Research Article
Compressed and Overlooked: A Radiologic-Clinical Overview of Abdominal Vascular Compression Syndromes
Akhil. M. Kulkarni1,2, Pranavi Kondaveeti2, and Suhasini Vittal Rao1*
1Consultant fetal medicine specialist, Davangere scan centre, Davangere, Karnataka, India.
2Department of Radiology, SSIMS & RC, Davangere, Karnataka, India.
2Department of Radiology, SSIMS & RC, Davangere, Karnataka, India.
*Corresponding author:Suhasini Vittal Rao, Consultant fetal medicine specialist, Davangere Scan Centre, Davangere, Karnataka, India, E-mail: drsuhasini2010@gmail.com
Copyright: © 2025 Akhil. M. Kulkarni, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information:Submission: 16/06/2025; Accepted: 05/08/2025; Published: 06/08/2025
Abstract
Abdominal vascular compression syndromes constitute a collection of infrequent but clinically important conditions caused by external compression of abdominal vessels or by vascular structures exerting pressure on hollow organs. Key syndromes include Median Arcuate Ligament Syndrome (MALS),
Nutcracker Syndrome (NCS), Superior Mesenteric Artery (SMA) syndrome, SMA-like syndrome, Retrocaval Ureter & Portal Biliopathy. These conditions are often underrecognized, as they can present with vague abdominal or urinary symptoms, while many affected individuals remain symptom-free. This review
focuses on essential CT imaging findings and clinical correlations that assist in diagnosing symptomatic patients.
Keywords:Vascular Compression Syndromes; MALS; Nutcracker Syndrome; SMA Syndrome; SMA Like Syndrome; Retrocaval Ureter; Portal Biliopathy
Introduction
Abdominal vascular structures may be subjected to compression
by surrounding anatomical elements, or conversely, may compress
adjacent hollow organs. The resulting symptoms tend to be
nonspecific, leading frequently to diagnostic delays or misdiagnoses.
Although these syndromes have been recognized for several decades,
they remain insufficiently understood. Untreated cases risk significant
complications. Because these conditions span multiple specialties,
they present substantial diagnostic challenges, underscoring the need
for heightened clinical awareness and recognition[1].
Anatomical variants or morphological changes predisposing
to vascular compression are sometimes incidentally detected in
asymptomatic patients undergoing imaging for unrelated reasons.
Therefore, clinical correlation is critical to avoid overdiagnosis, and
imaging findings alone should not be the basis for diagnosis[1].
Non-invasive imaging techniques like ultrasound, computed
tomography (CT), and magnetic resonance imaging (MRI) are useful
in identifying vascular compression syndromes affecting the abdomen
and pelvis. Of these, contrast-enhanced CT is typically the modality
of choice when clinical suspicion arises, owing to its ability to clearly
depict vascular structures and their anatomical relationships with
surrounding tissues[1,2].
This review outlines the underlying pathophysiology and
clinical manifestations of various abdominal vascular compression
syndromes, emphasizing key CT imaging findings that assist in
establishing a precise diagnosis.
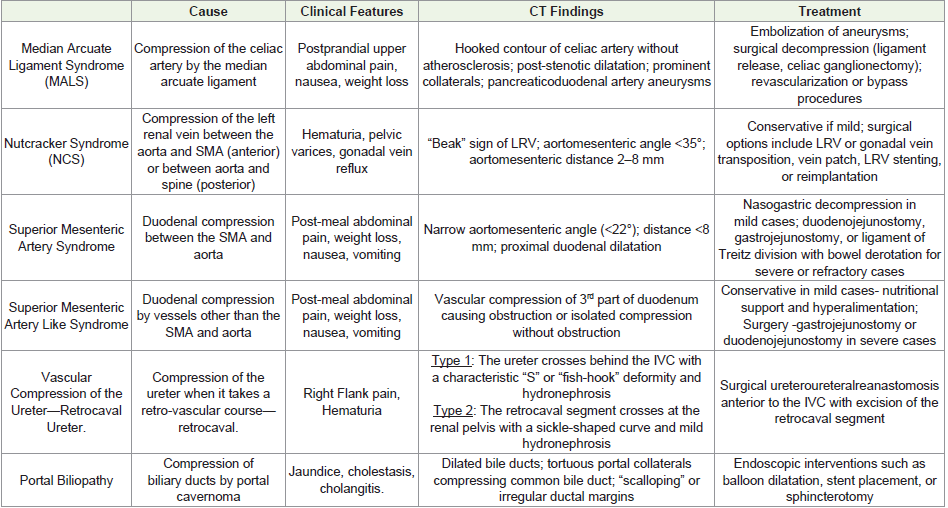
Median arcuate ligament syndrome:
Median Arcuate Ligament Syndrome (MALS), also referred to
as Dunbar syndrome or celiac artery compression syndrome, was
first described by Harjola in 1963 [1,3]. This condition arises from a
fibrous arch known as the median arcuate ligament, which connects
the diaphragmatic crura at the T12–L1 vertebral level and usually
courses above the origin of the celiac artery [4]In 10–24% of asymptomatic individuals, this ligament crosses
anteriorly over the proximal celiac artery [5]. While many such
compressions are clinically silent, a subset results in sufficient arterial
stenosis to impede blood flow and provoke symptoms [4].
