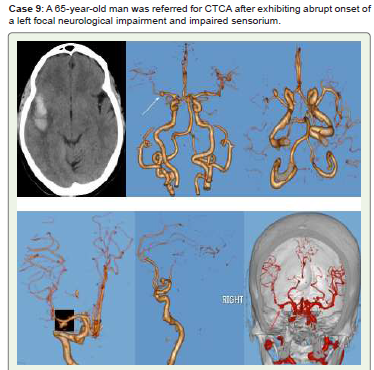
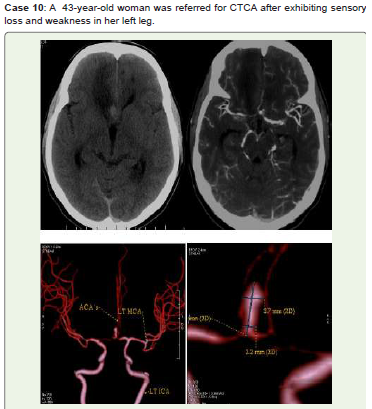
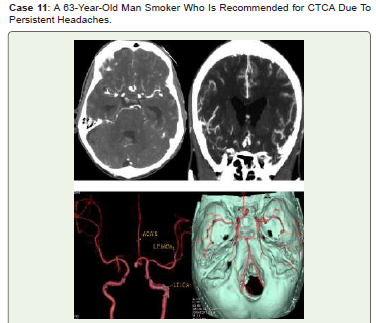
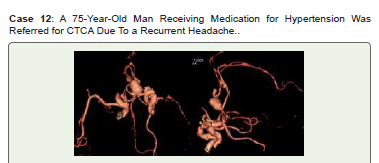
them (Figures 1A, 2A, 2B, 3A, 4A, 4B, 5A, 5B, 6A, 6B, and 7A).
Position, size, high-danger characteristics (irregular
protrusions and/or daughter sacs), patient characteristics
(age, gender, race), and extra risk factors (hypertension, prior
SAH, smoking) all influence the rupture hazard of CAA [1].
Saccular (berry) aneurysms are the most common type of intracranial
aneurysms, and they are found in predictable locations around the
circle of Willis. However, uncommon aneurysm types can occasionally
be found, such as dissecting, fusiform, serpentine, blood blister type,
traumatic, mycotic (or infectious), atheromatous, and enormous
aneurysms, all of which can cause focal bleeding (Figure. 1A, Figure
8A, 9A), thromboembolic episodes, or bulk effect [2] (Figure. 10A).
Conventional neurovascular imaging techniques such as CTCA,
magnetic resonance angiography (MRA), and digital subtraction
angiography (DSA) can be used to assess CAA, morphologic
features, and post-treatment appearance. The diagnostic accuracy
of noninvasive imaging for initial opinion and surveillance has
improved with to advancements in computed tomography and CTCA,
transcranial Doppler sonography, and MRA, even if DSA is still the
gold standard for CAA characterisation and treatment planning.
Hospitalization for aneurysmal subarachnoid hemorrhage decreased
slightly but significantly with increased use of neuroimaging and
treatment of unruptured CAA.
We examine the CTCA imaging methods and manifestations of CAA in this patient population’s assessment, monitoring, management, and results.
We examine the CTCA imaging methods and manifestations of CAA in this patient population’s assessment, monitoring, management, and results.
Discussion
Approximately 4% of people have intracranial aneurysms,
making them fairly common. The majority of these aneurysms are
asymptomatic, but they do present a slight but real risk of rupturing
and causing acute SAH [3].
The internal elastic lamina and a weakened or absent muscularis layer are typically the two layers of the normal artery wall that are lacking in all real cerebral aneurysms. All layers of the artery wall are lacking in intracranial pseudoaneurysms, which are typically cavitated paravascular hematomas that may or may not connect directly with the real arterial lumen. The “blood blister-like” aneurysm is uncommon yet deadly because of its tissue-paper thin wall. Round or lobulated focal outpouchings known as saccular aneurysms typically develop at major artery bifurcations, which are regions of severe hemodynamic stress [3].