Clinical features:
This syndrome most often affects individuals between 20 and 40
years of age, typically women with a lean body build [6]. The main
symptoms are chronic epigastric discomfort after meals, nausea, and
weight loss-signs of intermittent compression of the celiac artery [1].
Nonetheless, nearly 85% of those with this anatomical pattern do
not exhibit symptoms, and findings are often discovered incidentally
during CT imaging for unrelated issues [1,7,8] [Figure 2].CT imaging findings:
Multidetector CT (MDCT), due to its high spatial resolution,
enables clear visualization of the median arcuate ligament—thickness
beyond 4 mm is considered abnormal [8] [Figure 1B]. Since axial
images may not reveal the full extent of compression, sagittal
reconstructions are essential to assess both the ligament and celiac
artery origin. A distinctive hooked or “J-shaped” narrowing at the
origin of the celiac artery (Figure1A) serves as a key imaging sign
distinguishing MALS from atherosclerotic changes. The compression
is often persistent, even during inspiration. Additional findings may
include post-stenotic dilatation and development of collateral vessels
such as the pancreaticoduodenal arcade from the superior mesenteric
artery [9].Management:
Symptomatic cases of MALS are usually treated by surgically
releasing the median arcuate ligament, sometimes accompanied
by celiac ganglion removal or vascular bypass procedures. These
interventions generally lead to long-term relief from symptoms [10].Nutcracker Syndrome:
Nutcracker Syndrome refers to the entrapment of the left renal
vein (LRV), most commonly between the superior mesenteric artery
and the aorta, known as the “anterior nutcracker” variant. Less
frequently, compression occurs between the aorta and the vertebral
column when the LRV follows a retro-aortic or circum-aortic
course—termed the “posterior nutcracker” [1].It primarily affects otherwise healthy, thin women in their third
and fourth decade of life. The anterior variant is more common,
whereas the posterior variant (Figure 5) is rare and its clinical
relevance remains unclear [10].
Clinical features:
Nutcracker syndrome often presents in young to middle aged
adults, with women being more frequently affected. Because
diagnostic criteria are not well standardized, diagnosis is frequently
missed or delayed. Symptoms result from increased venous pressure in
the LRV and may be exacerbated by physical activity. Manifestations
range from microscopic haematuria to visible haematuria leading
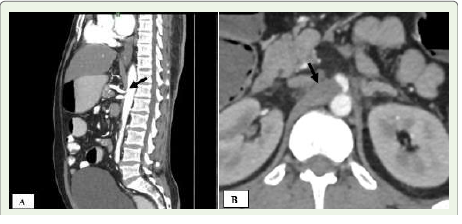
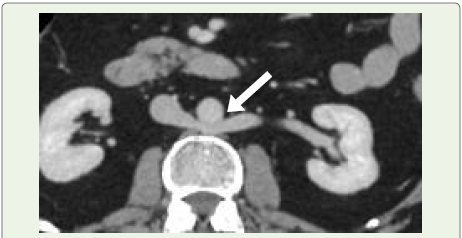
Figure 1A and B:A 49-year-old man presenting with abdominal pain after
meals. (A) Sagittal contrast-enhanced CT angiogram of the abdomen
reveals a prominent median arcuate ligament (black arrow), causing a
hooked deformity and narrowing at the proximal celiac artery with poststenotic
dilatation. (B) Axial contrast-enhanced CT angiogram demonstrates
the celiac artery origin being compressed by the thickened median arcuate
ligament (black arrow).
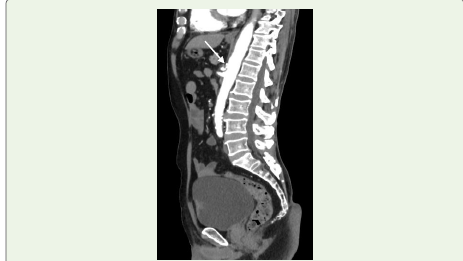
Figure 2:A 77-year-old man evaluated for haematuria and diagnosed with
bladder carcinoma (white arrow). Incidentally noted on sagittal contrast enhanced
CT angiography is a kinked proximal celiac artery with a hooked
contour and associated post-stenotic dilatation.
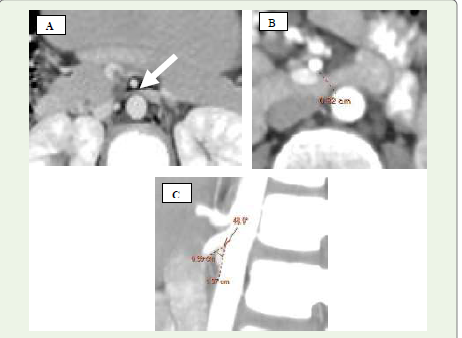
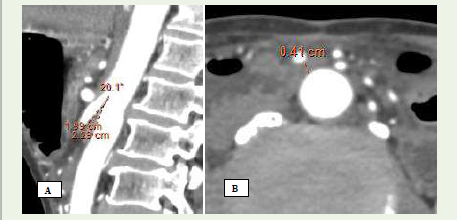
Figure 3 A-C: A 17-year-old male presenting with swelling in the left
scrotal region. (A) Axial contrast-enhanced CT image demonstrates the
characteristic “beak sign” (white solid arrow) due to narrowing of the left
renal vein. (B) Axial CT showing a reduced aortomesenteric distance (AMD)
measuring approximately 8 mm. (C) Sagittal contrast-enhanced CT reveals
a sharply decreased aortomesenteric angle (AMA) of around 48°.
Figure 4 A, B: (A) Coronal contrast-enhanced CT image displays a
prominently dilated left testicular vein (black arrow). (B) Axial contrast-enhanced
CT scan of the scrotum reveals a cluster of tortuous, enhancing
veins within the left scrotal sac (white solid arrow), consistent with a left-sided
varicocele.
Figure 5 Axial contrast-enhanced CT of a 33-year-old woman demonstrates
a retro-aortic course of the left renal vein (white arrow), an incidental finding
consistent with the posterior variant of Nutcracker Syndrome.
to anaemia, orthostatic proteinuria, and pain in the left flank due to
passage of clots. Reflux into the gonadal vein may lead to left-sided
varicocele in males and varicose veins in the pelvis or vulva in females
[1].
CT imaging findings:
On axial CT, the “beak sign” is seen as abrupt narrowing of the
LRV at the point where it passes between the SMA and aorta, forming
an acute angle [Figure 3A] [11,12]. Sagittal reconstructions help assess the aortomesenteric angle (AMA), which normally ranges from 38°–
56°; angles under 35° are suggestive of the syndrome [13,14]. Affected
individuals also demonstrate a reduced aortomesenteric distance
(AMD), typically narrowed from the normal 10–28 mm to 2–8 mm
[1,15] [Figure 3B], [Figure 3C]. A pre- to post-compression LRV diameter ratio exceeding 2.25 has been shown to yield 91% sensitivity
and specificity for diagnosis [16]. CT may also reveal dilated gonadal
veins and pelvic varicosities, although it cannot assess blood flow
velocity or direction [11] [Figure 4A], [Figure 4B].Management:
Management of mild or asymptomatic cases is conservative,
focusing on observation. More severe cases require intervention to
relieve venous obstruction and hypertension. Surgical options include
LRV transposition, external venous stenting, bypass procedures,
renal auto-transplantation, or nephrectomy [1].Superior Mesenteric Artery Syndrome:
Superior Mesenteric Artery (SMA) syndrome, first documented
by Rokitansky in 1842, results from compression of the third part of
the duodenum between the SMA and the aorta, leading to duodenal
obstruction [1,13].A hallmark diagnostic feature of SMA syndrome is the narrowing of the angle between the SMA and the aorta, which normally measures between 38° and 65°. In affected individuals, this angle is significantly reduced. Likewise, the aortomesenteric distance at the level where the duodenum crosses is typically reduced from its normal range of 10–28 mm [13,17,18].