In the arteries of the circle of Willis, saccular aneurysms usually affect the following arteries in order of frequency [4]. The middle cerebral artery (20%) (Figure1, Figure 2, Figure 9, Figure 13, Figure 24), the anterior communicating artery (30%) (Figure 3, Figure 4, Figure 7, Figure 10, Figure 11, Figure 12, Figure 23), the internal carotid artery (8%) (Figure 6, Figure 13, Figure 14, Figure 15, Figure 16, Figure 17, Figure 18, Figure 20), and the tip of the basilar artery (Figure 21), Pericallosal anterior cerebral artery (4%) (Figure 8, 12) and posterior inferior cerebellar artery (3%). Fusiform aneurysms (Dolichoectasia) (Figure 25) are long, spindle-shaped arterial dilations that may be linked to non-atherosclerotic pathology, such as connective tissue disorders, or atherosclerotic vascular disease [3]. Ectatic sinuous cerebral arteries, which are most commonly found in the vertebrobasilar system, give rise to dolicoectasia, which can have a diameter of several centimeters. Although symptoms of brainstem or cranial-nerve compression are typically present in these situations, they are not typically linked to SAH [5]. A ruptured artery or cystic medial necrosis are the causes of dissecting aneurysms. Thirty percent of affected people develop multiple aneurysms [5] - According to the Unruptured Cerebral Aneurysm Study [6], aneurysms should be classified as small (<5 mm), medium (>5-10 mm), large (>10 mm-25 mm), and giant (>25 mm) based on the maximum dome diameter. A neck diameter greater than 4 mm or a dome-to-neck ratio less than 2 were indicative of a wide-neck aneurysm.
Giant fusiform aneurysms are thought to be less common than 1% of all intracranial aneurysms, while “giant aneurysms”- intracranial aneurysms with a diameter more than 25 mm-represent between 3-5% of all aneurysms. A gigantic aneurysm presentation is characterized by the presence of a mural thrombus, cerebral bleeding episodes, and expansion symptoms [7].
The internal elastic lamina and a weakened or absent muscularis layer are typically the two layers of the normal artery wall that are lacking in all real cerebral aneurysms. All layers of the artery wall are lacking in intracranial pseudoaneurysms, which are typically cavitated paravascular hematomas that may or may not connect directly with the real arterial lumen. The “blood blister-like” aneurysm is uncommon yet deadly because of its tissue-paper thin wall. Round or lobulated focal outpouchings known as saccular aneurysms typically develop at major artery bifurcations, which are regions of severe hemodynamic stress [3].
In the arteries of the circle of Willis, saccular aneurysms usually affect the following arteries in order of frequency [4]. The middle cerebral artery (20%) (Figure1, Figure 2, Figure 9, Figure 13, Figure 24), the anterior communicating artery (30%) (Figure 3, Figure 4, Figure 7, Figure 10, Figure 11, Figure 12, Figure 23), the internal carotid artery (8%) (Figure 6, Figure 13, Figure 14, Figure 15, Figure 16, Figure 17, Figure 18, Figure 20), and the tip of the basilar artery (Figure 21), Pericallosal anterior cerebral artery (4%) (Figure 8, 12) and posterior inferior cerebellar artery (3%). Fusiform aneurysms (Dolichoectasia) (Figure 25) are long, spindle-shaped arterial dilations that may be linked to non-atherosclerotic pathology, such as connective tissue disorders, or atherosclerotic vascular disease [3]. Ectatic sinuous cerebral arteries, which are most commonly found in the vertebrobasilar system, give rise to dolicoectasia, which can have a diameter of several centimeters. Although symptoms of brainstem or cranial-nerve compression are typically present in these situations, they are not typically linked to SAH [5]. A ruptured artery or cystic medial necrosis are the causes of dissecting aneurysms. Thirty percent of affected people develop multiple aneurysms [5] - According to the Unruptured Cerebral Aneurysm Study [6], aneurysms should be classified as small (<5 mm), medium (>5-10 mm), large (>10 mm-25 mm), and giant (>25 mm) based on the maximum dome diameter. A neck diameter greater than 4 mm or a dome-to-neck ratio less than 2 were indicative of a wide-neck aneurysm.
Giant fusiform aneurysms are thought to be less common than 1% of all intracranial aneurysms, while “giant aneurysms”- intracranial aneurysms with a diameter more than 25 mm-represent between 3-5% of all aneurysms. A gigantic aneurysm presentation is characterized by the presence of a mural thrombus, cerebral bleeding episodes, and expansion symptoms [7].
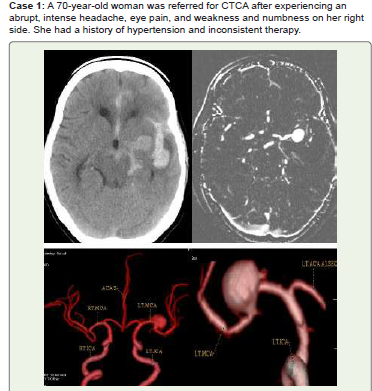
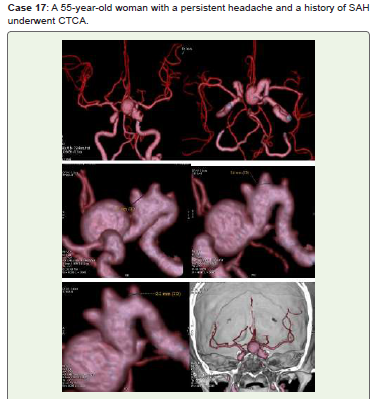
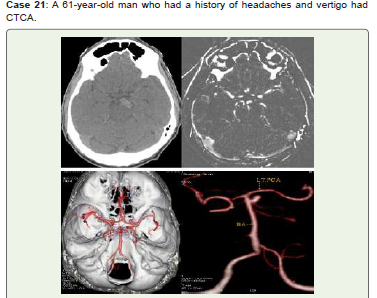
Figure 1:Acute SAH is visible along the anterior interhemispheric fissure,
left sylvian fissure, interpeduncular cistern, left ambient cistern, and left
basal ganglia hemorrhage on noncontrast CT (A). A significant saccular
aneurysm is visible at the left middle cerebral artery (MCA) bifurcation in
CTCA maximum intensity projection (MIP) (B) and 3D volume rendered
images (3D-VR) (C, D). She received medical care and was recommended
for endovascular coiling or surgical clipping.