Clinical features:
Patients typically show signs of upper gastrointestinal obstruction
such as abdominal pain following meals, nausea, vomiting, and
unintended weight loss—symptoms linked to delayed gastric
emptying. Other potential causes must be excluded, and diagnosis
relies on identifying the anatomical abnormality using imaging
studies [19].Imaging findings:
On CT, the AMA is decreased (6°–22°) and the AMD reduced
(2–8 mm) [Figure 7A], [Figure 7B]. Characteristic findings include
gastric and proximal duodenal dilation [Figure 6A], [Figure 6B]
with abrupt narrowing at the compression site. CT helps localize the
obstruction and exclude other causes. Importantly, decreased AMA
and AMD without symptoms do not suffice for diagnosis [20,21,22].Management:
The first-line approach involves conservative therapy, including
fluid resuscitation, electrolyte correction, and nutritional support often
through nasojejunal feeding. Patients are advised to consume
small, frequent meals and may benefit from positional adjustments
like lying prone or in the left lateral decubitus position. If nonsurgical
methods fail, operative options such as duodenojejunostomy,
gastrojejunostomy, or division of the ligament of Treitz with bowel
derotation (Strong’s procedure) may be considered [1,3,21].Superior mesentery artery-like syndrome:
SMA-like syndrome is a newly described condition that mimics
the presentation of SMA syndrome but results from compression of
the duodenum by vascular structures other than the SMA and aorta.
These may include mesenteric or retroperitoneal arteries and veins
[23].Clinical features:
This syndrome clinically mimics SMA syndrome and carries
similar risks if untreated [23].Imaging findings:
On imaging, compression of the third portion of the duodenum
may be observed between multiple anterior (mesenteric) and posterior
vascular structures. Anatomical variations in the suspensory ligament
of the duodenum may contribute to this phenomenon. CT scans may
reveal either duodenal compression leading to obstruction [Figure 8A], [Figure 8B] or isolated vascular compression without evidence
Figure 6 A, B: A 76-year-old man presenting with postprandial abdominal
discomfort, vomiting, and a history of weight loss. (A) Sagittal contrast-enhanced
CT of the abdomen shows a markedly distended stomach (white
arrow). (B) Axial contrast-enhanced CT angiogram reveals dilatation of the
first and second portions of the duodenum (yellow arrow) due to compression
of the third part by the superior mesenteric artery and aorta.
Figure 7 A and B: (A) Sagittal contrast-enhanced CT demonstrates a
sharply reduced angle between the superior mesenteric artery and aorta
(AMA ~ 20°). (B) Axial contrast-enhanced CT image shows a decreased
aortomesenteric distance (AMD) measuring approximately 4 mm.
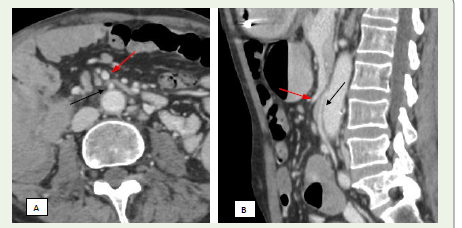
Figure 8 A,B: A 50-year-old man presenting with abdominal pain and
vomiting. (A & B) Axial and sagittal contrast-enhanced CT images of the
abdomen show compression of the third portion of the duodenum by the
ileocolic vein (red arrow) and the inferior mesenteric artery (black arrow),
resulting in gastric overdistension due to stagnant intraluminal contents—
findings consistent with SMA-like Syndrome.
of obstruction (Figure 9A) (Figure 9B). CT imaging is particularly
helpful in identifying the precise location of the duodenum and
ruling out alternative causes of obstruction [21,24].
Management:
Management begins conservatively, focusing on nutritional
support and hyperalimentation to restore retroperitoneal fat.
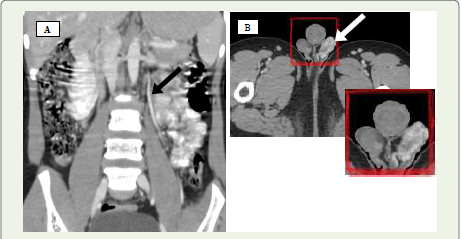
Figure 9 A,B: A 29-year-old woman with complaints of indigestion, vomiting,
and weight loss. (A) Coronal contrast-enhanced CT scan of the abdomen
demonstrates a duplicated inferior vena cava (IVC) on the left side (black
arrow). (B) Contrast-enhanced axial CT of abdomen shows compression of
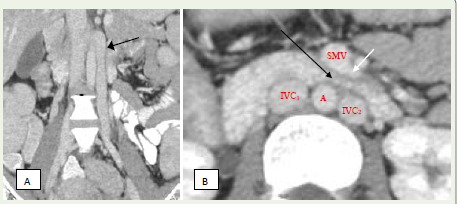
3rd part of duodenum (black arrow) by aorta (A), duplicated IVC (IVC2) and
SMV & its jejunal tributary (black arrow) at the level of L3 vertebra with no
signs of bowel obstruction—SMA-Like Syndrome.