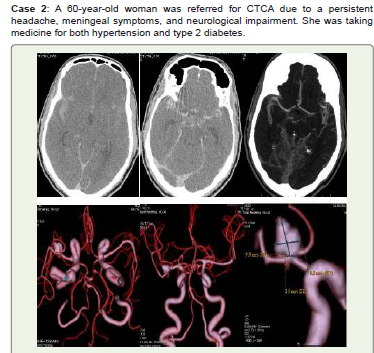
Figure 2:Acute SAH is visible in basal cisterns, sylvian fissures,
interhemispheric fissures, and along the tentorium cerebelli on noncontrast
CT (A, B). A medium multilobed Saccular aneurysm measuring 9.5x6 mm
with a neck diameter of 3.5 mm is shown at the right MCA bifurcation in
CTCA (C) and 3D-VR images (D, E, and F). She had conservative care and
was recommended for endovascular therapy and /or surgical clipping.
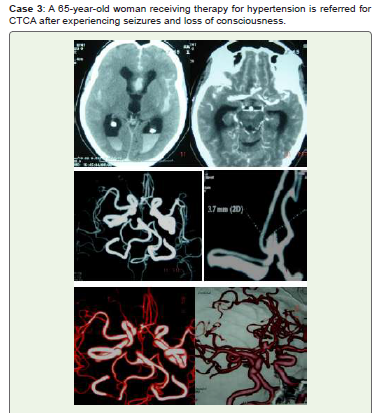
Figure 3:Noncontract CT (A) reveals intraventricular hemorrhage in the
posterior horns of lateral ventricles as well as acute SAH in Sylvian fissures,
interhemispheric fissures, and along cerebral sulci. Anterior communicating
artery (ACOM) saccular aneurysm is visible on CTCA (B), MIP (C, D), and
3D-VR (E, F) images. During conservative treatment, the patient passed away.
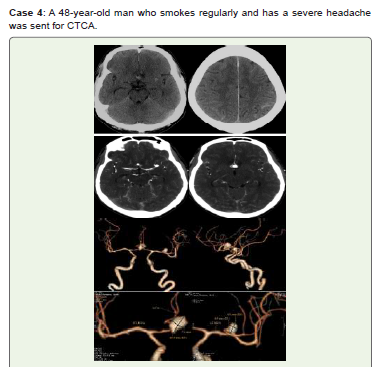
Figure 4:Acute SAH is visible at the tentorium cerebelli and cerebral sulci
interhemispheric fissure on noncontrast CT (A, B). Multilobed medium ACOM
saccular aneurysms measuring 10.8x7.6 mm and left MCA bifurcation small
saccular aneurysms measuring 6.1x4.9 mm are visible in CTCA axial (C, D)
and 3D-VR images (E–H). He received conservative care and did not return
for a follow-up.
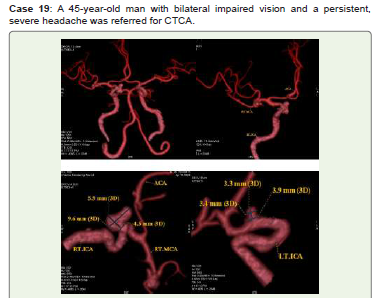
Figure 5:Acute SAH is seen on noncontrast CT (A, B). Multiple saccular
aneurysms of intracranial left clinoid ICA (C5), ACOM, right MCA, and
fusiform aneurysm of left posterior cerebral artery (PCA) are seen on CTCA
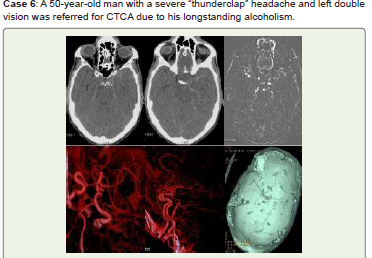
3D-VR images (C-H). Managed conservatively & on regular follow up.
Figure 6:(A-F). Acute SAH is visible along the left parasellar cistern,
suprasellar cistern, and tentorium cerebelli on noncontrast CT (A, B). The
left intracavernous ICA (C4) saccular aneurysm is seen in CTCA MIP (C)
and 3D-VR pictures (D-F). Endovascular therapy was recommended for him.
Figure 7:Noncontrast CT (A), shows acute SAH. A medium saccular
aneurysm of ACOM with a neck diameter of 3.4 mm is visible on CTCA (B-D),
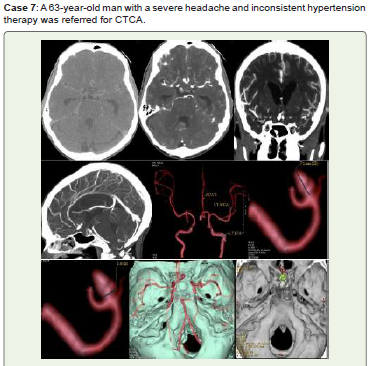
3D-VR pictures (E-G), and 3D-VR virtual dissection (H, I).
Endovascular coiling and/or surgical clipping were recommended for him.
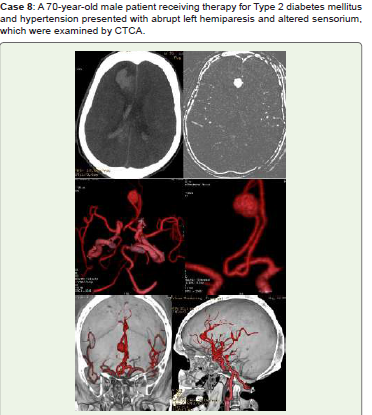
Figure 8:Noncontrast axial (A) show Right frontal lobe lobulated hematoma
with surrounding edema, acute SAH and intraventricular hemorrhage, MIP
(B), 3D-VR (C, D), and virtual dissection of skull images (E, F) all show a
large right precallosal ACA (A3) saccular aneurysm projecting to the right.
He received conservative treatment and advised endovascular coiling and/
or surgical clipping, and frequent follow-up.
Figure 9:Right temporal lobe lobulated hematoma with surrounding edema
is visible on CT axial non-contrast (A), whereas right MCA (M1) saccular
aneurysm projecting inferiorly is visible on 3D-VR (B-E) and virtual dissection
(F) images. Treated consevatively & on regular follow up.
Figure 10:Right frontal lobe infarct is seen on CT Axial non-contrast (A),
MIP (B), and 3D-VR images (C, D) show a large ACOM saccular aneurysm
with a neck diameter of 2.2 mm and a high ellipticity index, squeezing the
right ACA’s A2 segment and generating a mass effect. He was scheduled
for immediate endovascular treatment but did not show up for the follow-up.
Figure 11:ACOM saccular aneurysm extending anteriorly is seen in CTCA
axial (A), coronal (B), 3D-VR (C), and virtual dissection of skull vault pictures
(D). He is routinely monitored.
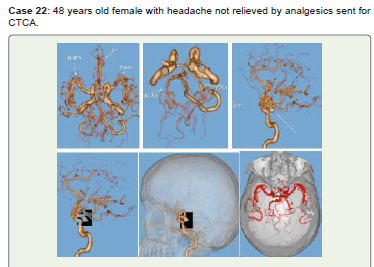
Figure 12:Large saccular aneurysms of ACOM and smaller saccular
aneurysms of bilateral A2 segments of ACA are shown in CTCA 3D-VR
pictures. He is being monitored on a regular basis.
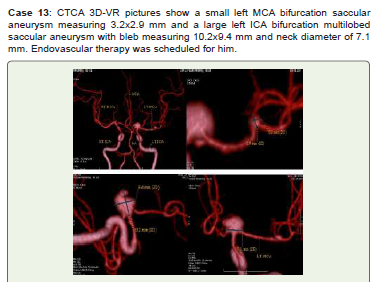
Figure 13:CTCA 3D-VR pictures show a small left MCA bifurcation saccular
aneurysm measuring 3.2x2.9 mm and a large left ICA bifurcation multilobed
saccular aneurysm with bleb measuring 10.2x9.4 mm and neck diameter of
7.1 mm. Endovascular therapy was scheduled for him.
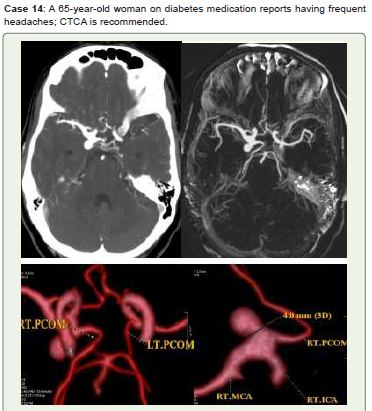
Figure 14:CTCA Axial (A), MIP (B), and 3D-VR images (C, D) show a medium
saccular aneurysm with a neck diameter of 4 mm at the intersection of the right
ICA bifurcation and the origin of the posterior communicating artery (PCOM).