Surgery (gastrojejunostomy or duodenojejunostomy) is considered if
symptoms persist [23,25].
Vascular Compression of the Ureter—Retrocaval Ureter:
The ureter may rarely follow a retrovascular path, such as retroiliac
or retrocaval routes, which can lead to compression [26]. Retrocaval
ureter is a congenital anomaly caused by abnormal embryological
development of the inferior vena cava (IVC) [27]. Under typical
development, the intrarenal segment of the IVC originates from the
right supracardinal vein, which lies dorsal to the ureter, maintaining
the ureter’s lateral position. However, in retrocaval ureter, the ventral
subcardinal vein fails to regress, causing the ureter to pass posterior
to and encircle the IVC during embryogenesis. This aberrant course
results in its compression between the IVC and vertebral column,
eventually leading to progressive hydronephrosis [28].Clinical features:
Clinically, patients often report right flank pain, although many
remain asymptomatic [28].Imaging findings:
Imaging distinguishes two types:Type 1: The ureter crosses behind the IVC near L3 with a characteristic “S” or “fish-hook” deformity and hydronephrosis in over 50% of casesalso known as the low loop retrocaval ureter [Figure 10] [Figure 11]
Type 2: The retrocaval segment crosses at the renal pelvis with a sickle-shaped curve and mild hydronephrosis, which is less common [29].
Management:
Management is tailored based on symptom intensity and renal
function status. For mild, asymptomatic cases, watchful waiting
may be appropriate. In symptomatic individuals, surgical correction
through ureteroureteral reanastomosis anterior to the IVC—along
with removal of the retrocaval segment—is commonly performed
and typically produces favorable results [30].
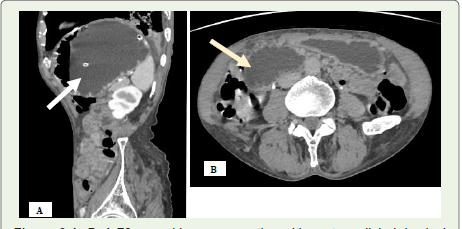
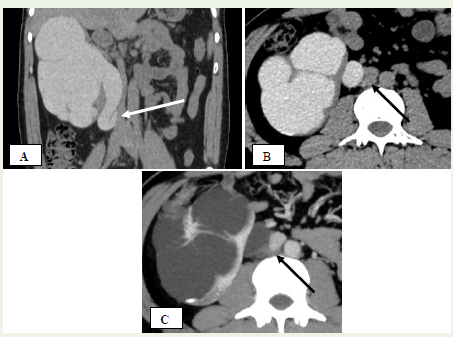
Figure 10 A, B, C: A 26-year-old man with repeated episodes of right flank
pain. (A) Coronal reconstructed image from the excretory phase of contrast-enhanced
CT IVU reveals right-sided hydronephrosis and a characteristic
‘fish-hook’ appearance of the upper ureter caused by its retrocaval path
(white arrow). (B) and (C) Axial CT images during excretory and nephrogenic
phases show the right ureter coursing posterior to the IVC (black arrow).
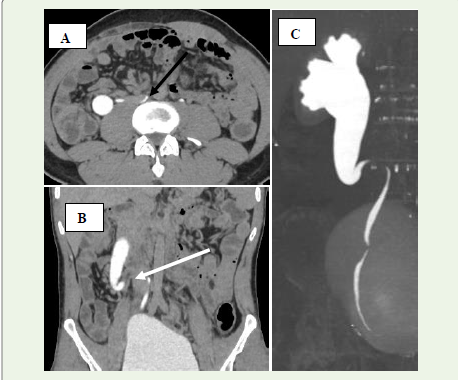
Figure 11 A 28-year-old man presenting with recurrent right flank pain.