Endovascular therapy and follow-up were recommended for her.
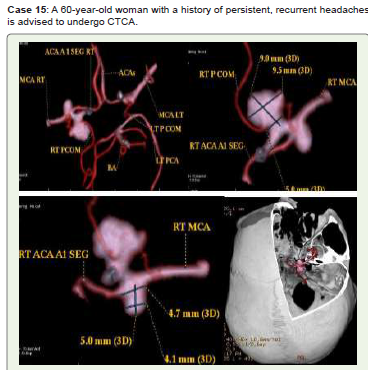
Figure 15:CTCA 3D-VR pictures (A–C) and virtual dissected skull bone
(D) show a small saccular aneurysm with a neck diameter of 4.7 mm that
originates from the right ICA bifurcation and measures 5 x 4.1 mm. Another
medium multilobed saccular aneurysm, measuring 9.5 x 9 mm with a neck
diameter of 5 mm, originated at the intersection of the right PCOM and MCA.
Endovascular therapy and further monitoring were recommended for her.
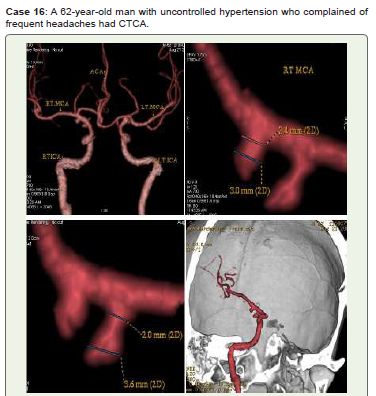
Figure 16:Two medium saccular aneurysms with a high ellipticity index in the
right distal communicating ICA (C7) with neck diameters of 2 mm and 2.4 mm,
respectively, are seen in CTCA 3D-VR (A-C) and virtual dissected skull vault
(D) pictures. Endovascular therapy was recommended for him.
Figure 17:Left ICA bifurcation large saccular aneurysm and three small
saccular aneurysms of terminal communicating ICA (C7) with neck diameters
of 3.7 mm, 3.6 mm, and 2.2 mm are visible in CTCA 3D-VR (A-E) and virtual
dissection of skull (F) pictures. Endovascular therapy and follow-up were
recommended for her.
Figure 18:CT Noncontrast axial (A), CTCA MIP (B), and virtual dissection
of the skull (C) images show a left supraclinoid ophthalmic ICA (C6) small
saccular aneurysm. Endovascular therapy and routine follow-up were
recommended for him.
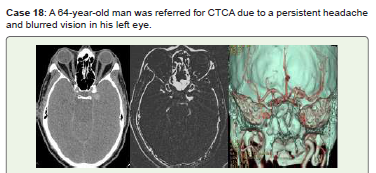
Figure 19:Bilateral distal ophthalmic ICA(C6) saccular aneurysms measuring
right medium 9.6x5.9 mm with neck diameter of 4.5 mm and left small 3.3x3.4
mm with neck diameter of 3.9 mm are shown on CTCA 3D-VR (A-D) images.
Endovascular treatment was recommended for him, but he did not show up
for the follow-up.
Figure 20:CTCA 3D-VR (A-D) pictures show a right petrous ICA (C2) small
saccular aneurysm measuring 3.1x4.5 mm and a left ostio-proximal small
PCOM saccular aneurysm measuring 2.7x24 mm with a neck diameter of 2.8
mm. Endovascular therapy was scheduled for him.
Figure 21:CT Axial non-contrast (A) reveals linear hyperdensity anterior
to midbrain on left side, MIP (B), Virtual dissection of skull (C) and 3D -VR
(D) images reveal distal basilar artery saccular aneurysm with high ellipticity
index projecting to the left side. He was advised endovascular treatment & on
interval follow up.
Figure 22:CTCA 3D-VR (A-D) and virtual dissection of the skull (E, F) images
show an inferiorly projecting left ostio-proximal PCOM saccular aneurysm.
Endovascular therapy and routine follow-up were recommended for her.
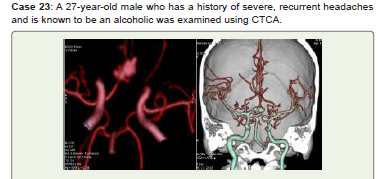
Figure 23:A multilobed medium saccular aneurysm is visible at the
intersection of the A1 segment of the right ACA and ACOM in CTCA 3D-VR
(A) and virtual bone dissection (B) pictures. Endovascular coiling and/or
neurosurgical clipping were recommended for him.
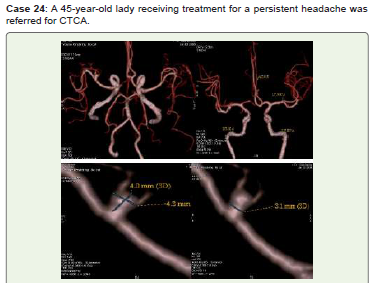
Figure 24:A small saccular aneurysm of the M1 segment of the right MCA,
measuring approximately 4x4.3 mm with a neck diameter of 3.1 mm, is visible
in CTCA 3D-VR (A-D) pictures. Surgical clipping and/or endovascular coiling
were recommended, and she was lost for follow-up.
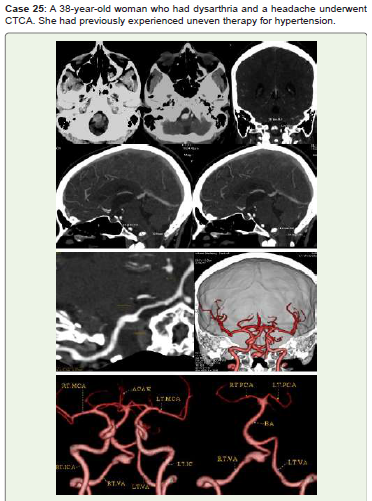
Figure 25:An oval hyperdensity is visible anterior to the medulla in CT axial
non-contrast (A). A large right vertebral fusiform aneurysm (Dolichoectasia)
measuring 12.8x12.8x11.9 mm with a posterior intramural thrombus measuring
11.1x5.9 mm is visible in axial, coronal, and sagittal contrast images (B-F),
virtual dissection (G), and 3D-VR images (H, I). Endovascular therapy and
further monitoring were recommended for her.
Microaneurysms, also called Charcot-Bouchard aneurysms, are
linked to persistent hypertension and usually develop in small blood
arteries (less than 300 micrometers in diameter), most frequently the
lenticulo-striate vessels of the basal ganglia [8]. Intracranial bleeding
is frequently caused by Charcot-Bouchard aneurysms [9].
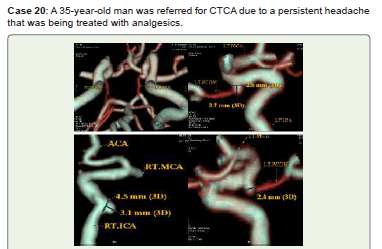
A family history of aneurysms, a variety of hereditary illnesses, age above 50, female gender, present cigarette smoking, and cocaine usage are all contributing factors to the development of aneurysms. Rare causes of intracranial aneurysms include head trauma, intracranial tumors or neoplastic emboli, and infections from bacterial or fungal colonization of artery walls [10].
Intracranial aneurysms have been linked to fibromuscular dysplasia, coarctation of the aorta, and pheochromocytoma, most likely due to the elevated blood pressure that these disorders induce [11].
A family history of aneurysms, a variety of hereditary illnesses, age above 50, female gender, present cigarette smoking, and cocaine usage are all contributing factors to the development of aneurysms. Rare causes of intracranial aneurysms include head trauma, intracranial tumors or neoplastic emboli, and infections from bacterial or fungal colonization of artery walls [10].
Intracranial aneurysms have been linked to fibromuscular dysplasia, coarctation of the aorta, and pheochromocytoma, most likely due to the elevated blood pressure that these disorders induce [11].
SAH is 1.6 times more prevalent in women, making them more
vulnerable to aneurysm rupture [12-14]. Only 10–15% of cerebral
aneurysms exhibit symptoms [15, 16], with the majority being
discovered by chance while being evaluated for other ailments.
The bulk effect or possibly a little amount of blood leakage that
irritates the meninges but is insufficient to qualify as a hemorrhage
are the main causes of the CAA symptoms [12, 17]. Given that
10% to 43% of SAH sufferers report having a sentinel headache
two months prior, these symptoms could be an early warning sign
of an imminent rupture.preceding the rupture [18]. SAH patients
typically have poor clinical conditions, which might include severe
headaches, disorientation, neurological impairments, and even coma.
With a 25% to 50% death rate, SAH is a terrible occurrence. Only
about one-third of cases have a favorable outcome since over 50% of
survivors experience permanent impairment [19].
According to statistics from the International Study of Unruptured
Intracranial Aneurysms (ISUIA), aneurysms with a diameter of 10
mm or greater are critically vulnerable to rupture. The average annual
rupture rate for aneurysms smaller than 10 mm in diameter without
a history of SAH was 0.05%; however, the rupture rate was ten times
higher for aneurysms of the same size with a history of SAH [20].
For the detection of cerebral aneurysms, a recent study using CTCA
with a multidetector row scanner found a sensitivity of 95.1% to 98%
and a specificity of 100%. The published series shown that DSA is still
more sensitive than CTCA in detecting aneurysms less than 3 mm [21,
22]. In addition to screening, CTCA may identify the morphometric
features of intracranial aneurysms, such as the sizes of the sac and
neck as well as the existence of daughter sacs and leaving branches.