(A) Axial CT image during the excretory phase demonstrates the retrocaval
trajectory of the right ureter (black arrow). (B) Coronal reconstructed image from
the excretory phase reveals hydronephrosis with the upper ureter forming a ‘fishhook’
configuration due to its retrocaval route (white arrow). (C) A 3D-rendered
CT IVU image highlights the same finding—right-sided hydronephrosis and
‘fish-hook’ shaped ureter consistent with a retrocaval course
Portal Biliopathy:
Portal biliopathy refers to a collection of abnormalities affecting
the bile ducts and gallbladder, typically seen in individuals with
extrahepatic portal vein obstruction or portal hypertension [31].The condition arises primarily through two mechanisms: first, by direct mechanical pressure on the biliary tract from a portal cavernoma [Figure 12] and second, through peribiliary fibrosis triggered by inflammation or ischemia following thrombosis in the
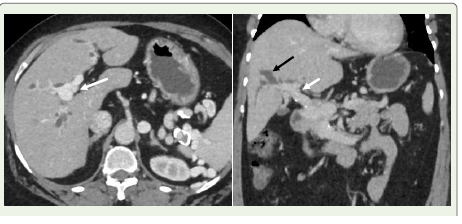
Figure 12:A 55-year-old woman diagnosed with portal hypertension. (A) Axial
contrast-enhanced CT image reveals cavernous transformation of the portal
vein (white arrow). (B) Coronal contrast-enhanced CT scan shows dilated
biliary ducts (black arrow) secondary to external compression from the portal
cavernoma (white arrow), indicative of portal biliopathy.
small venous channels within the bile duct wall [4,9,32]. A portal
cavernoma is a network of dilated collateral veins surrounding the
common bile duct, resulting from cavernous transformation. It is
composed of paracholedochal veins (also known as the plexus of
Petren) and epicholedochal veins (plexus of Saint), both of which run
along the bile duct wall [33].
There are three main types of portal biliopathy: varicoid, fibrotic,
and mixed. The varicoid form is due to compression and distortion
of the bile duct by large external collaterals (paracholedochal veins),
while the fibrotic form is associated with thickening and narrowing
of the bile duct caused by intramural compression from smaller
collateral veins (epicholedochal veins) [4].
Clinical features:
Around 70–100% of individuals who exhibit radiologic features
of portal biliopathy do not initially experience symptoms [32]. Only a
small subset presents with clinical signs such as prolonged cholestasis
or jaundice [4,9,31].Imaging findings:
Contrast-enhanced CT is typically performed to rule out
alternative causes of biliary dilation, especially since portal biliopathy
can resemble malignant distal bile duct strictures, such as those from
pancreatic cancer or cholangiocarcinoma [34]. The most characteristic
radiologic sign, apart from detecting a portal cavernoma, is the
abrupt bending or “kinking” of the common bile duct, usually due to
external pressure from enlarged paracholedochal veins [35].Management:
While the majority of cases are asymptomatic and do not need
active intervention, patients who develop symptoms—especially
from biliary strictures—can be treated effectively using endoscopic
approaches such as balloon dilatation, stenting, or performing a
sphincterotomy [36].Conclusion
Although often underrecognized, abdominal vascular syndromes
constitute an important and frequently overlooked clinical entity.
Their presentations can vary widely—from subtle, nonspecific
symptoms to severe and obvious clinical signs—necessitating
increased awareness and a collaborative, multidisciplinary approach
to diagnosis and management. Advances in imaging modalities,
particularly high-resolution CT and magnetic resonance angiography
(MRA), have improved early detection, thereby facilitating timely
intervention and enhancing patient outcomes. Despite these
technological improvements, abdominal vascular syndromes are still
commonly misdiagnosed or diagnosed late, due to their complex
clinical manifestations and overlap with more prevalent abdominal
diseases. Clinicians and radiologists alike should consider these
entities in the differential diagnosis when assessing patients with
unexplained abdominal pain, weight loss, or similar symptoms.
A thorough grasp of the underlying vascular anatomy, associated
pathophysiological mechanisms, and key imaging findings is essential
for ensuring effective patient care.
Disclosure: The authors have no conflicts of interest to disclose.
None of the authors received outside funding for the production of
this original manuscript and no part of this article has been previously
published elsewhere.
References
Citation
Akhil MK, Pranavi K, Suhasini VR. Compressed and Overlooked: A Radiologic-Clinical Overview of Abdominal Vascular Compression Syndromes. Indian J Appl Radiol. 2025;11(1): 216.