An ECG-gated, time-resolved, volumetric imaging sequence, fourdimensional
(4D) CTCA generates angiographic images using patientspecific
average input rates with a rather excellent spatial resolution
(≈0.5 mm³) [23]. The method is being applied more frequently
to improve hemodynamic assessments of brain aneurysms using
computational fluid dynamics (CFD). 4D CTCA has been shown in
several investigations to detect specific regions of abnormal aneurysm
wall motion, which may indicate a higher risk of rupture [24].
For cerebral aneurysms greater than 3 mm, the three-dimensional
(3D) time-of-flight (TOF) MRA sensitivity is 93%–97%, while for
aneurysms smaller than 3 mm, it is 85%-93% [25].
To see cerebral arteries and their aneurysms, 4D MR employs
gadolinium contrast and TOF sequences. 4D MR uses postprocessing
techniques and ECG-synchronized 3-D phase-contrast
MRI to noninvasively measure 3-D intracranial blood flow [26]. In
specific regions of interest, 4D MR may evaluate blood flow volumes,
flow velocities, and wall shear stress (WSS). A number of modest
investigations have shown that aberrant WSS may be linked to thin
aneurysm walls and aneurysm development [27], which could have
therapeutic implications.
Research indicates that an inflammatory mechanism that
results in artery wall remodeling, atherosclerotic conformation, and
aneurysm growth is the cause of aneurysms [28]. Focal enhancement
aneurysms are more likely to be bigger [28] and have a higher rupture
risk score [29].
When evaluating unstable unruptured intracranial aneurysms,
MR high-resolution vascular wall imaging (HR-VWI) has emerged
as a valuable tool. The evaluation of aneurysm wall enhancement as
a biomarker of aneurysm wall inflammation, aneurysm growth, and
rupture shows encouraging compliance [30]. According to data, there
is a significant negative predictive value for the absence of aneurysm
wall augmentation. When there are several intracranial aneurysms,
MR HR-VWI can assist in identifying the ruptured aneurysm
and may even lead to rupture risk classification during aneurysm
surveillance [31].
With 3D rotating DSA, the operator may see the aneurysm’s
size, shape, and neck as well as the parent-vessel properties in more
detail. Additionally, this method is helpful in obtaining suitable
working angles for potential endovascular expected anatomy and
embolization following open surgical surgery [32, 33]. With the
exception of vascular overlap, 4D DSA allows 3D viewing from any
chosen spatial projection at any moment throughout the passage of
the contrast bolus for aneurysm investigation [34].
Due to early IVUS catheters’ enormous size and poor navigability,
intravascular ultrasonography (IVUS) has been limited in its
ability to evaluate cerebral disease. Furthermore, IVUS is known
to overestimate the true lumen’s size, which could be caused by
inadequate lumen–intima interface imaging [35].
An alternative intravascular imaging method called optical
coherence tomography (OCT) employs interferometry and light
backscatter to see the architecture of the artery wall and lumen with
a spatial resolution ten times greater than that of DSA or IVUS
[36]. OCT can scan lumen size, intimal flaps, patency of perforators
and tiny branches, stent apposition, thrombus development,
endothelialization, and all layers of the artery wall with an axial
resolution of 10 to 20 μm, which may be useful in predicting aneurysm
rupture [37].
The 2D and 3D morphology of aneurysms can be evaluated in
a variety of ways, such as aneurysm width, perpendicular height,
maximum height (from the midpoint of the neck), numerous lobes,
blebs, aneurysm angle, aneurysm volume, neck width aspect ratio
(height divided by neck width), and parent-vessel diameter size ratio
(maximum diameter to parent-vessel diameter) [38].
The following suggestions are included in the American Heart
Association [39] consensus guidelines for the standardized reporting
on imaging for intracranial aneurysms and endovascular repair:
1. When there are several aneurysms, each one needs to be
explained separately.
2. High-resolution planar reconstructed images are used to
provide aneurysm dome measures in millimeters in three orthogonal
planes. To prevent attenuating the precise lumen size and morphology,
the source images shouldn’t be over-windowed.
3. The location of the aneurysm is described on the base of the
neck origin vessel and, often, the nearest branchship. For example,
the closest branch vessel, such as the meningo-hypophyseal trunk,
superior hypophyseal, ophthalmic, anterior choroidal, or posterior
communicating artery, or position at the carotid terminus, should
be used to name ICA aneurysms. Bony landmarks, such as the
supraclinoid, should not be used for description since they are less
accurate.
4. Although additional coordinate planes (anterior or posterior,
superior or inferior, medial or lateral) are also frequently used,
aneurysm orientation can be characterized as right of midline,
midline, or left of midline. It is also necessary to discuss the impacts
of mass on nearby structures.
5. Neck sizes smaller than 4 mm and aneurysm dome-to-neck
ratios larger than 2:1 indicate aneurysm morphology that can be
coiled. The neck’s dimensions in comparison to the parent vessel
diameter is also an important determinant for use of intraluminal
adjunctive devices or flow diverters.
When using flow diverters or intraluminal adjunctive devices, vessel diameter is another crucial factor. The best way to visualize complex aneurysm morphology is via 3D volume-rendering software. The three management approaches are surgical clipping, endovascular coiling, and observation (Figure 26). Fusiform aneurysms are currently treated using a variety of techniques, including bypasses, vascular stents, wrapping, and trapping. Treatment for cerebral aneurysms is becoming safer and more efficient thanks to nonoperative endovascular methods. It is debatable what should be done when cerebral aneurysms remain unruptured. The initial interval imaging is typically obtained after three to six months of treatment, since most aneurysm recurrences happen in the first year after treatment [40]. The subsequent picture is taken between six months and a year later if it is stable at that point. Imaging may be stretched out to intervals of one to five years if the aneurysm continues to show stability. Higher-threat aneurysms require a longer (perhaps lifetime) follow-up period [41]. The gold standard for vascular imaging, DSA is typically chosen for the first follow-up imaging following aneurysm therapy. For long-term surveillance, MRA is the preferred noninvasive imaging technique. Since gadolinium contrast carries a relatively low risk [39], CE-MRA is typically chosen. The acquisition duration is quicker than TOF-MRA, and TOF-MRA is less sensitive for aneurysms that perfuse slowly [42]. Because there is less susceptibility artifact associated with stents and flow diverters with high metal surface coverage and flow-related signal loss is eliminated by the use of contrast material, the CE-MRA exhibits better detection of post-treatment intracranial aneurysm occlusion, parent vessel visualization, and luminal measurements even though its inherent spatial resolution is lower than that of the 3D TOF technique [43]. Although CTCA is an inexpensive, noninvasive substitute for MRI, beam hardening artifacts from clips, coils, and stents severely impair the image quality [44].
When using flow diverters or intraluminal adjunctive devices, vessel diameter is another crucial factor. The best way to visualize complex aneurysm morphology is via 3D volume-rendering software. The three management approaches are surgical clipping, endovascular coiling, and observation (Figure 26). Fusiform aneurysms are currently treated using a variety of techniques, including bypasses, vascular stents, wrapping, and trapping. Treatment for cerebral aneurysms is becoming safer and more efficient thanks to nonoperative endovascular methods. It is debatable what should be done when cerebral aneurysms remain unruptured. The initial interval imaging is typically obtained after three to six months of treatment, since most aneurysm recurrences happen in the first year after treatment [40]. The subsequent picture is taken between six months and a year later if it is stable at that point. Imaging may be stretched out to intervals of one to five years if the aneurysm continues to show stability. Higher-threat aneurysms require a longer (perhaps lifetime) follow-up period [41]. The gold standard for vascular imaging, DSA is typically chosen for the first follow-up imaging following aneurysm therapy. For long-term surveillance, MRA is the preferred noninvasive imaging technique. Since gadolinium contrast carries a relatively low risk [39], CE-MRA is typically chosen. The acquisition duration is quicker than TOF-MRA, and TOF-MRA is less sensitive for aneurysms that perfuse slowly [42]. Because there is less susceptibility artifact associated with stents and flow diverters with high metal surface coverage and flow-related signal loss is eliminated by the use of contrast material, the CE-MRA exhibits better detection of post-treatment intracranial aneurysm occlusion, parent vessel visualization, and luminal measurements even though its inherent spatial resolution is lower than that of the 3D TOF technique [43]. Although CTCA is an inexpensive, noninvasive substitute for MRI, beam hardening artifacts from clips, coils, and stents severely impair the image quality [44].
Conclusion
The diagnosis, monitoring, therapy, and post-treatment
monitoring of cerebral aneurysms all depend on imaging. In order
to more precisely guide implicit treatment, it is crucial to accurately
assess the size, morphology, position, and rupture status of a cerebral
aneurysm as well as to identify certain imaging characteristics that may
indicate a higher risk of rupture. The majority of intracranial aneurysm
morphologies may be accurately and noninvasively diagnosed by
radiologists using CTCA quickly in emergency room setting, which
is necessary to select the best course of action. Therefore, it seems
that employing CTCA as the main imaging technique for SAH is a
successful strategy, and DSA should only be used in situations when
there is doubt. Non-invasive imaging techniques like as transcranial
Doppler sonography, CTCA, and MRA are more suitable for serially
monitoring aneurysms because of invasive angiography’s risks.
Treatment for cerebral aneurysms is becoming safer and more
efficient thanks to nonoperative endovascular procedures.
References
Citation
Kumar K, Raveendran C, Murugaiah V, Senthilnathan V. Computed Tomography Cerebral Angiography Evaluation of Cerebral Arterial Aneurysms-Case Series. Indian J Appl Radiol. 2026;12(1): 231.